Fueling the Fire: The Shocking Truth About Combustion Reactions!
DOFPro Team

Combustion Reactions
Combustion reactions are rapid oxidation reactions. One usually burns a fuel (typically coal, oil or natural gas) in air (or rarely pure oxygen). The principal atomic species in combustion reactions are carbon, hydrogen, sulfur and oxygen.
Solid Fuels: Principally coal, coke, wood, charcoal, and solid waste.
Liquid Fuels: Principally hydrocarbons and alcohols.
Gaseous Fuels: Principally natural gas.
Coal
Coal is really nasty stuff. There is no other way to put it.
| Component | Percentage |
|---|---|
| carbon [\(\mathrm{C}\)] | 75–90 |
| hydrogen [\(\mathrm{H}\)] | 4.5–5.5 |
| nitrogen [\(\mathrm{N}\)] | 1–1.5 |
| sulfur [\(\mathrm{S}\)] | 1–2 |
| oxygen \(\mathrm{[O}\)] | 5–20 |
| ash | 2–10 |
| moisture [\(\mathrm{H_2O}\)] | 1–10 |
http://www.chemistryexplained.com/Ce-Co/Coal.html
| Component | Bituminous | Subbituminous | Lignite |
|---|---|---|---|
| SiO2 | 20–60 | 40–60 | 15–45 |
| Al2O3 | 5–35 | 20–30 | 10–25 |
| Fe2O3 | 10–40 | 4–10 | 4–15 |
| CaO | 1–12 | 5–30 | 15–40 |
| MgO | 0–5 | 1–6 | 3–10 |
| SO3 | 0–4 | 0–2 | 0–10 |
| Na2O | 0–4 | 0–2 | 0–6 |
| K2O | 0–3 | 0–4 | 0–4 |
| LOI | 0–15 | 0–3 | 0–5 |
https://www.fhwa.dot.gov/publications/research/infrastructure/structures/97148/cfa51.cfm
Trace Elements in Coal Fly Ash
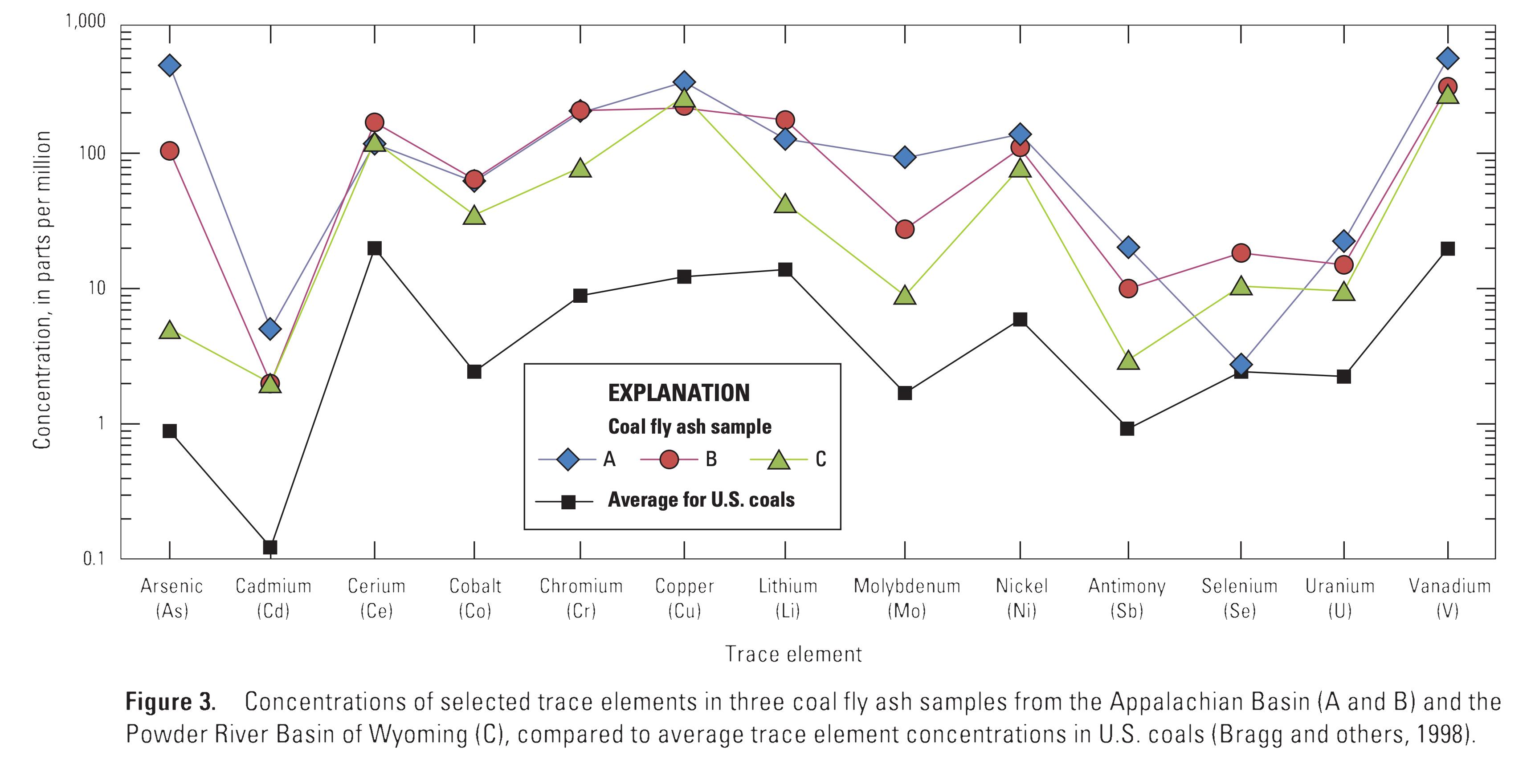
https://pubs.usgs.gov/fs/2015/3037/pdf/fs2015-3037.pdf
Heating Value
The heating value of a fuel is the negative of its heat of combustion. There are two types
Higher Heating Value (HHV): Assumes the final state of the water is liquid (same as for heat of combustion).
Lower Heating Value (LHV): Assumes the final state of the water is vapor.
\[ HHV = LHV + n_\mathrm{H_2O} \Delta \hat{H}_v\ (\mathrm{H_2O},\ 25\ ^\circ\mathrm{C}) \]
\[ HV_\mathrm{mix} = \sum x_i HV_i \]
Adiabatic Flame Temperature
If we operate a combustor such that we extract no heat from the combustor, all of the enthalpy of reaction has to go into heating the combustion products.
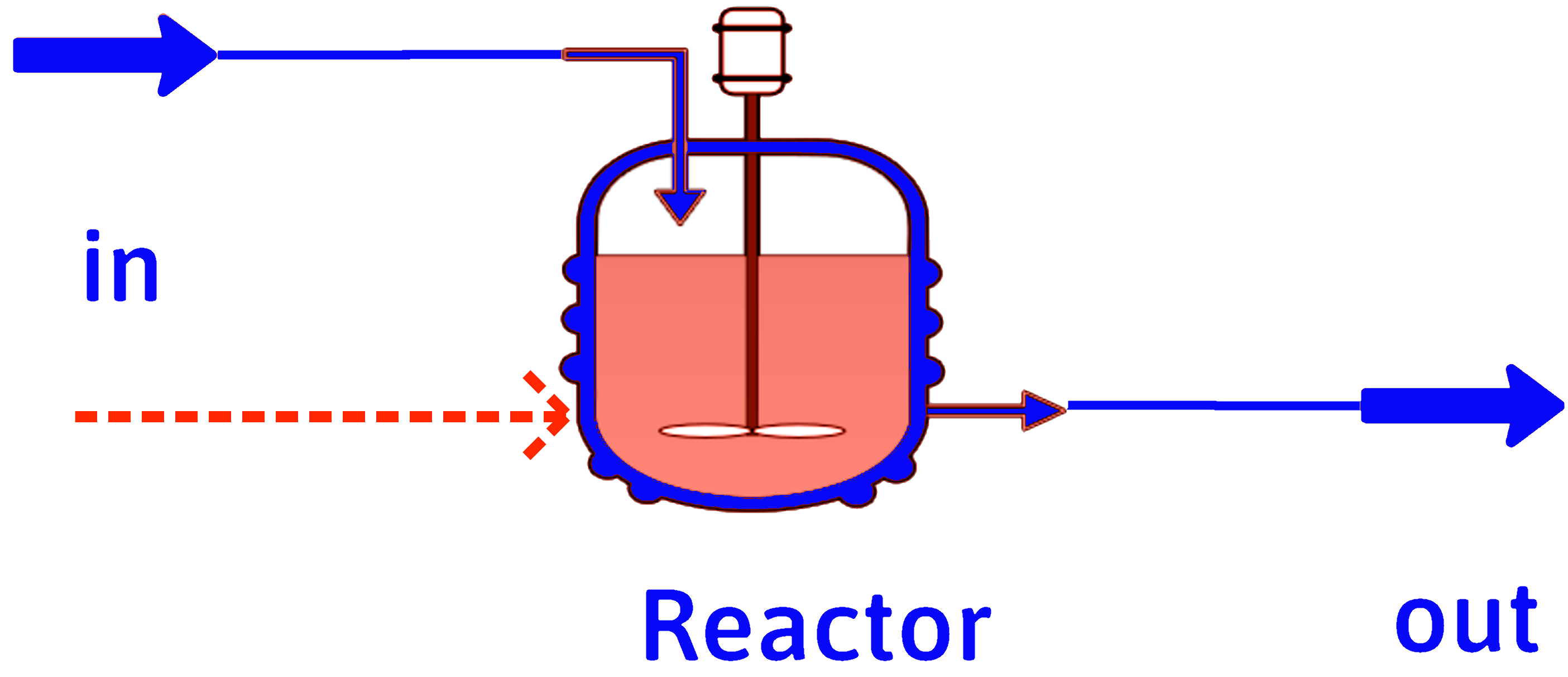
\(T_\mathrm{in}\)
\(T_\mathrm{out} = T_\mathrm{af}\)
\(Q\text{ or }\dot{Q} = 0\)
\(\Delta H\text{ or }\Delta \dot{H} = 0\)
\[ \sum_\mathrm{out} \dot{n}_j \int\limits_{T_\mathrm{ref}}^{T_\mathrm{out} = T_\mathrm{af}} C_{pj} dT - \sum_\mathrm{in} \dot{n}_i \int\limits_{T_\mathrm{ref}}^{T_\mathrm{in}} C_{pi} dT + \sum_\mathrm{rxns} \dot{\xi}_k(\Delta \hat{H}_r^\circ)_k = \dot{Q} = 0 \]
The adiabatic flame temperature is the maximum temperature a fuel-air mixture can reach when it burns. The maximum adiabatic flame temperature occurs when burning with stoichiometric air. To be useable, some heat has to be extracted from the burning mixture, which lowers the maximum temperature.
Many of the \(C_p\) formulas found in the literature do not go to a high enough temperature range to permit calculation of \(T_\mathrm{af}\) for near-stoichiometric fuel-air mixtures.
To process materials at higher temperatures than adiabatic flame temperatures, some other technique such as electromagnetic, or laser heating must be used.
Flammability Limits
For fuel-air mixtures that are either too fuel lean or too fuel rich, the oxidation reaction does not generate enough heat to sustain a flame or reaction. The limits of fuel-air mixtures are called the flammability limits.
The Flash Point is the temperature above which the vapor pressure is high enough to create a flammable mixture.
The Autoignition Temperature is the temperature above which a fuel-air mixture will combust without an additional source of spark or flame.
| Species | LFL/LEL in vol. % | UFL/UEL in vol. % | Flash Point | Autoignition temperature |
|---|---|---|---|---|
| Acetone | 2.6–3 | 12.8–13 | −17 °C | 465 °C, 485 °C |
| Benzene | 1.2 | 7.8 | −11 °C | 560 °C |
| n-butane | 1.6 | 8.4 | −60 °C | 420–500 °C |
| CO | 12[6] | 75 | Fl. gas | 609 °C |
| Diesel fuel | 0.6 | 7.5 | >62 °C | 210 °C |
| Ethanol | 3–3.3 | 19 | 12.8 °C | 365 °C |
| Gasoline | 1.4 | 7.6 | <−40 °C | 246–280 °C |
| Hydrogen | 4/18.3[25] | 75/59 | Fl. gas | 500–571 °C |
| Isopropanol | 2[6] | 12 | 12 °C | 398–399 °C |
| Jet A-1 fuel | 0.6–0.7 | 4.9–5 | >38 °C | 210 °C |
| Methane | 5.0 | 14.3 | Fl. gas | 580 °C |
| Propylene | 2 | 11.1 | −108 °C | 458 °C |
| Veg. oil | 327 °C |
The Takeaways
- We have added the energetics to the stoichiometry of combustion reactions.
- Coal is a particularly dirty fuel. It takes extreme efforts to burn it cleanly
- The Higher Heating Value (HHV) is the heat of combustion when liquid water is assumed as a combustion product.
- The Lower Heating Value (LHV) is the heat of combustion when water vapor is assumed as a combustion product.
- The adiabatic flame temperature is the highest temperature a stoichiometric fuel-air mixture can reach when burned.
- For safety reasons, always check your flammability limits.
Thanks for watching!
The previous video in the series is in the link in the upper left. The next video in the series is in the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.
The DOFPro Team

