Feeling the Heat? How to Get Your Best Estimate.
DOFPro Team

Intro
Always use reliable data if you have them. If not, then estimate
\(\ \ \ \ \ C_p\)
\(\ \ \ \ \ \Delta \hat{H}_v\)
\(\ \ \ \ \ \Delta \hat{H}_m\)
\(\ \ \ \ \ \Delta \hat{H}_s\)
for use in the First Law.
The standard reference is The Properties of Gases and Liquids. Current edition is the sixth.
Estimating Formulas
If the tabulated data are missing use a formula like Kopp’s rule,
\[ (C_p)_{\mathrm{compound}} = \sum_\text{all atoms} C_{pa} \]
How about mixtures?
Lacking other data, use a weighted average.
| Element | Solids | Liquids |
|---|---|---|
| C | 7.5 | 12 |
| H | 9.6 | 18 |
| B | 11 | 20 |
| Si | 16 | 24 |
| O | 17 | 25 |
| F | 21 | 29 |
| P | 23 | 31 |
| S | 26 | 31 |
| All others | 26 | 33 |
The \(C_p \Delta T\) changes are known as sensible heat changes. During a phase change, heat is added without a change in temperature. Such changes are latent heat changes.
- Latent heat of vaporization, \(\hspace{4pt} \Delta \hat{H}_v\)
- Latent heat of fusion, \(\hspace{4pt} \Delta \hat{H}_m\)
- Latent heat of sublimination, \(\hspace{4pt} \Delta \hat{H}_s\)
\(\hspace{1mm}\)
Why \(\Delta \hat{H}\)? Why not \(\Delta \hat{U}\)?
Vaporize one pound of water at constant \(T\) and \(P\). What kind of system?
\(\hspace{1mm}\)
The \(1\)st law is: \(\Delta U = Q + W\)
\(\hspace{1mm}\)
For a closed system the pressure-volume work is:
\(\hspace{1mm}\)
Now if \(P =\) constant
\(\hspace{1mm}\)
So the \(1\)st law becomes:
\(\hspace{1mm}\)
If a phase change takes place in a constant volume closed system, then \(\Delta \hat{U}\) must be evaluated as
\(\hspace{1mm}\)
\(\hspace{1mm}\)
\(\hspace{1mm}\)
How do you evaluate latent heats?
- Tables
- Correlations
- Educated Guesses
\(\hspace{1mm}\)
Trouton’s Rule (within 30% accuracy)
\(\hspace{1mm}\)
water, low-MW alcohols
Chen’s equation (within 2% accuracy)
\(\hspace{1mm}\)
with \(T\)’s in Kelvin, \(P\) in atm, and \(\Delta \hat{H}\) in \(\mathrm{kJ/mol}\)
\(\hspace{1mm}\)
Also Clausius and Claperyon backwards
And finally if you have \(\Delta \hat{H}_v\) at one temperature and you need it at another, the Watson correlation
\(\hspace{1mm}\)
Trouton’s Rule for Solids (unspecified accuracy)
For metallic elements
For inorganic compounds
For organic compounds
Trouton’s Rule Plot
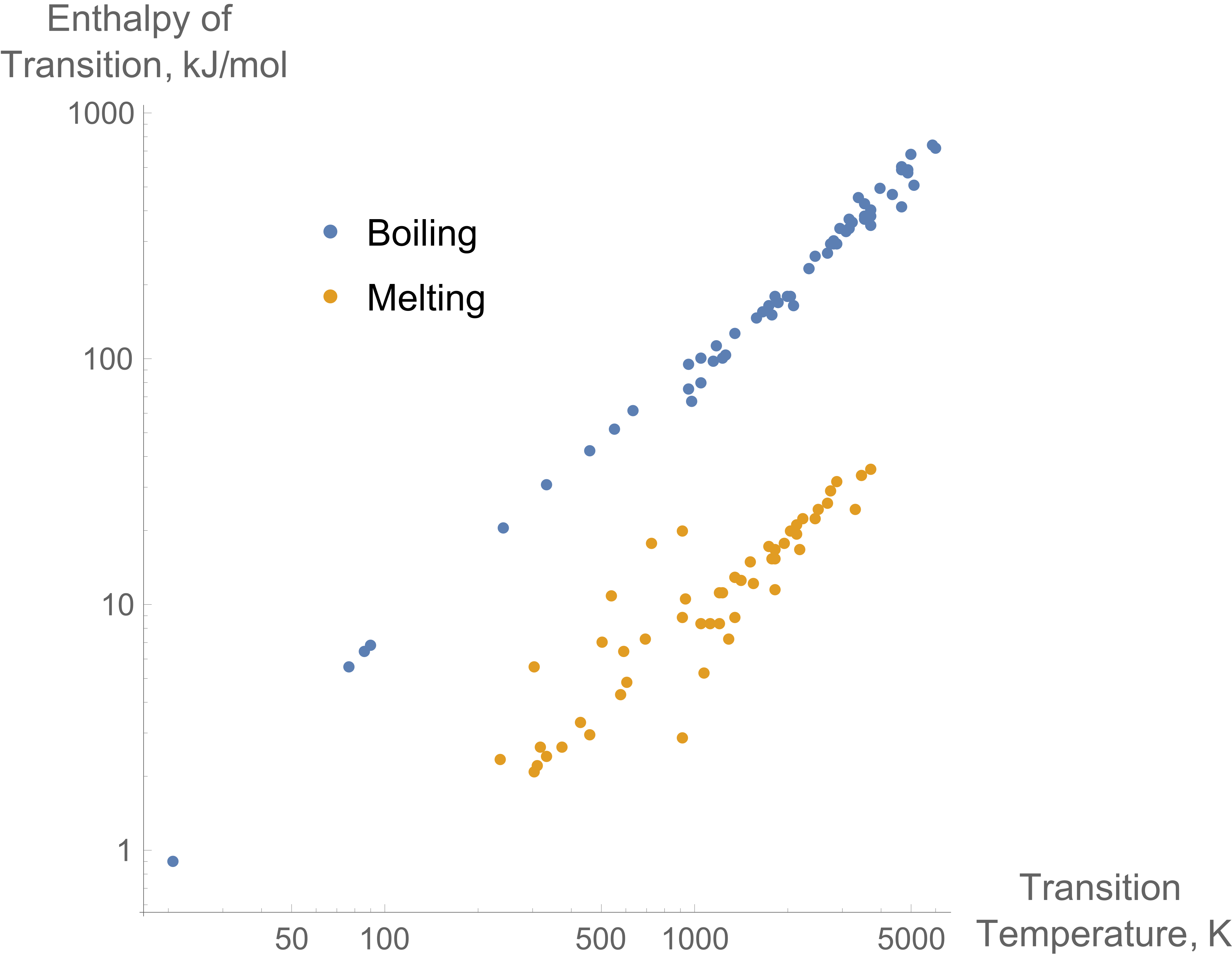
Valuable Numbers to Know
Approximate Heat Capacities
| Gas Type | \(C_v\) | \(C_p\) |
|---|---|---|
| Monatomic | \(\frac{3}{2}R\) | \(\frac{5}{2}R\) |
| Diatomic | \(\frac{5}{2}R\) | \(\frac{7}{2}R\) |
\(C_p\text{ in } \left[\mathrm{\dfrac{Btu}{lb_m\ ^{\circ} F}}\right] \text{ or } \left[\mathrm{\dfrac{cal}{g\ ^{\circ} C}}\right]\)
Steam \(\approx 0.5\)
Water \(\approx 1.0\)
Ice \(\approx 0.5\)
\(\Delta \hat{H}_v \approx 1000\ \left[\mathrm{\dfrac{Btu}{lb_m}}\right]\)
\(\ \Delta \hat{H}_m \approx 150\ \left[\mathrm{\dfrac{Btu}{lb_m}}\right]\)
The Takeaways
- Estimating formulas are useful when you can’t find reliable data for heat capacities and for specific enthalpies for phase changes.
- Kopp’s rule is useful for heat capacities.
- Trouton’s rule, Chen’s equation, and the Clausius Clapeyron equation used inversely are useful for heats of vaporization
- The Watson correlation is useful for estimating heats of vaporization at temperatures other than the normal boiling point.
- Trouton’s rule for solids is useful for heats of fusion.
Thanks for watching!
The previous video in the series is in the link in the upper left. The next video in the series is in the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.
The DOFPro Team

