Steam: Get the Quality Right AFTER You Table It! Part 2
DOFPro Team

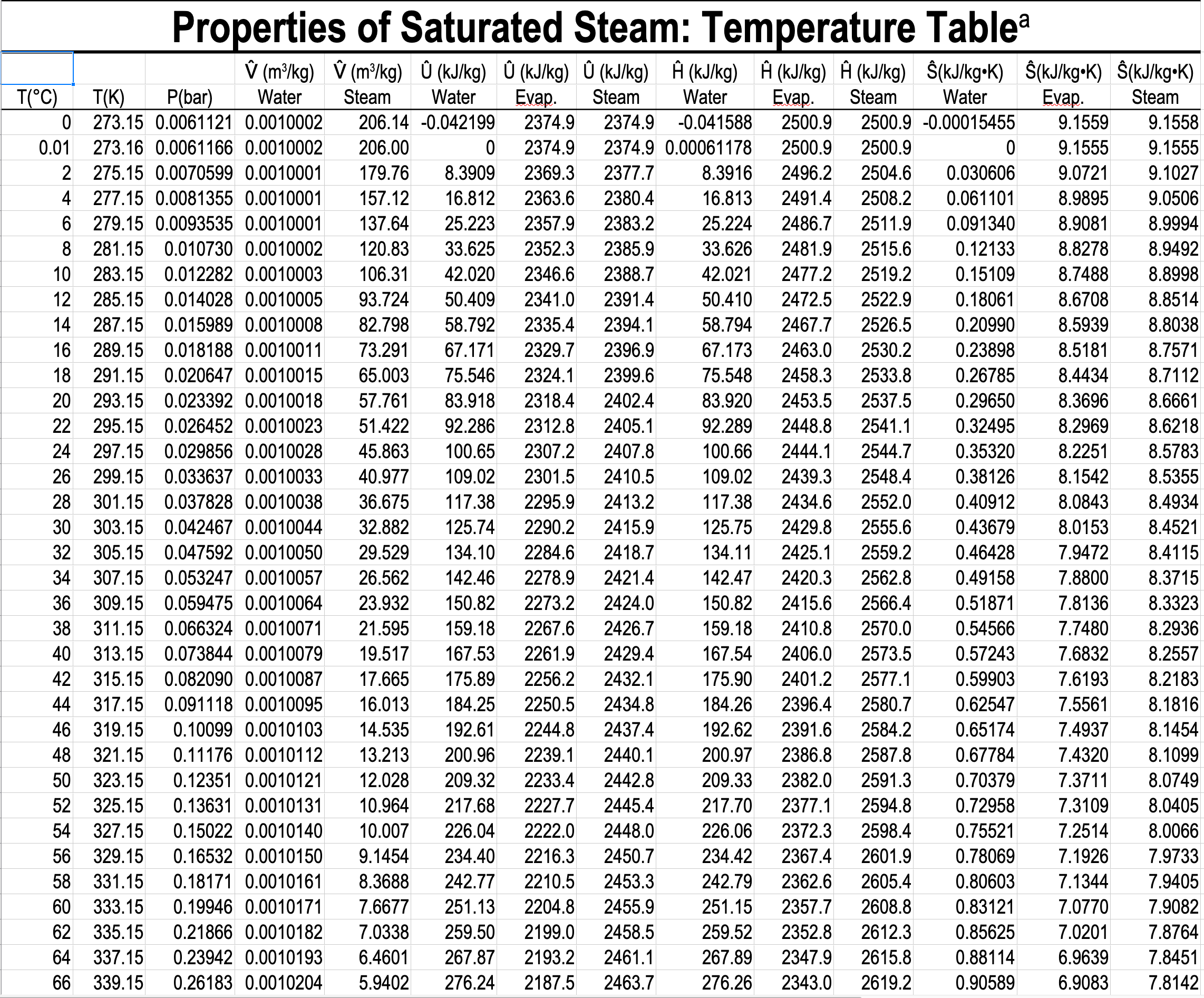
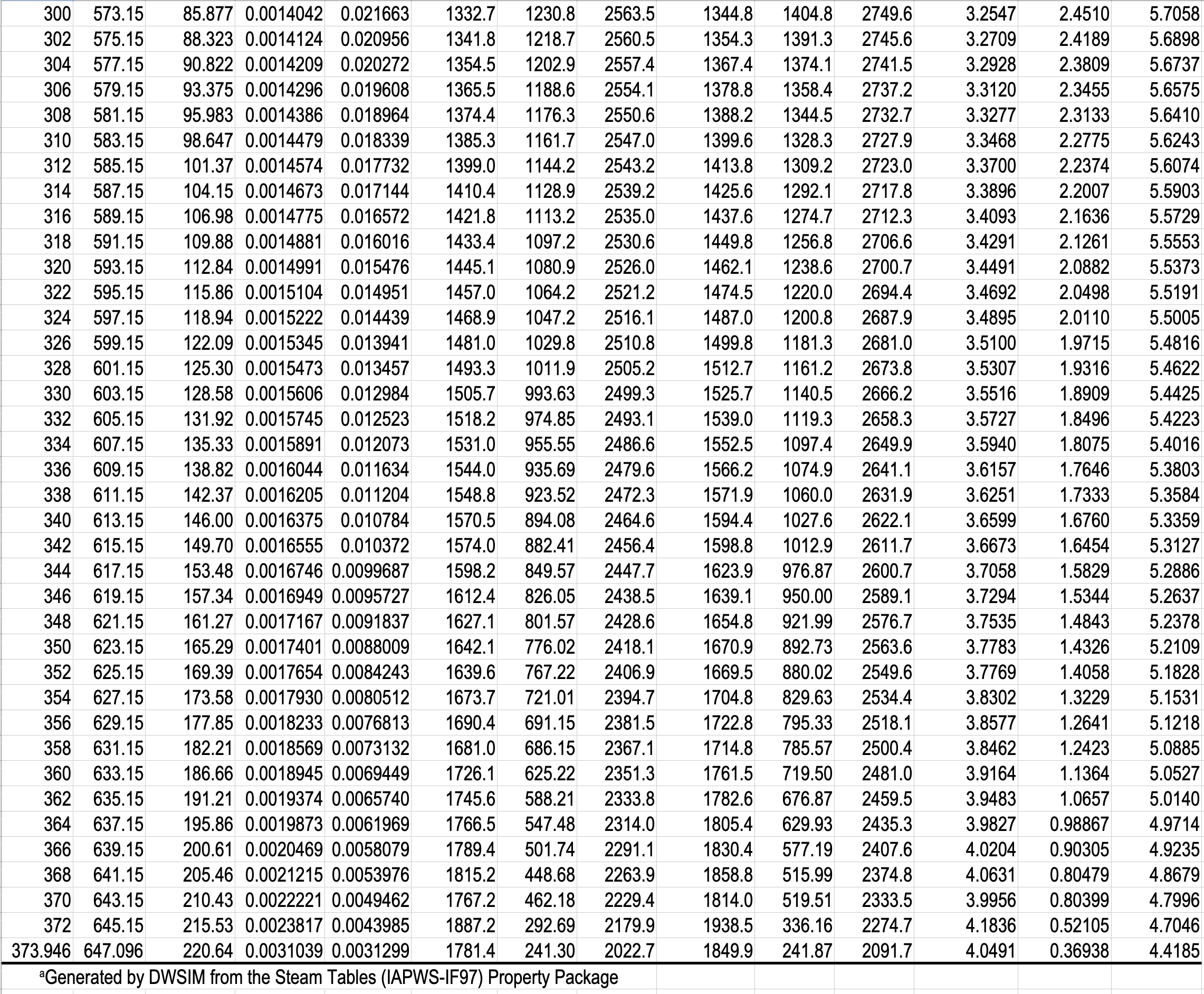
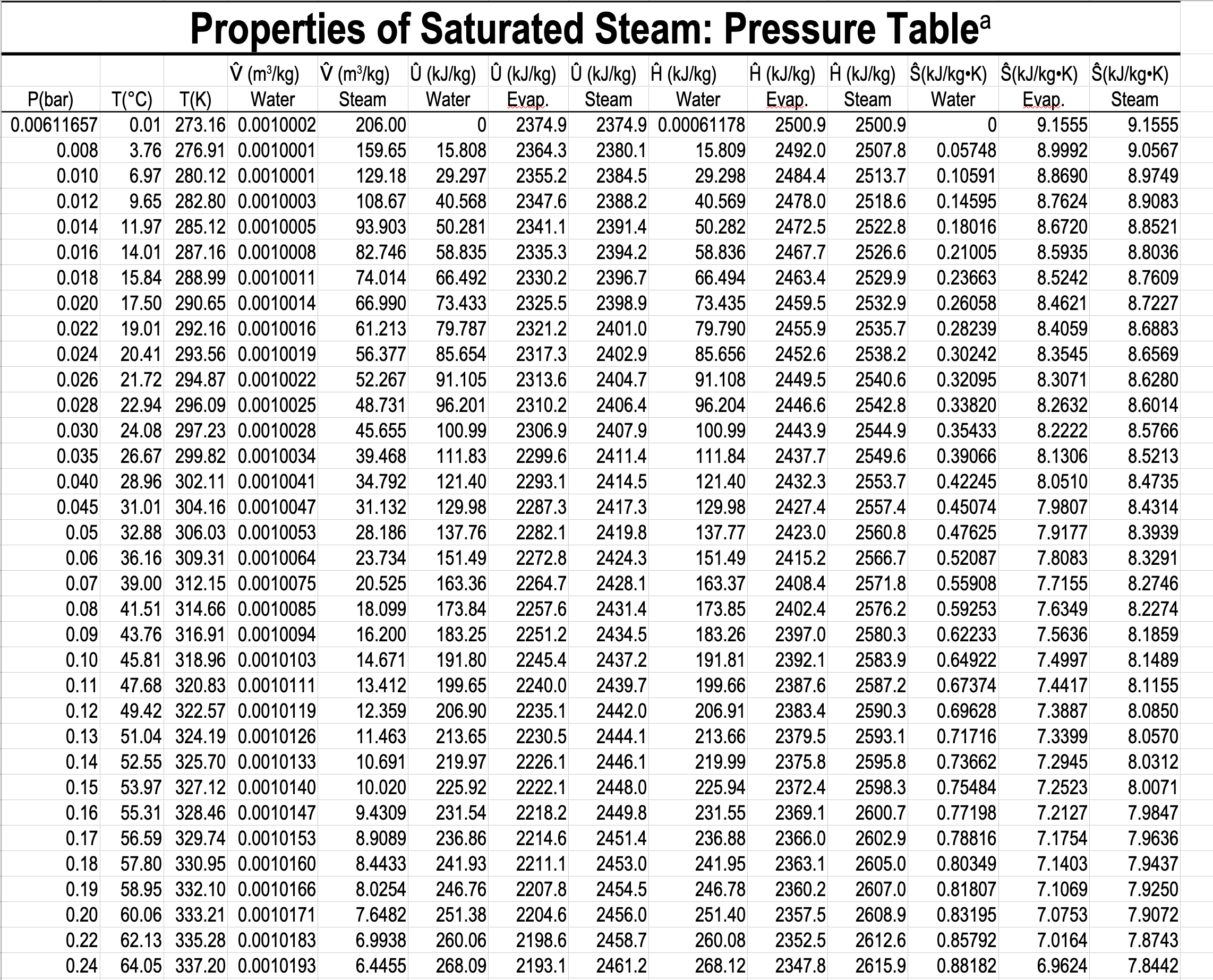
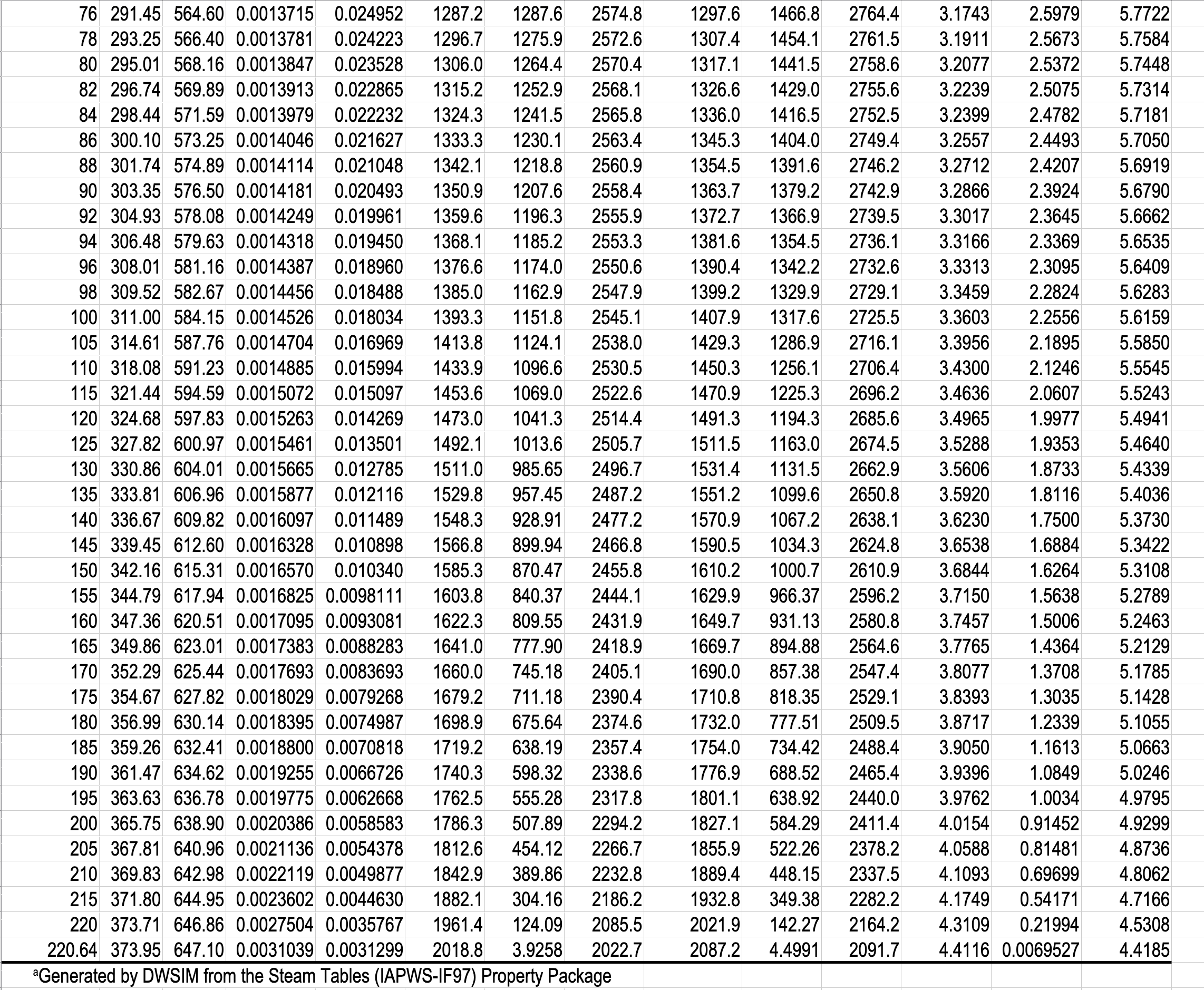
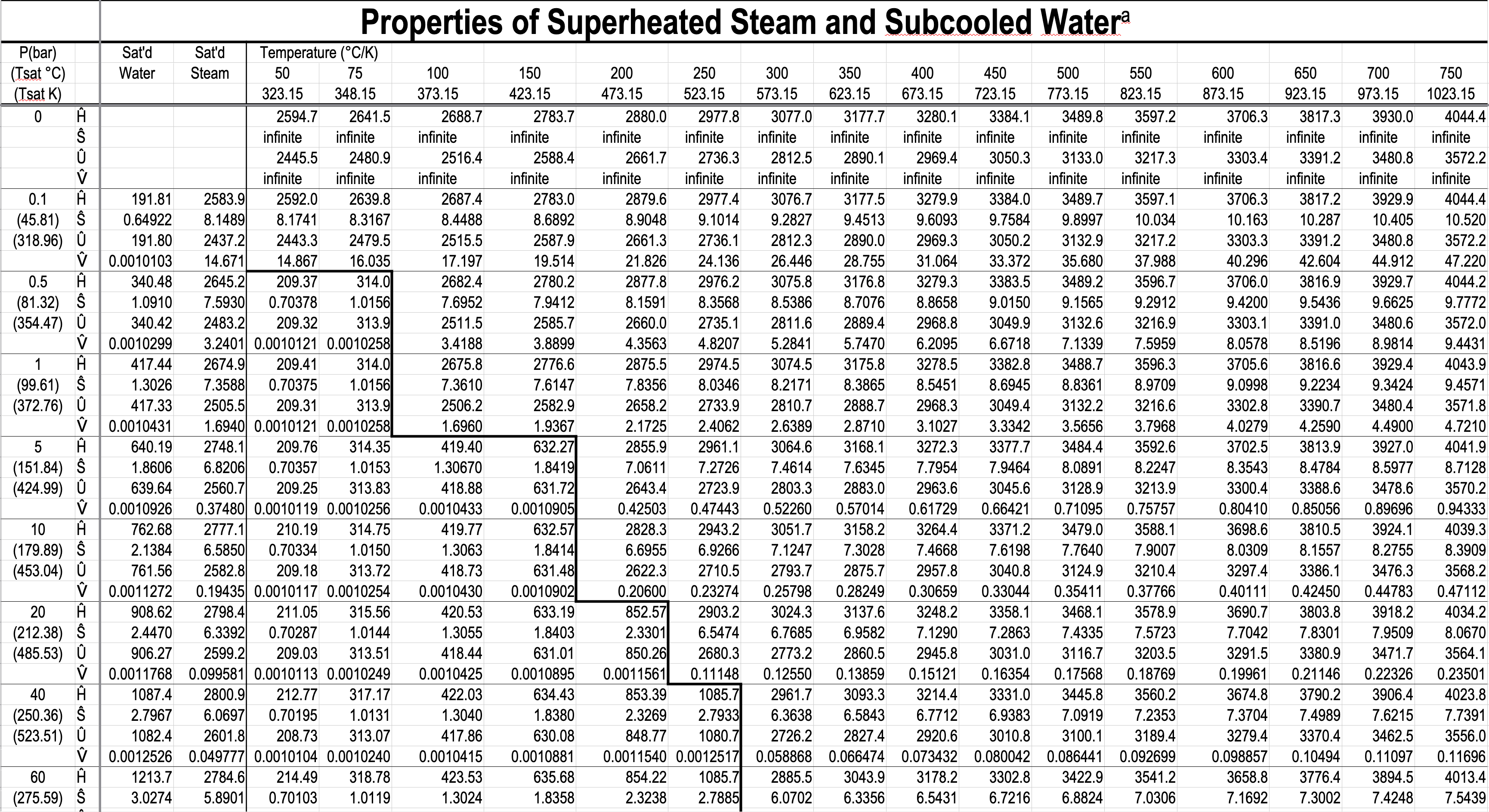
In the single-phase region (superheated steam)
\(\ \ \ \ DF = 2\)
Must specify two properties to define the thermodynamic state
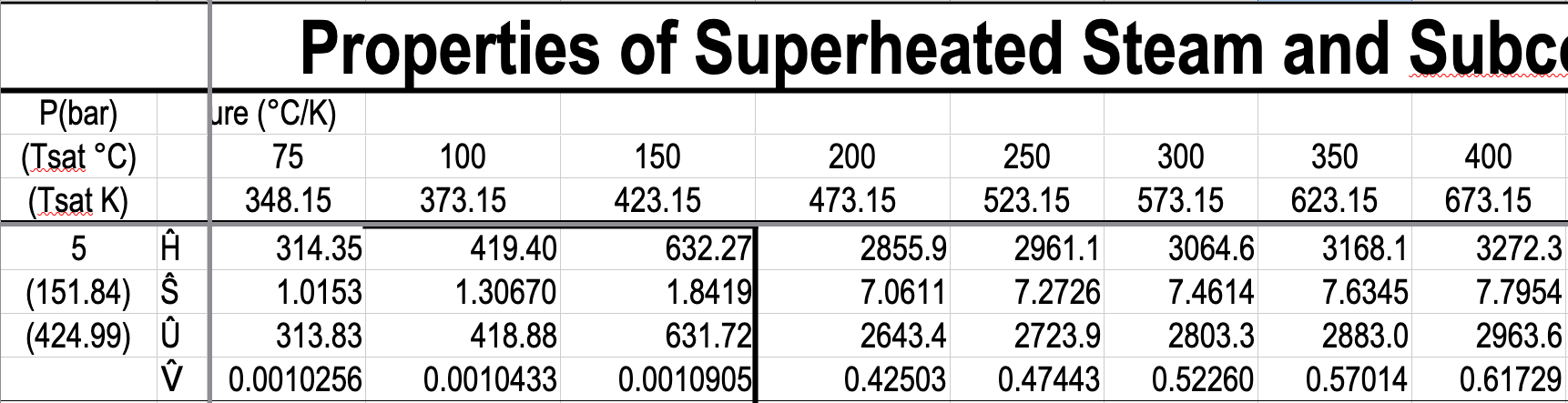
e.g. What is the specific volume of steam at \(5\ \mathrm{bar}\) and \(300\ ^\circ\mathrm{C}\)?
e.g. What is the temperature of steam with a specific volume of \(0.50000\) \(\dfrac{\textrm{m}^3}{\textrm{kg}}\) at \(5\) bar?
\(\dfrac{T - 250}{0.50000 - 0.47443}\ =\ \dfrac{300 - 250}{0.52260-0.47443} \hspace{5pt} \Rightarrow T = 276.54 ^{\circ} \mathrm{C}\)
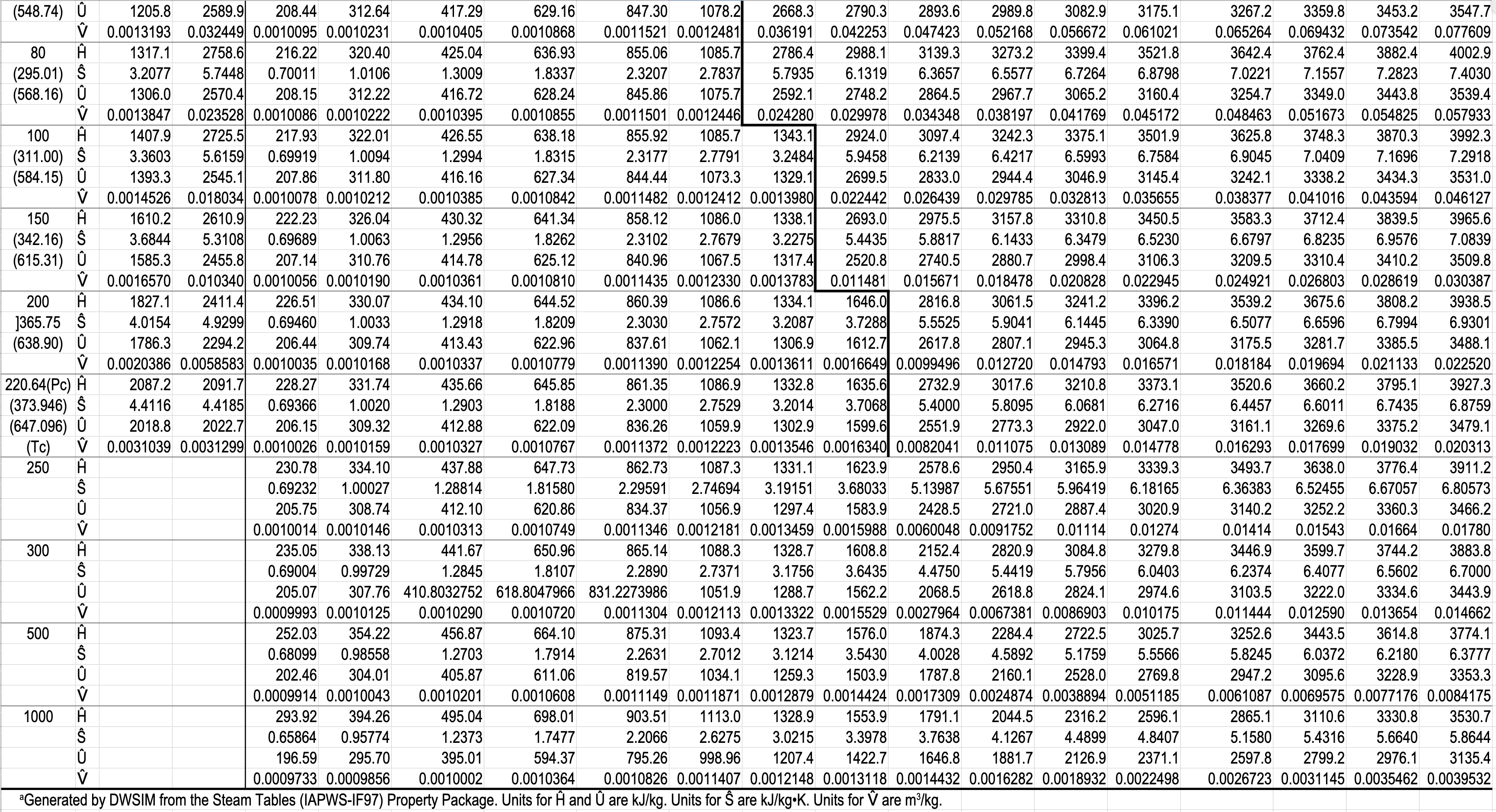
In the two-phase region (saturated steam and water)
\(\ \ \ \ DF = 1\)
Have two phases (water and steam) in equilibrium.
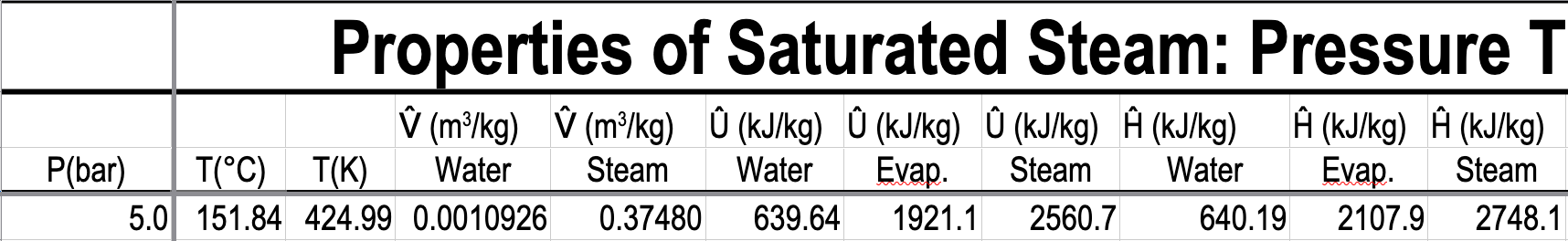
e.g. What is the temperature, and what are the specific enthalpies of saturated water and saturated steam at \(5.0\) bar?
In the two-phase region (saturated steam and water)
We know the properties of the two phases.
What is the property of the overall mixture?
Define the quality.
\(\ \ \ \ x \equiv \dfrac{m_\mathrm{steam}}{m_\mathrm{steam} + m_\mathrm{water}} = \dfrac{m_\mathrm{steam}}{m_\mathrm{total}}\)
- What is the pressure and specific internal energy of a mixture of steam and water at \(88\ ^\circ\mathrm{C}\) and a quality of \(80\%\)?
- What is the quality of a mixture of steam and water at \(0.65017\) bar, and a specific enthalpy of \(2000\ \mathrm{kJ/kg}\)?
What is the pressure and specific internal energy of a mixture of steam and water at \(88\ ^\circ\mathrm{C}\) and a quality of \(80\%\)?
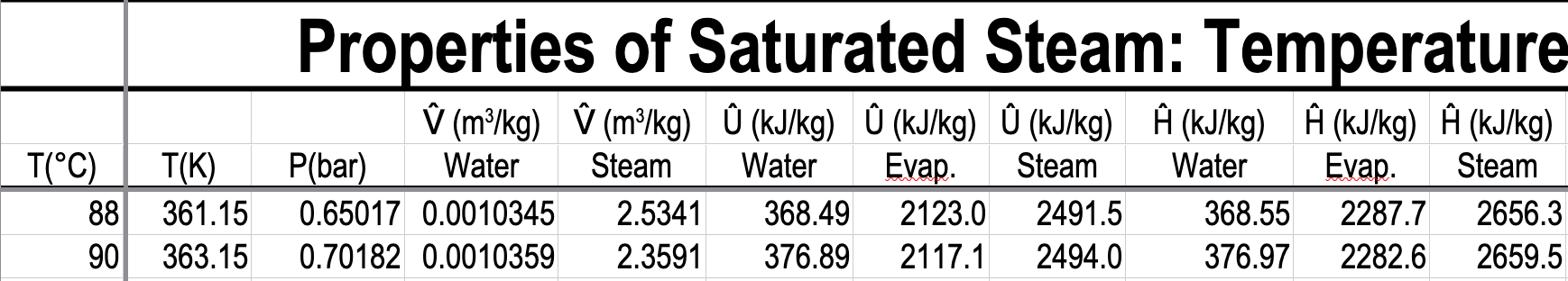
\(P=0.65017\ \mathrm{bar}\)
\(\hat{U}_{\text{mix}} = x\hat{U}_{v} + (1-x)\hat{U}_{l} = 0.8 \mathbf{\cdot} 2491.5 + 0.2 \mathbf{\cdot} 368.49 = 2066.9\ \mathrm{kJ/kg}\)
\(\hat{U}_{\text{mix}} = x\hat{U}_{v}\ +\ (1-x)\hat{U_{l}} = x(\hat{U}_{v} - \hat{U}_{l}) + \hat{U}_{l} = x\Delta \hat{U}_{lv} + \hat{U}_{l}\)
\(\hat{U}_{\text{mix}} = x\Delta \hat{U}_{lv} + \hat{U}_{l} = 0.8\mathbf{\cdot} 2123.0 + 368.49 = 2066.9\ \mathrm{kJ/kg}\)
What is the quality of a mixture of steam and water at \(0.65017\) bar, and a specific enthalpy of \(2000\ \mathrm{kJ/kg}\)?

\(\hat{H}_{\text{mix}} = x\hat{H}_{v}\ +\ (1-x)\hat{H_{l}} = x(\hat{H}_{v} - \hat{H}_{l}) + \hat{H}_{l} = x\Delta \hat{H}_{lv} + \hat{H}_{l}\)
\(\\\)
\(x = \dfrac{\hat{H}_{\mathrm{mix}} - \hat{H}_{l}}{\hat{H}_{v} - \hat{H}_{l}} = \dfrac{2000 - 368.55}{2656.3 - 368.55} = 0.7131 = 71.31\)%
\(\\\)
\(x = \dfrac{\hat{H}_{\text{mix}} - \hat{H}_{l}}{\Delta\hat{H}_{lv}} = \dfrac{2000 - 368.55}{2287.7} = 0.7131 = 71.31\)%
The Takeaways
- In the single phase region we have to specify two properties.
- The single-phase table is set up to be easy to use for temperature and pressure but we definitely don’t have to specify just temperature and pressure.
- In the two phase region we have to specify one property such as temperature or pressure, and then we often have to calculate the properties of a mixture using the quality.
Thanks for watching!
The previous video in the series is in the link in the upper left. The next video in the series is in the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.
The DOFPro Team

