Germain Hess and the Enthalpy of Doom
DOFPro Team

Introduction
- The enthalpy descriptions associated with different sorts of reactions
- How you use those for calculating
- First-law relationships
- Properties of reactions
First Law for a Reactor
If we have a chemical reaction, such as
\[ a\mathrm{A} + b\mathrm{B} \rightarrow c\mathrm{C} + d\mathrm{D} \]
we can run it in an isothermal chemical reactor, either as an open steady-state system or as a closed constant-pressure system.
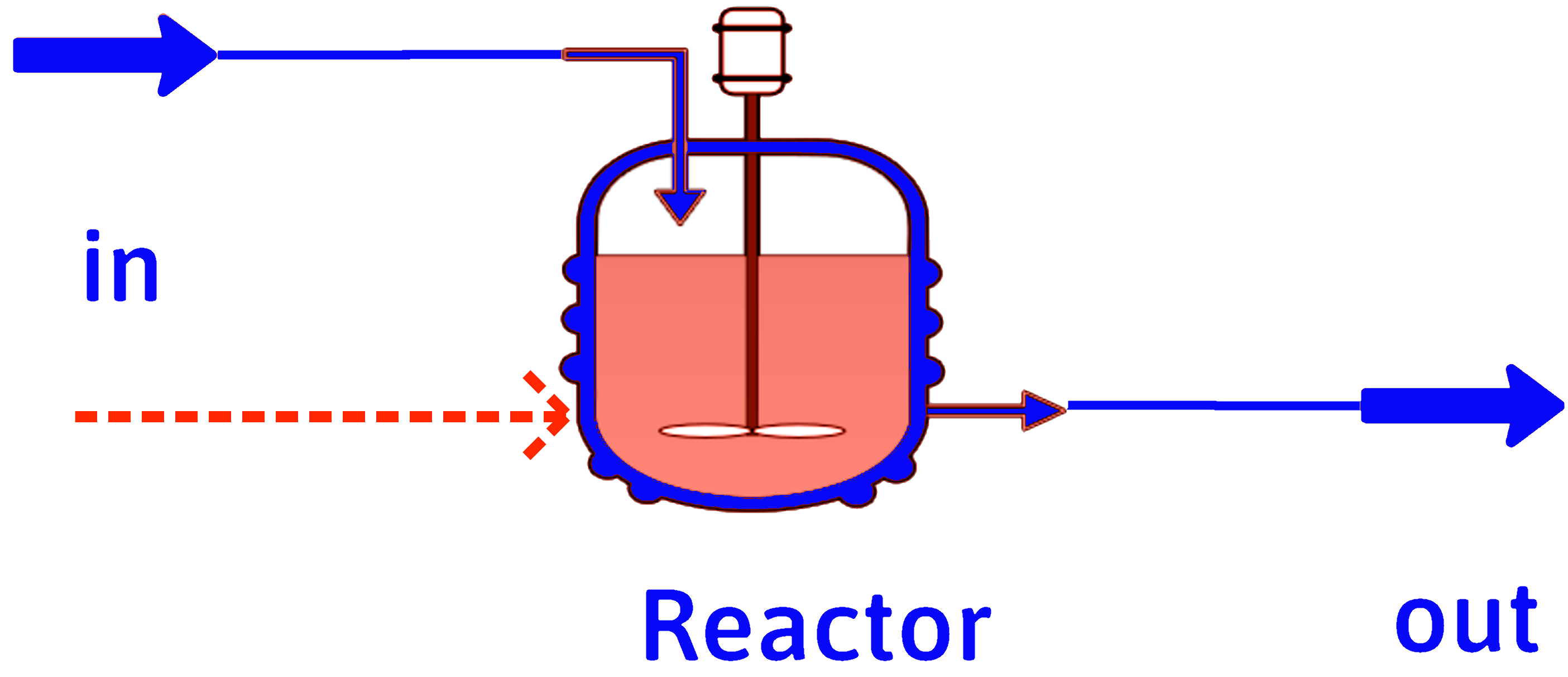
\(T_\mathrm{in}\)
\(T_\mathrm{out} = T_\mathrm{in}\)
\(a\mathrm{A} + b\mathrm{B} \rightarrow c\mathrm{C} + d\mathrm{D}\)
\(Q\text{ or }\dot{Q}\)
\[ \Delta \hat{H}_r = \dfrac{\Delta H}{\xi} = \dfrac{Q}{\xi} \text{ or } \dfrac{\Delta \dot{H}}{\dot{\xi}} = \dfrac{\dot{Q}}{\dot{\xi}} \]
The enthalpy of reaction or heat of reaction depends on how you write the chemical equation. If you double the stoichiometric coefficients, you double the heat of reaction.
The heat of reaction is a function of temperature. At the standard reference temperature (\(25\ ^{\circ}\text{C}\) for the DOFPro videos), the heat of reaction is the standard heat of reaction. Wikipedia has reasonable table of heats of combustion, which are heats of reaction for burning fuels in air. They are discussed further later in the video.
To calculate the heat of reaction at a temperature other than the standard temperature, you have to take into account any sensible heat changes and any phase changes since \(H\) is a state function.
Heat of Formation
The enthalpy of formation or heat of formation is the standard heat of reaction for forming one mole of product, starting from pure elements at their standard form of aggregation at \(T_\mathrm{ref}\). For example, the standard states at \(25\text{ } ^{\circ}\text{C}\) for oxygen and nitrogen are the diatomic gases \(\mathrm{O_2 (g)}\) and \(\mathrm{N_2 (g)}\). The standard heat of formation for any element in its standard form of aggregation is \(0\) by definition. Wikipedia has a reasonably good table of standard enthalpies of formation.
You can calculate the standard heat of reaction for any reaction as the sum of the heats of formation times the stoichiometric coefficients. Don’t forget phase changes.
\[ \Delta \hat{H}^{\circ}_r = \sum \nu_i \Delta \hat{H}^{\circ}_{f, i} \]
Hess’s Law
The energetics of any reaction can be calculated by algebraically adding together any set of reactions that gives you the same overall reaction as the one you want.
Example
\(\mathrm{C_2H_6} + \dfrac{7}{2} \mathrm{O_2} \rightarrow 2\mathrm{CO_2} + 3\mathrm{H_2O}: \hspace{10pt}\Delta \hat{H}^{\circ}_{r1} = -1559.8\ \mathrm{kJ/mol}\)
\(\mathrm{C} + \mathrm{O_2} \rightarrow \mathrm{CO_2}: \hspace{10pt}\Delta \hat{H}^{\circ}_{r2} = -393.5\ \mathrm{kJ/mol}\)
\(\mathrm{H_2} + \dfrac{1}{2} \mathrm{O_2} \rightarrow \mathrm{H_2O}: \hspace{10pt}\Delta \hat{H}^{\circ}_{r3} = -285.8\ \mathrm{kJ/mol}\)
Add \(2\) Equation \(2\)’s to \(3\) Equation \(3\)’s and subtract \(1\) Equation \(1\)
\[ 2 \mathrm{C} + 3\mathrm{H_2} + 2\mathrm{O_2} + \dfrac{3}{2} \mathrm{O_2} - \dfrac{7}{2} \mathrm{O_2} \rightarrow 2\mathrm{CO_2} + 3\mathrm{H_2O} - 2\mathrm{CO_2} - 3\mathrm{H_2O} + \mathrm{C_2H_6} \]
- \(\hspace{10pt} 2\mathrm{C} + 3\mathrm{H}_2 \rightarrow \mathrm{C}_2 \mathrm{H}_6\)
\[ \Delta \hat{H}^{\circ}_{r4} = 2\Delta \hat{H}^{\circ}_{r2} + 3\Delta \hat{H}^{\circ}_{r3} - \Delta \hat{H}^{\circ}_{r1} \]
\[ = 2(-393.5) + 3(-285.8) - (-1559.8) = -84.6 \mathrm{kJ/mol} \]
which is also the heat of formation for ethane.
If you use different sets of equations to calculate the same final equation with Hess’s law, you may find that rounding and truncation errors keep the results from being identical. The most current value for the enthalpy of formation of ethane from NIST is \(–84. \pm0.4\ \mathrm{kJ/mol}\), which almost encompasses the value we calculated.
The Heat of Combustion
If you have a combustion reaction for a given species, as discussed in the Flame On, Torch! video, the heat of reaction for complete combustion of one mole of fuel is called the heat of combustion. As a reminder, for the principal species and complete combustion, the combustion products are \(\mathrm{C}\mathrm{O}_2\), \(\mathrm{H}_2\mathrm{O}\), and \(\mathrm{S}\mathrm{O}_2\). \(\mathrm{C}\mathrm{O}_2\) and \(\mathrm{S}\mathrm{O}_2\) are gases at standard conditions. What about water?
The value called the enthalpy of combustion assumes that water is liquid at \(25\ ^{\circ}\mathrm{C}\) and \(1\ \mathrm{atm}\). It is alternately called the Higher Heating Value (HHV). The enthalpy of combustion calculated assuming water is vapor at \(25\ ^{\circ}\mathrm{C}\) is called the Lower Heating Value. The details will be discussed in Fueling the Fire: The Shocking Truth About Combustion Reactions!
The Takeaways
- The standard heat of formation is the standard heat of reaction for forming one mole of product, starting from pure elements at their standard form of aggregation at \(T_\mathrm{ref}.\)
- The heat of reaction is the sum of the heats of formation of the individual species times their stoichiometric coefficients. Don’t forget phase changes.
- Hess’s law states that the energetics of a reaction is the algebraic sum of the energetics of any set of constituent reactions.
- The heat of combustion is the heat of reaction for a combustion reaction.
Thanks for watching!
The previous video in the series is in the link in the upper left. The next video in the series is in the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.
The DOFPro Team

