Resistance is Futile. Entropy Always Wins
DOFPro Team

The Second Law of Thermodynamics
- No apparatus can operate in such a way that its only effect (in system and surroundings) is to convert heat absorbed by a system completely into work.
- It is impossible by a cyclic process to convert the heat absorbed by a system completely into work.
- No process is possible which consists solely in the transfer of heat from one temperature level to a higher one.
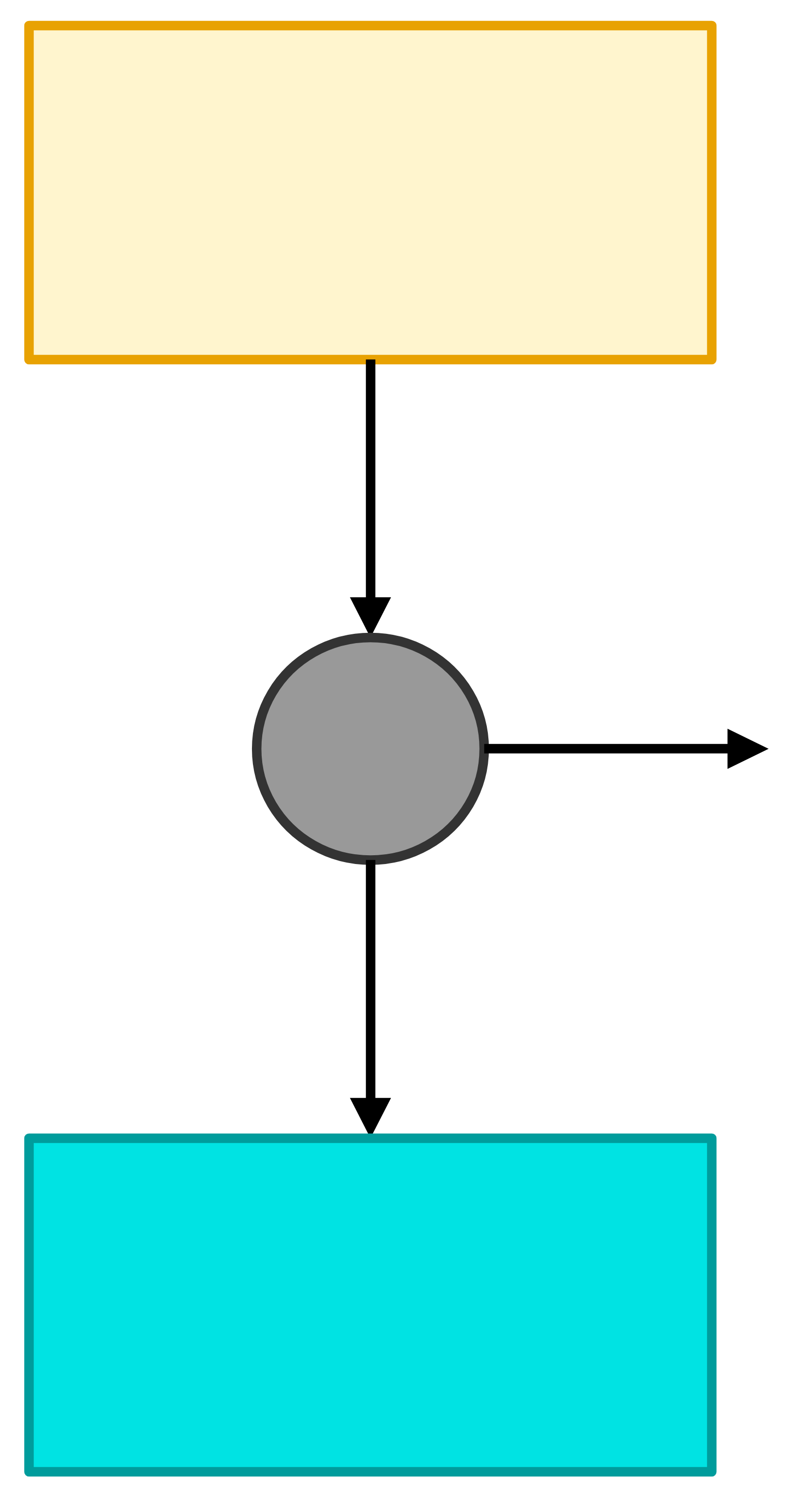
\(T_\mathrm{hot}\)
\(T_\mathrm{cold}\)
\(|Q_\mathrm{hot}|\)
\(|W|\)
\(|Q_\mathrm{cold}|\)
Imagine two isothermal heat reservoirs (they can absorb or provide an infinite amount of heat at a constant temperature) at temperatures \(T_\mathrm{hot}\) and \(T_\mathrm{cold}\). Let’s extract an amount of heat, \(Q_\mathrm{hot}\), from the hot reservoir and convert as much of the heat as possible into work and reject the remaining heat, \(Q_\mathrm{cold}\), into the cold reservoir.
- What is the maximum possible amount of work?
- What is the minimum amount of heat rejected?

\(T_\mathrm{hot}\)
\(T_\mathrm{cold}\)
\(|Q_\mathrm{hot}|\)
\(|Q_\mathrm{cold}|\)
\(|W|\)
The efficiency of any heat engine operating between the two heat reservoirs is defined as:
\[\eta \equiv \frac{|W|}{|Q_\mathrm{hot}|}\]
What is the relationship between \(Q_\mathrm{hot}\) and \(Q_\mathrm{cold}\)?
Assume a cyclic process on a closed system with negligible kinetic and potential energy changes.
1st Law (system is engine)
\[\Delta{U} = Q + W = 0\]
\[-W = Q_\mathrm{hot} + Q_\mathrm{cold}\]
\[|W| = |Q_\mathrm{hot}| - |Q_\mathrm{cold}|\]

\(T_\mathrm{hot}\)
\(T_\mathrm{cold}\)
\(|Q_\mathrm{hot}|\)
\(|Q_\mathrm{cold}|\)
\(|W|\)
- What is the maximum possible amount of work?
- What is the minimum amount of heat rejected?
Any engine which operates between these two reservoirs and produces the greatest possible work while rejecting the minimum amount of heat is called a Carnot engine. The efficiency of a Carnot engine is:
\[\eta \equiv \frac{|W|}{|Q_\mathrm{hot}|} = 1 - \frac{T_\mathrm{cold}}{T_\mathrm{hot}}\]
What is the relationship between \(Q_\mathrm{hot}\) and \(Q_\mathrm{cold}\) for a Carnot engine?

\(T_\mathrm{hot}\)
\(T_\mathrm{cold}\)
\(|Q_\mathrm{hot}|\)
\(|Q_\mathrm{cold}|\)
\(|W|\)
From the 1st Law (system is engine)
\[-W = Q_\mathrm{hot} + Q_\mathrm{cold}\]
Combining with the efficiency
\[\frac{Q_\mathrm{hot} + Q_\mathrm{cold}}{Q_\mathrm{hot}} = 1 - \frac{T_\mathrm{cold}}{T_\mathrm{hot}}\]
or
\[\frac{Q_\mathrm{hot}}{T_\mathrm{hot}}=-\frac{Q_\mathrm{cold}}{T_\mathrm{cold}}\]

\(T_\mathrm{hot}\)
\(T_\mathrm{cold}\)
\(|Q_\mathrm{hot}|\)
\(|Q_\mathrm{cold}|\)
\(|W|\)
Imagine an actual reversible cycle

Approximate the cycle with infinitesimal Carnot engines
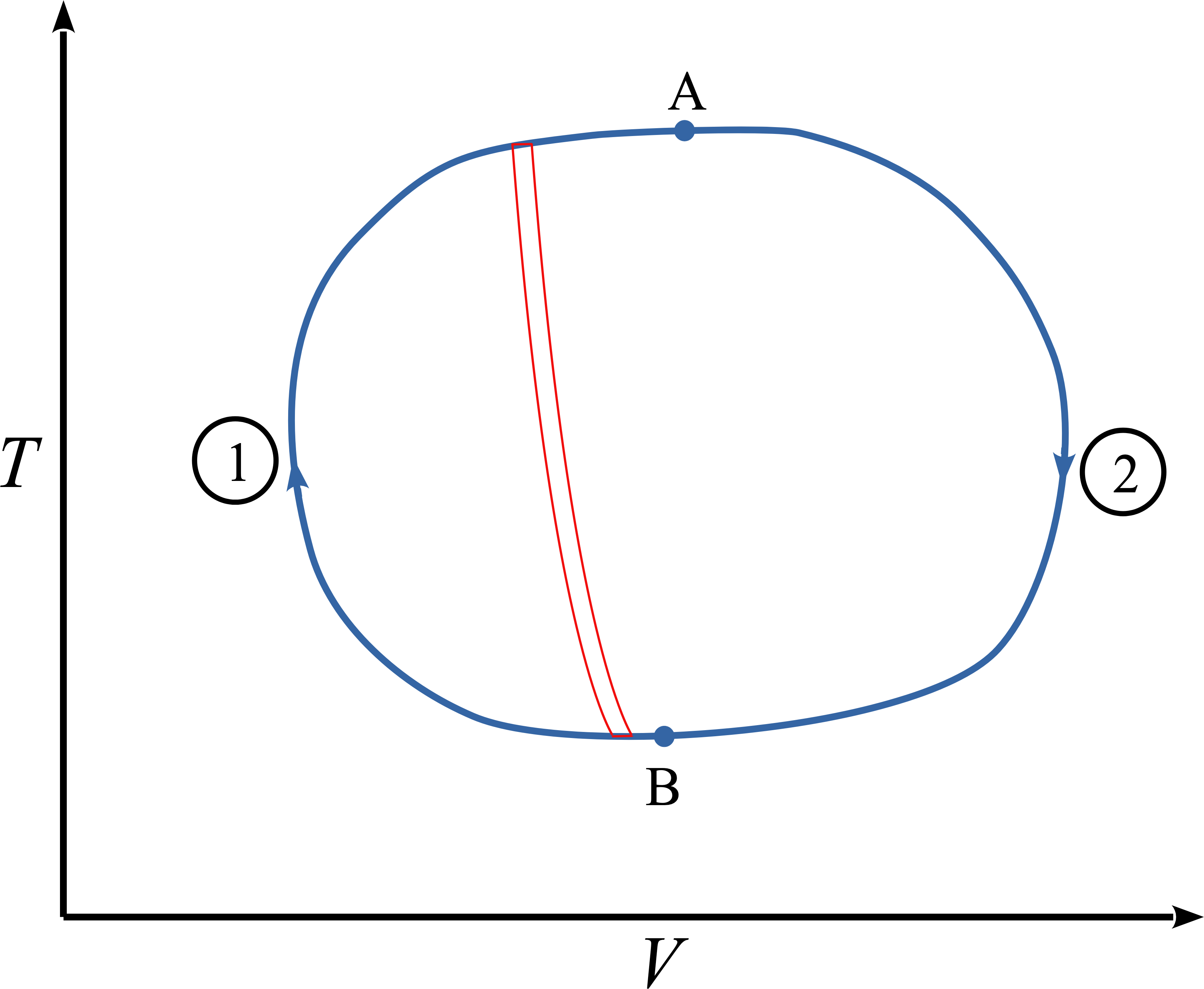
\[\frac{dQ_\mathrm{hot}}{dT_\mathrm{hot}} + \frac{dQ_\mathrm{cold}}{dT_\mathrm{cold}}=0\]
Integrate over all
\[\int \frac{dQ}{T}=\int \frac{dQ_\mathrm{hot}}{dT_\mathrm{hot}} + \int \frac{dQ_\mathrm{cold}}{dT_\mathrm{cold}}=0\]

Note that if the loop integral is zero,
\[\oint\limits_{\substack{\mathrm{A–B} \\ \mathrm{path\ 1}}} \frac{dQ}{T} = \oint\limits_{\substack{\mathrm{A–B} \\ \mathrm{path\ 2}}} \frac{dQ}{T}\]
What kind of function is independent of path?
Let us define a new state function, called entropy.
\(\Delta S = \int\limits_\mathrm{A}^\mathrm{B} \dfrac{dQ_\mathrm{Carnot}}{T}\)
or
\(dS = \dfrac{dQ_\mathrm{carnot}}{T}\)
If
\(Q \neq Q_\mathrm{Carnot}\)
then
\(dS = \dfrac{dQ}{T} + \dfrac{dW_\mathrm{lost}}{T}\)
Since \(S\) is a state function, any path can be used to evaluate \(\Delta S\).
Mathematical Statement of the Second Law
or
\(S\) can be thought of as the reversible normalized heat transfer or as the degree of randomness or disorder.
Remember:
can be less than zero. But
What is \(\Delta S\) for a cyclic process?
Example
Analyze the following process. Is it possible?

\(T_\mathrm{hot} = 500\ \mathrm{K}\)
\(T_\mathrm{cold} = 300\ \mathrm{K}\)
\(|Q_\mathrm{hot}| = 1\ \mathrm{kJ/s}\)
\(|Q_\mathrm{cold}| = 300\ \mathrm{J/s}\)
\(|W| =\ ?\)
1st Law around engine
Closed system
\(\Delta U = 0 = Q + W\)
\(Q = 1\ \mathrm{kJ/s} - 300\ \mathrm{J/s} = 700\ \mathrm{J/s}\)
\(0 = 700\ \mathrm{J/s} + W\)
\(W = -700\ \mathrm{J/s} = -700\ \mathrm{W}\)
Example (cont.)
Analyze the following process. Is it possible?

\(T_\mathrm{hot} = 500\ \mathrm{K}\)
\(T_\mathrm{cold} = 300\ \mathrm{K}\)
\(|Q_\mathrm{hot}| = 1\ \mathrm{kJ/s}\)
\(|Q_\mathrm{cold}| = 300\ \mathrm{J/s}\)
\(|W| =\ ?\)
2nd Law
\(\Delta S_\mathrm{system} = 0\), Why?
\(\Delta S_\mathrm{surroundings} =\ ?\)
\(\Delta S_\mathrm{500\ K\ reservoir} = \frac{-1000\ \mathrm{J/s}}{500\ \mathrm{K}} = -2 \frac{\mathrm{W}}{\mathrm{K}}\)
\(\Delta S_\mathrm{300\ K\ reservoir} = \frac{300\ \mathrm{J/s}}{300\ \mathrm{K}} = 1 \frac{\mathrm{W}}{\mathrm{K}}\)
\(\Delta S_\mathrm{total} = 0 + (-2) + (1)\)
\(= -1 \frac{\mathrm{W}}{\mathrm{K}} < 0\)
What is the maximum possible work?
Example:
What is the maximum work?

\(T_\mathrm{hot} = 500\ \mathrm{K}\)
\(T_\mathrm{cold} = 300\ \mathrm{K}\)
\(|Q_\mathrm{hot}| = 1\ \mathrm{kJ/s}\)
\(|Q_\mathrm{cold}| =\ ?\)
\(|W| =\ ?\)
2nd Law
\(\Delta S_\mathrm{surr.} = \Delta S_\mathrm{500\ K\ res.} + \Delta S_\mathrm{300\ K\ res.}\)
\(\Delta S_\mathrm{500\ K\ res.} = \frac{-1000\ \mathrm{J/s}}{500\ \mathrm{K}} = -2 \frac{\mathrm{W}}{\mathrm{K}}\)
\(\Delta S_\mathrm{300\ K\ res.} = 0 - \Delta S_\mathrm{500\ K\ res.}\)
\(= 2 \frac{\mathrm{W}}{\mathrm{K}} = \frac{|Q_\mathrm{cold}|}{300\ \mathrm{K}}\)
\(|Q_\mathrm{cold}| = 2 \frac{\mathrm{W}}{\mathrm{K}} \cdot 300\ \mathrm{K} = 600\ \mathrm{W}\)
\(Q_\mathrm{net} = 1\ \mathrm{kW} - 600\ \mathrm{W} = 400\ \mathrm{W}\)
\(0 = 400\ \mathrm{W} + W\)
\(W = -400\ \mathrm{W}\)
Example:
What would you get from the Carnot efficiency?

\(T_\mathrm{hot} = 500\ \mathrm{K}\)
\(T_\mathrm{cold} = 300\ \mathrm{K}\)
\(|Q_\mathrm{hot}| = 1\ \mathrm{kJ/s}\)
\(|Q_\mathrm{cold}| =\ ?\)
\(|W| =\ ?\)
Using Carnot efficiency
\(\eta = \frac{|W|}{|Q_\mathrm{hot}|} = 1 - \frac{T_\mathrm{cold}}{T_\mathrm{hot}}\)
\(= 1 - \frac{300}{500} = 0.400\)
\(|W| = \eta |Q_\mathrm{hot}|\)
\(= 0.400 \cdot 1\ \mathrm{kW} = 400\ \mathrm{W}\)
\(W = -400\ \mathrm{W}\)
\(|Q_\mathrm{cold}| = 1000\ \mathrm{W} - 400\ \mathrm{W} = 600\ \mathrm{W}\)
The Takeaways
- The thermal efficiency, \(\eta\), of a heat engine is the work output (what you want) divided by the heat input (what you have to pay for).
- Entropy is a state function.
- The entropy change of a heat reservoir is the heat transferred to or from the reservoir divided by the absolute temperature of the reservoir.
- The Second Law of Thermodynamics states that the sum of the entropy change for the system and for the surroundings must be greater than or equal to zero.
- The Carnot thermal efficiency, \(\eta_\mathrm{Carnot} = 1 - \dfrac{T_\mathrm{cold}}{T_\mathrm{hot}}\)
Thanks for watching!
The previous video in the series is in the link in the upper left. The next video in the series is in the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.
The DOFPro Team

