
Dry Bulbs, Wet Bulbs,
and My Will to Live:
Mastering the Psych Chart
DOFPro Team
Sling Psychrometer

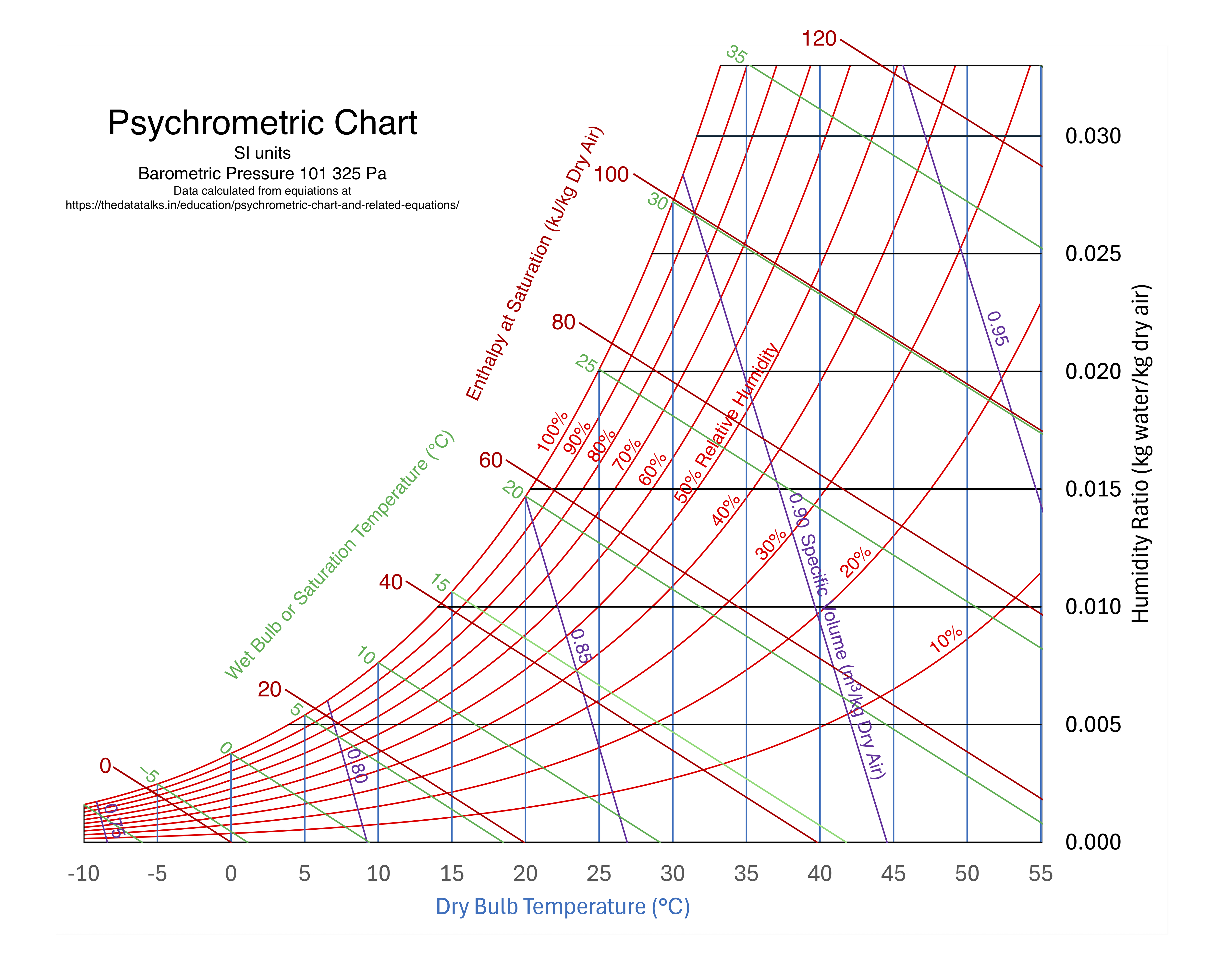
Lines on Psychrometric Chart
- Humid volume, \(\hat{V}\)
- Dry-bulb temperature, \(T_\mathrm{db}\)
- Wet-bulb temperature, \(T_\mathrm{wb}\)
- Absolute humidity, \(h_a\)
- Relative humidity, \(h_r\)
- Enthalpy at saturation, \(\hat{H}_\mathrm{sat}\)
- Enthalpy deviation, \(\hat{H}_\mathrm{dev}\)
- Adiabatic saturation line

Humid Volume

Dry-Bulb Temperature

Wet-Bulb Temperature

Absolute Humidity

Relative Humidity

Enthalpy at Saturation

Enthalpy Deviation
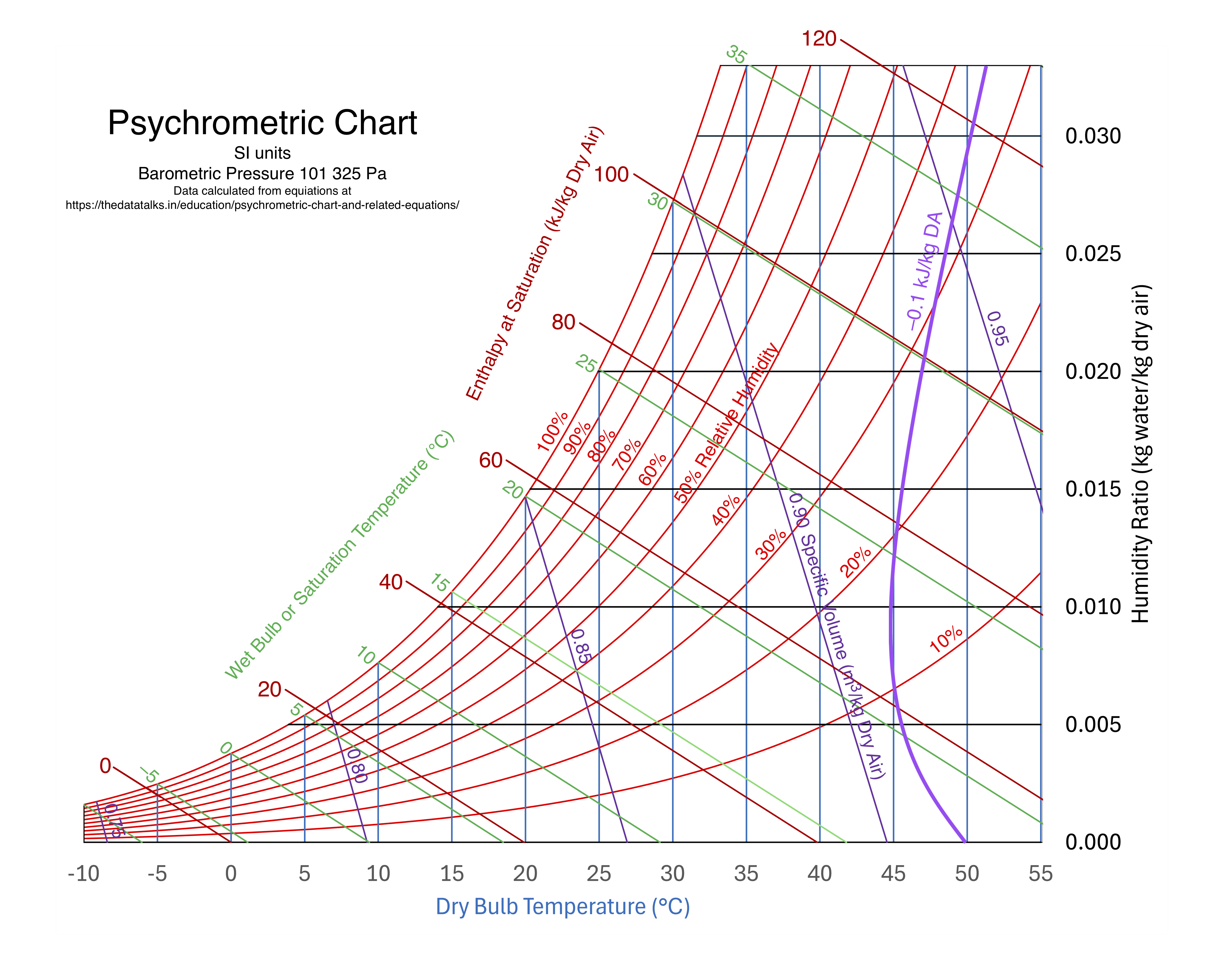
Adiabatic Saturation Line

First Example
You set out a cold root brew on a day when the temperature is 86 degrees Fahrenheit, which you may recall was \(30\ ^\circ\mathrm{C}\), and the relative humidity is \(30\%\).
- At what temperature is the brew when water quits condensing on the can?
- What is the dew point?
- What is the wet bulb temperature?
- What is the absolute humidity?
- What is the specific volume?
- What is the enthalpy relative to liquid water and dry air at \(0\ ^\circ\mathrm{C}\)?
First Example

\[\colorbox{white}{$\displaystyle T_\mathrm{dp} = 10.55\ ^\circ\mathrm{C}$}\]
\[\colorbox{white}{$\displaystyle T_\mathrm{wb} = 17.95\ ^\circ\mathrm{C}$}\]
\[\colorbox{white}{$\displaystyle h_a = 0.00795\ \mathrm{kg\ H_2O/kg\ DA}$}\]
\[\colorbox{white}{$\displaystyle \hat{V} = 0.870\ \mathrm{m^3/kg\ DA}$}\]
\[\colorbox{white}{$\displaystyle \hat{H}_\mathrm{sat} = 50.86\ \mathrm{kJ/kg\ DA}$}\]
\[\colorbox{white}{$\displaystyle \hat{H}_\mathrm{dev} = -0.37\ \mathrm{kJ/kg\ DA}$}\]
\[\colorbox{white}{$\displaystyle \hat{H} = \hat{H}_\mathrm{sat} + \hat{H}_\mathrm{dev} = 50.86 - 0.37 = 50.49\ \mathrm{kJ/kg\ DA}$}\]
Second Example
We have hot air at \(43\ ^\circ\mathrm{C}\) and \(10\%\ h_r\) entering an adiabatic clothes dryer containing 5 kg of water and assorted clothes and exiting at \(90\%\ h_r\).
- How many cubic meters of air are required to completely dry the clothes?
- What is the steady-state temperature of the exiting air?
- What is the steady-state temperature of the clothes until they are almost completely dry?
Second Example

\[\colorbox{white}{$\displaystyle h_a = 0.0054\ \mathrm{kg\ H_2O/kg\ DA}$}\]
\[\colorbox{white}{$\displaystyle h_a = 0.0144\ \mathrm{kg\ H_2O/kg\ DA}$}\]
\(m_\mathrm{DA} = \dfrac{5}{0.0144 - 0.0054} = 556\ \mathrm{kg\ DA}\)
\[\colorbox{white}{$\displaystyle \color{purple}\hat{V} = 0.903\ \mathrm{m^3/kg\ DA}$}\]
\(V = m_\mathrm{DA} \hat{V} = 556 \cdot 0.903 = \boxed{502\ \mathrm{m^3\ DA}}\)
\[\colorbox{white}{$\displaystyle \boxed{\color{blue}T_\mathrm{db} = 21.27\ ^\circ\mathrm{C}}$}\]
\[\colorbox{white}{$\displaystyle \boxed{\color{green}T_\mathrm{wb} = 20.1\ ^\circ\mathrm{C}}$}\]
The Takeaways
- The psychrometric chart is a graphical way to solve problems involving the air-water vapor system. Given any two of: \(T_\mathrm{db}\), \(T_\mathrm{wb}\) or \(\hat{H}_\mathrm{sat}\) , \(h_r\), \(h_a\) or \(T_\mathrm{dp}\) , or \(\hat{V}\), you can read the remaining values off the diagram.
- Psychrometric charts are useful for humidifying and dehumidifying process calculations, and heating, ventilation, and air conditioning calculations.
- Psychrometric charts have largely been supplanted with online calculators or phone apps.
Thanks for watching!
The previous video in the series is in the link in the upper left. The next video in the series is in the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.
The DOFPro Team

