Steam: Get the Quality Right AFTER You Table It! Part 1
DOFPro Team

Gibbs Phase Rule
\(\ \ \ \ \ \boxed{DF\ =\ 2+c - \Pi}\)
- A Degree of Freedom, \(DF\), is a coordinate or variable.
- A Component, \(c\), is a chemical species, e.g. \(\mathrm{Al_2O_3}\).
- A Phase, \(\Pi\), is a physical entity with a uniform composition, temperature, pressure, and specific volume.
- For a single component system with one phase \(DF = 2 + 1 - 1 = 2\).
- For a single component system with two phases \(DF = 2 + 1 - 2 = 1\).
Steam-Water \(T\text{-}\hat{S}\) Diagram
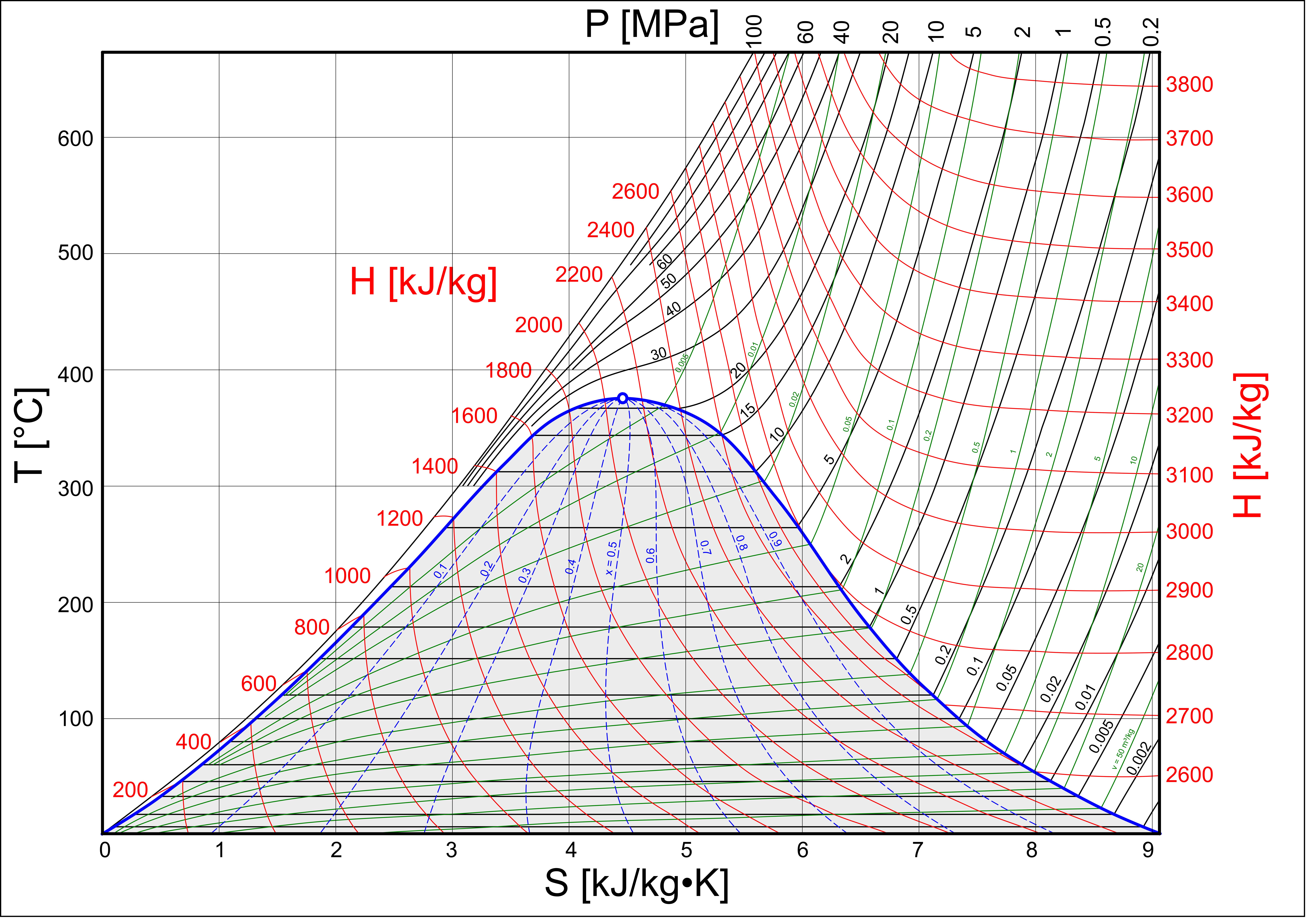
Kaboldy, T-s diagram, Font and spacing, CC BY-SA 3.0
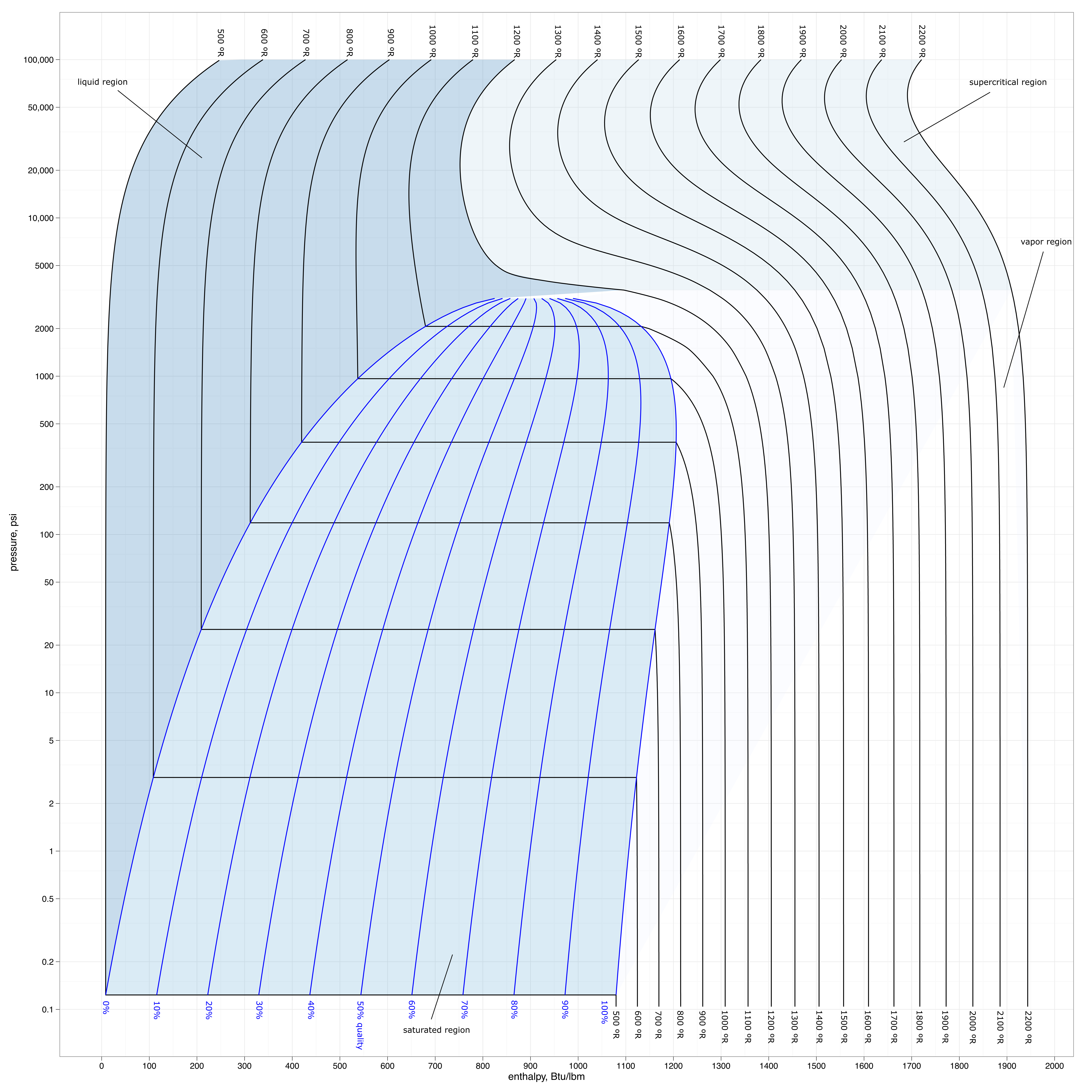
Steam-Water \(P\text{-}\hat{H}\) Diagram











The Takeaways
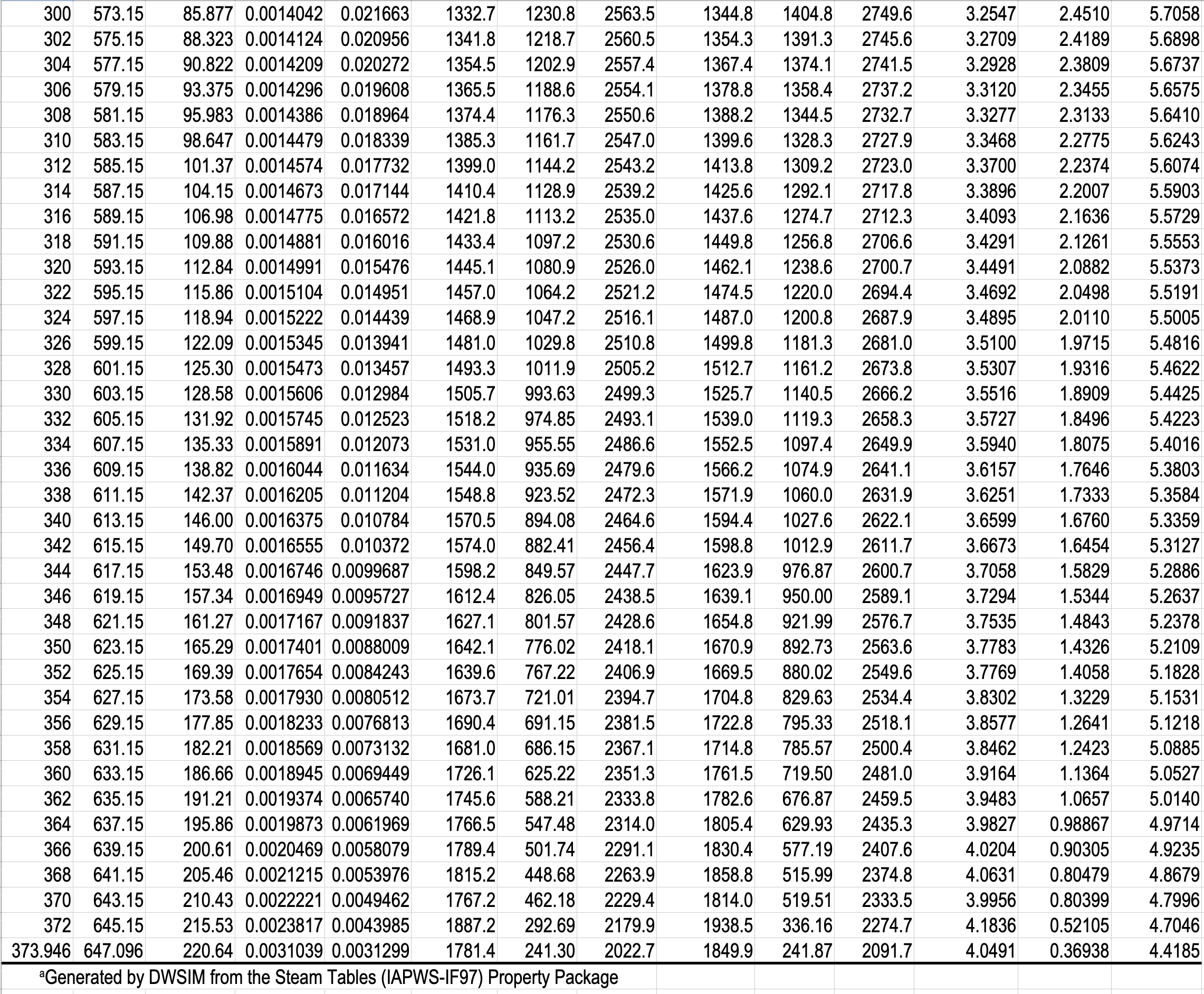
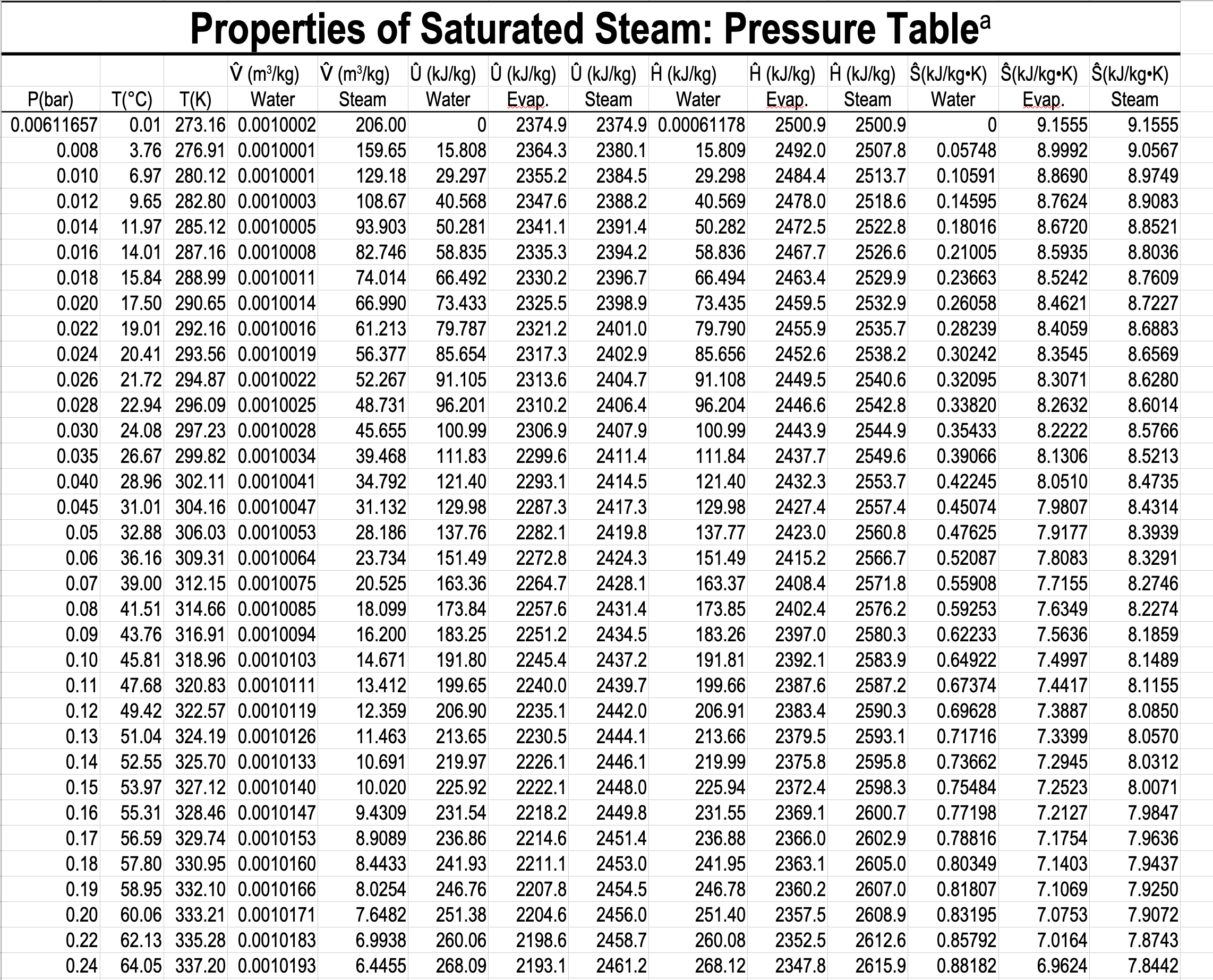
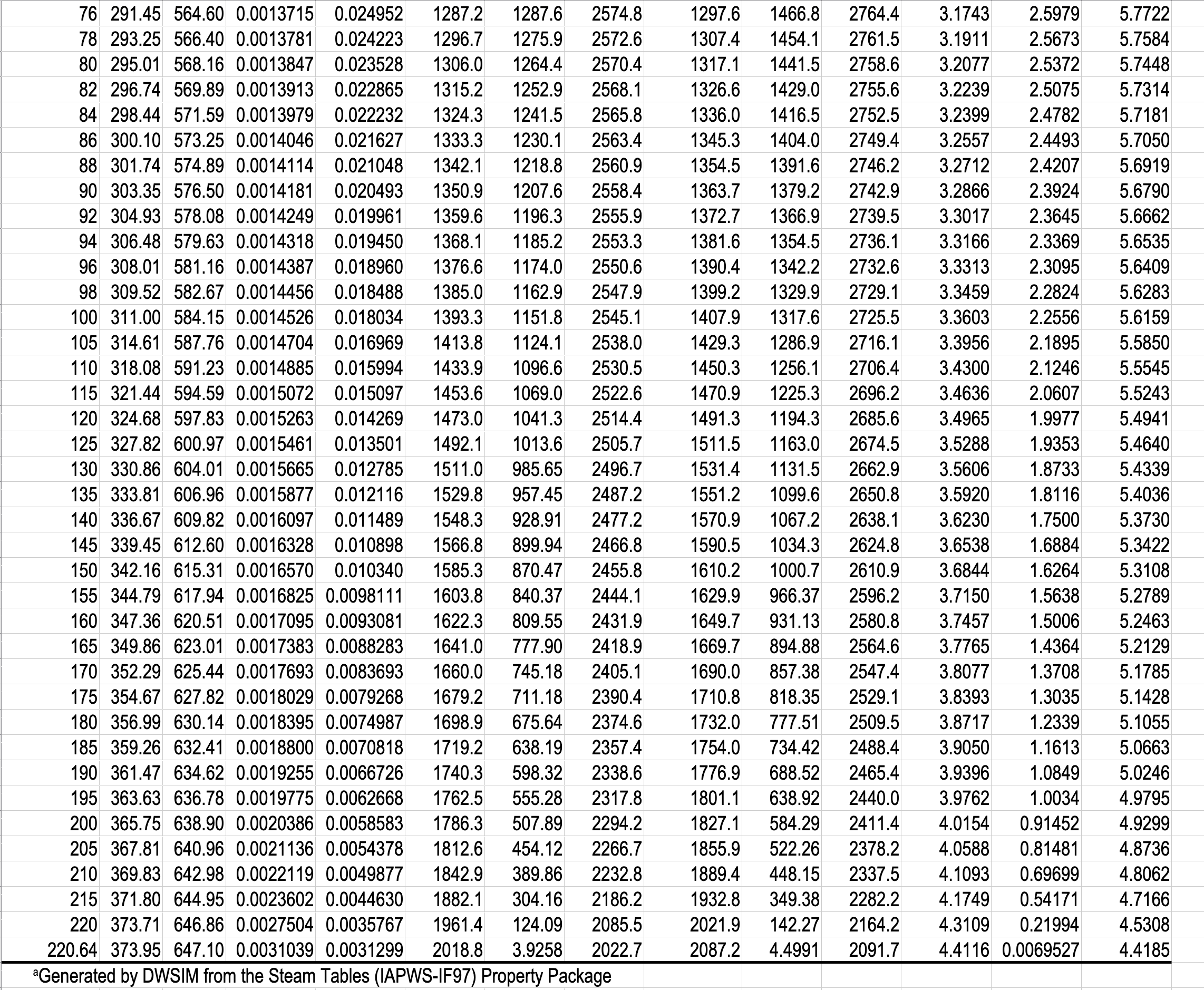
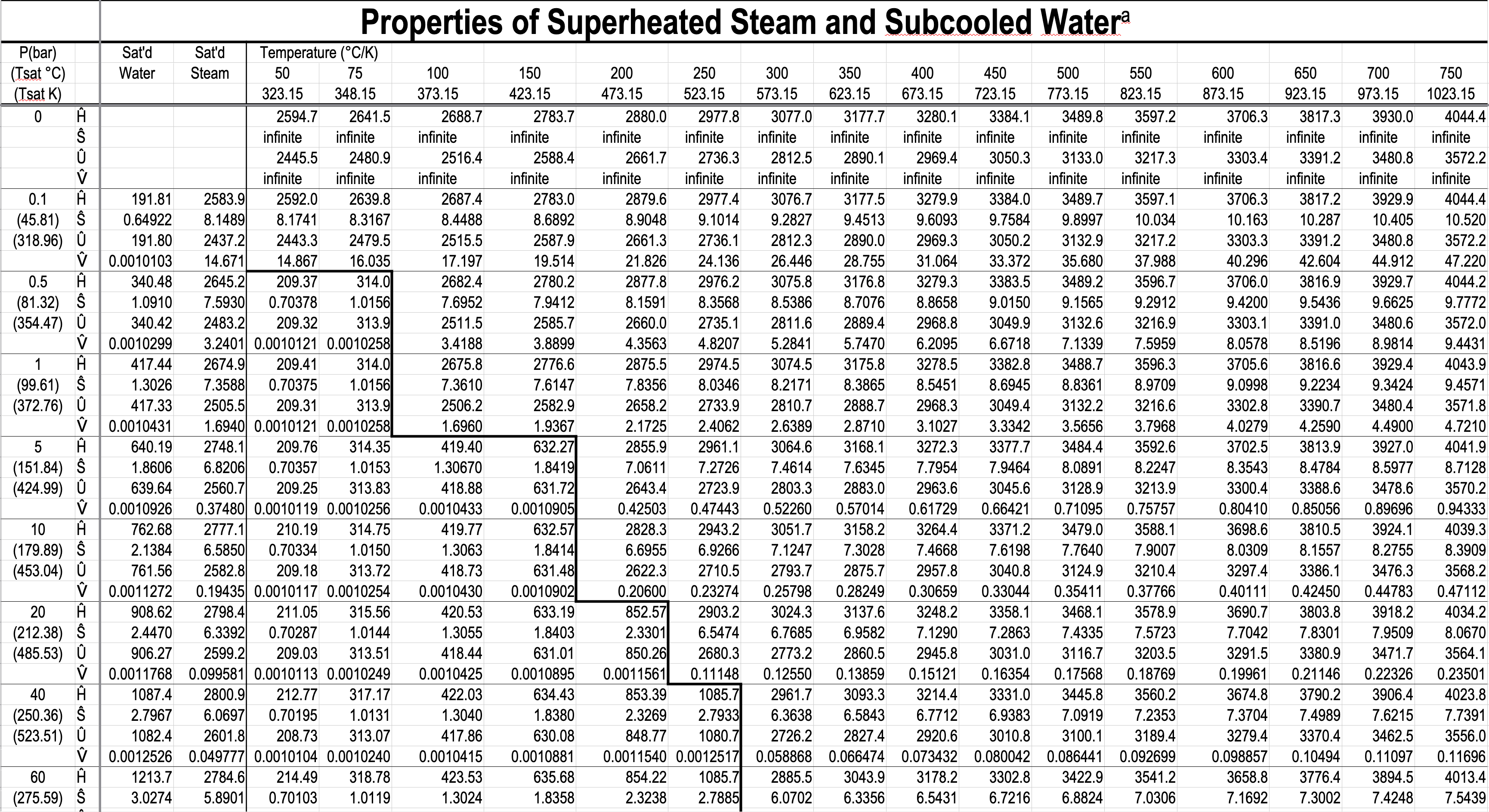
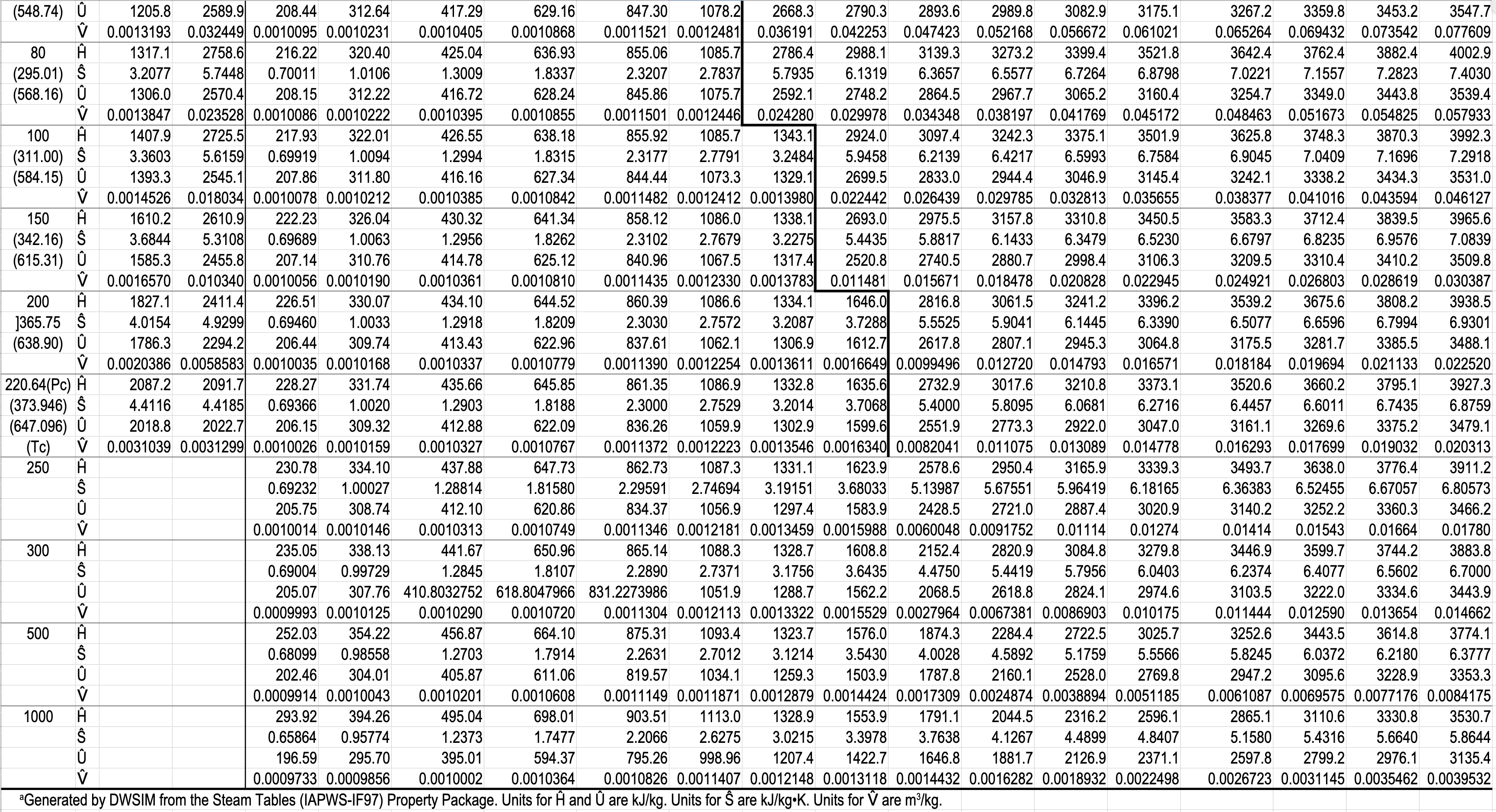
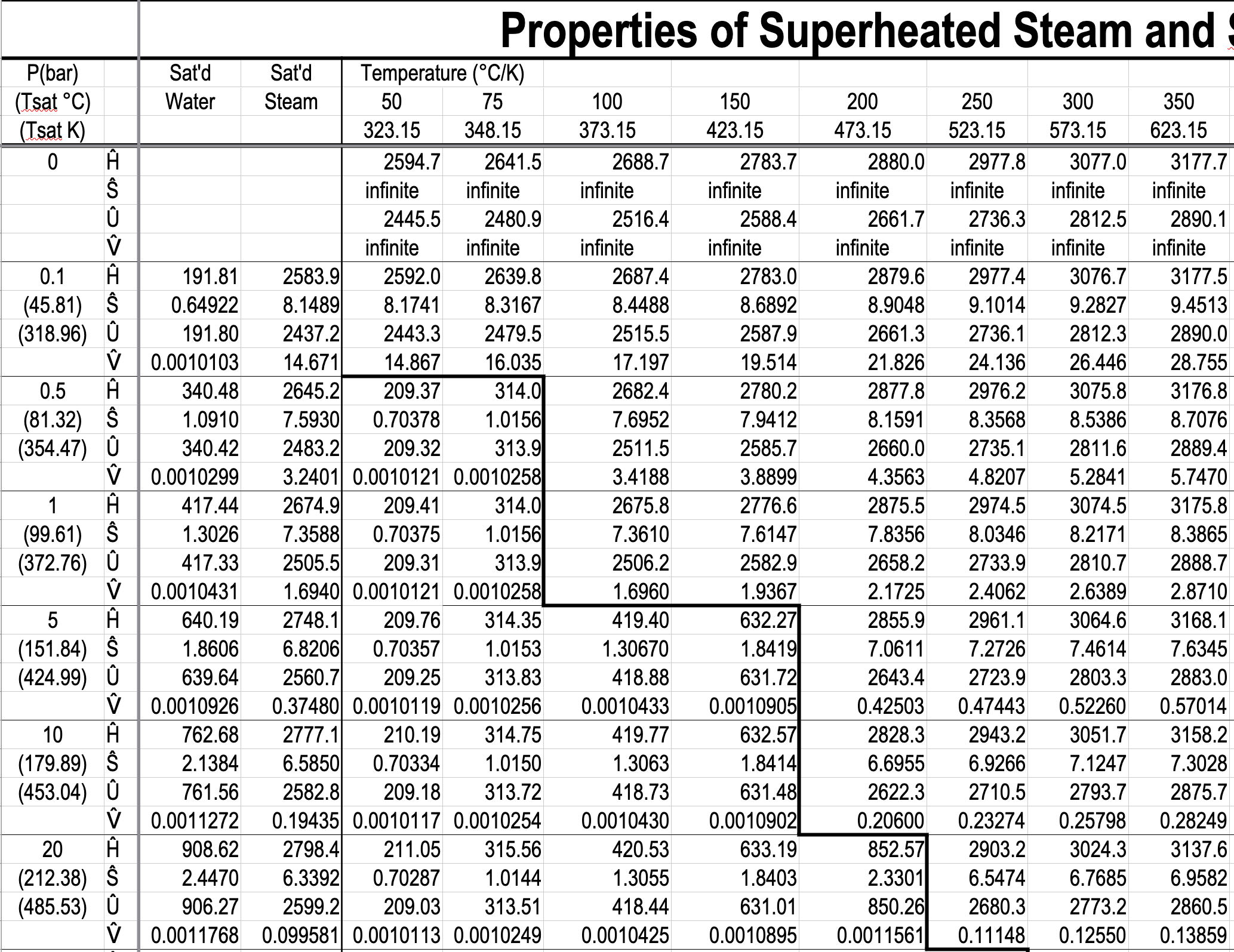
- Gibbs phase rule dictates how thermodynamic tables like the steam tables are arranged.
- The Temperature Table and the Pressure Table have the same information in them. The choice is based on whether you have the temperature or the pressure of the two-phase mixture.
- The Superheated Steam and Subcooled Water Table has single-phase regions for both subcooled water and superheated steam.
- You have to specify a single property in the two-phase tables, but you have to specify two properties in the single-phase table.
Thanks for watching!
The previous video in the series is in the link in the upper left. The next video in the series is in the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.
The DOFPro Team

