React Harder: Formation Heats and Alternate Endings
DOFPro Team

Introduction
React Hard: The First Law Gets Chemical and
React Harder: Formation Heats and Alternate Endings
present
- three sets of calculations for the 1st Law in reacting systems.
- based on A Combustible Mixture! video
- Review React Hard: The First Law Gets Chemical before watching this video.
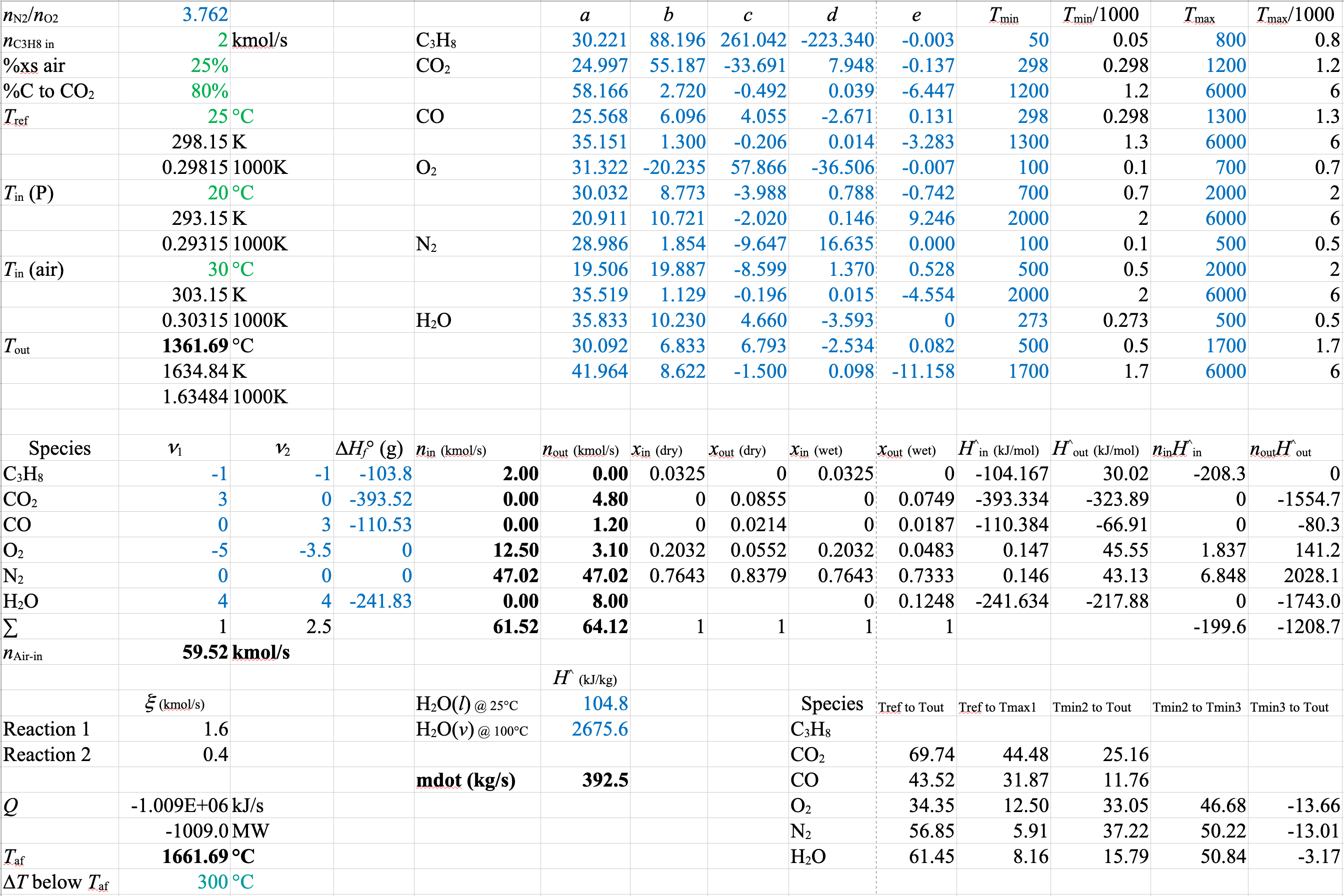
Energetics for Equations 3 and 4
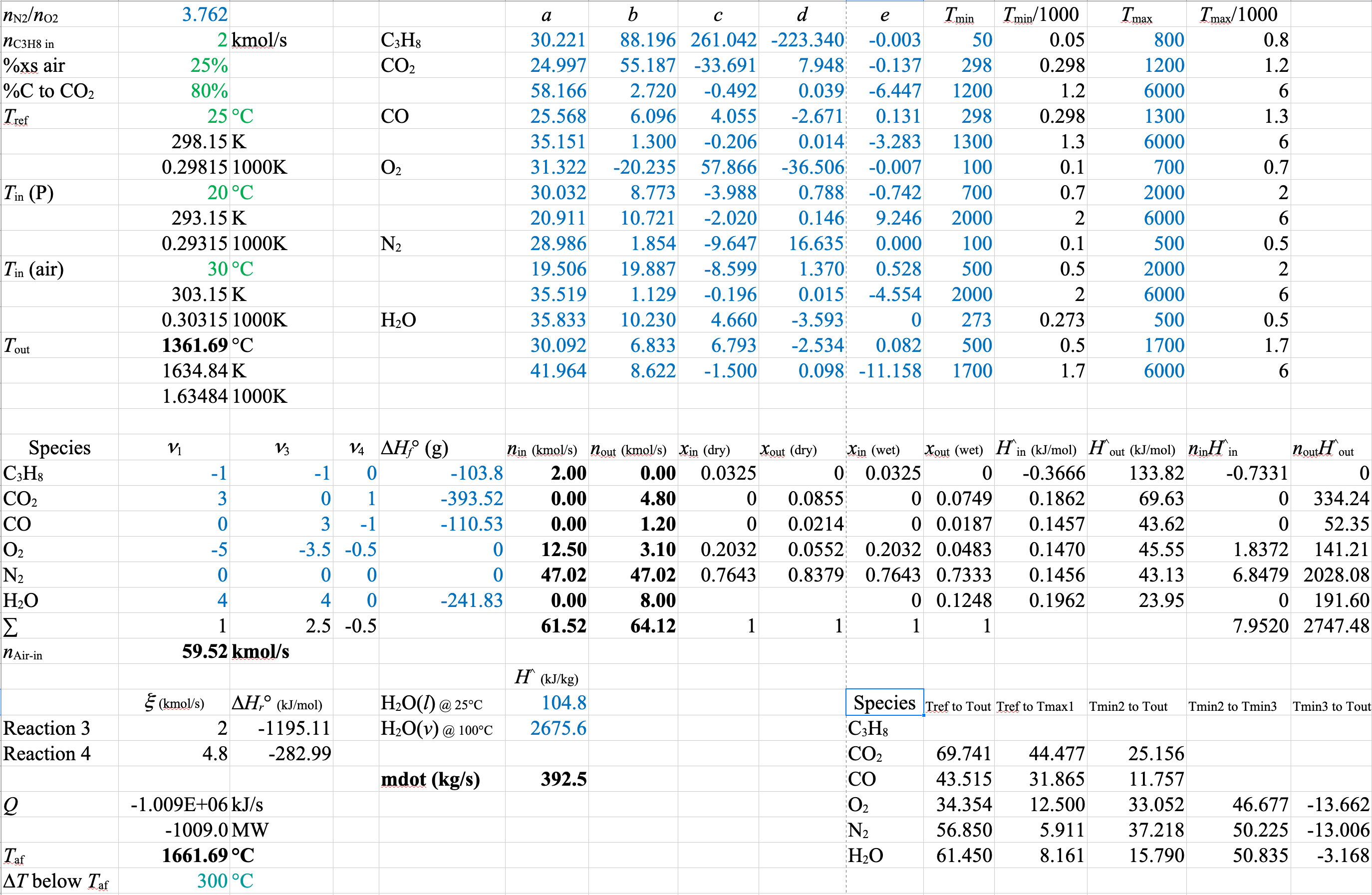
Heat of Formation Method
\[\sum\limits_{\substack{\mathrm{out} \\ \mathrm{streams}}} \dot{n}_j \hat{H}_j\ - \sum\limits_{\substack{\mathrm{in} \\ \mathrm{streams}}} \dot{n}_i \hat{H}_i = \dot{Q},\hspace{120px} \hat{H}_i = \Delta \hat{H}^{\circ}_{f, i} + \int\limits_{T_\mathrm{ref}}^T C_{pi} (T) dT \]
Which Method?
- All three gave identical results.
- The heat of reaction method requires you to calculate the heats of reaction.
- The heats of formation method requires you to add the heats of formation to the specified species specific enthalpies.
- The net effort appears to be about the same.
I personally usually use the heat of reaction method because I like to see what the reactions are contributing to the overall energy balance compared to what the sensible heat changes are contributing.
- Either method works.
- Implement your chosen method correctly.
The Takeaways
- All three ways of working the problem gave the same answers.
- Sometimes you have to do piecewise integration on your heat capacity equations.
- The method you choose depends on your personal preference.
Thanks for watching!
The Full Story companion video is in the link in the upper left. The companion video in the series, Flame On, Torch!, is in the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.

