Entropy Made Me Do It: Turbines, Compressors, and Other Second-Law Shenanigans (Part 1)
DOFPro Team

Introduction
These videos, Entropy Made Me Do It, Part 1 and Part 2 derive the equations for common items in power and refrigeration machinery.
Part 1 discusses
- the turbine or expander
- the adiabatic compressor
- the isothermal compressor
Part 2 discusses
- the pump
- the nozzle
- the valve or throttle
Up First
Turbine – Steam or Combustion Gases
Expander – Compressed Gases
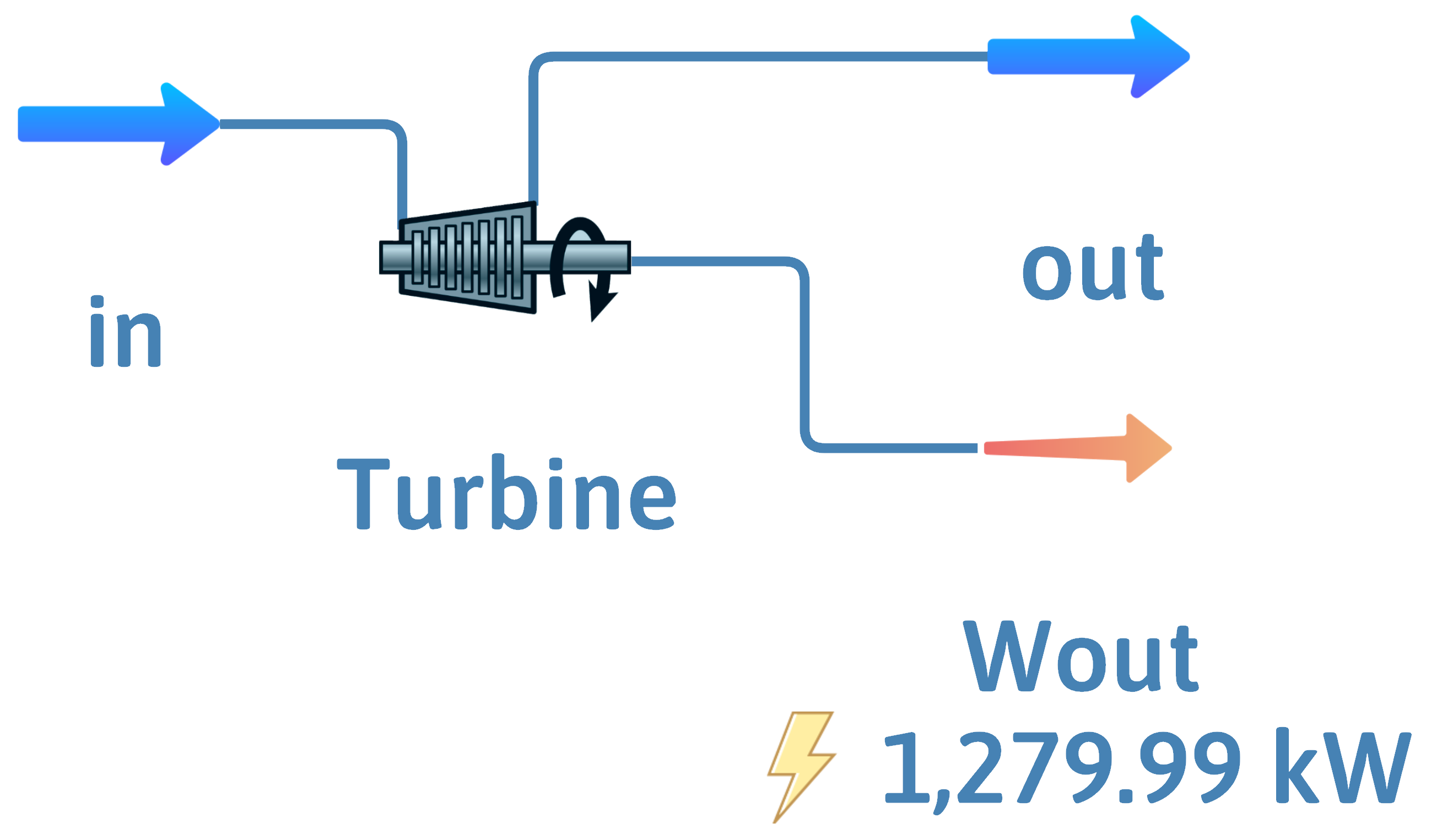
![]()
Inputs – \(\dot{m}\) or \(\dot{n}\), \(T\) and \(P\)
Outputs – \(\dot{m}\) or \(\dot{n}\), \(T\) or \(P\)
Goal is work output
Then
Compressor
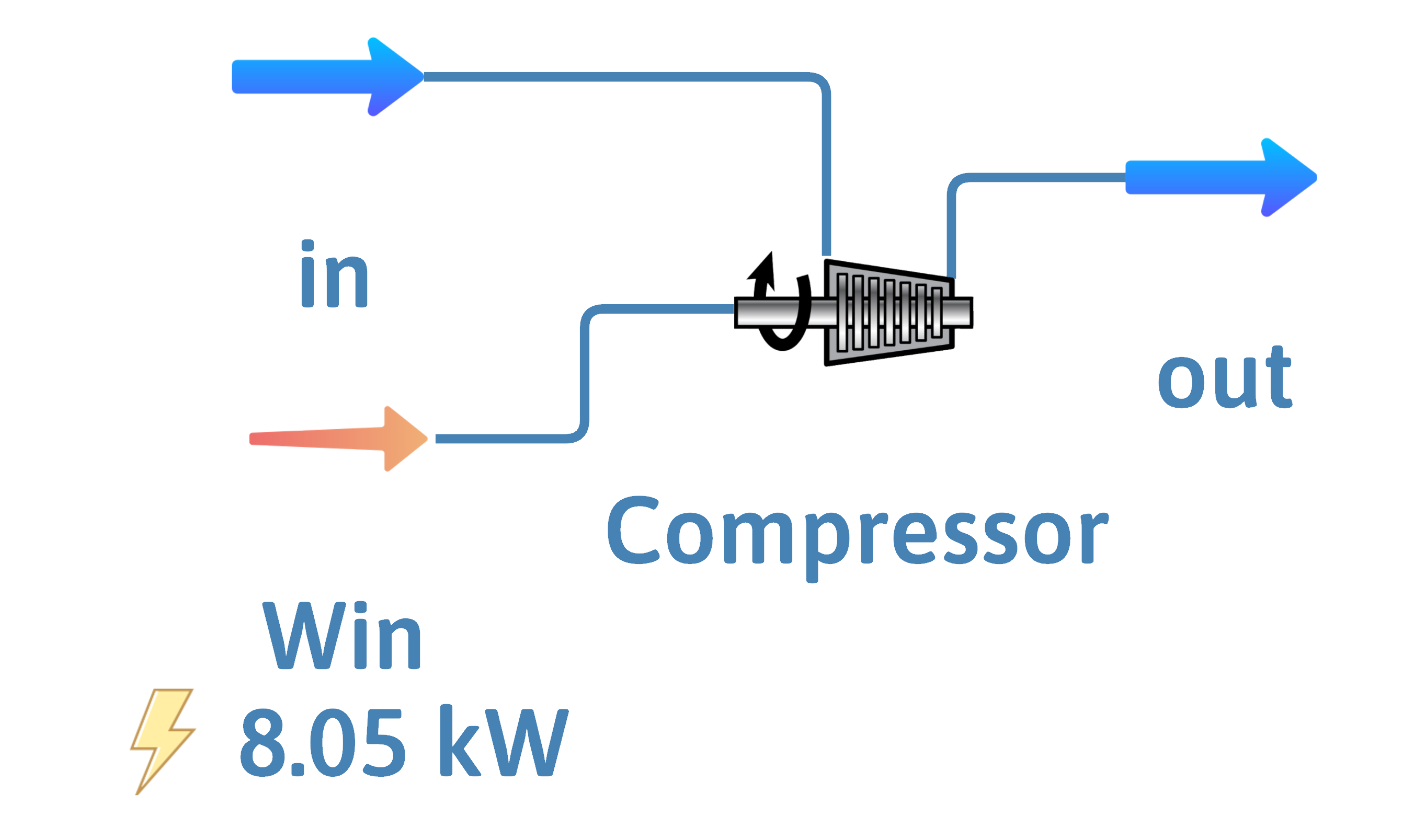
![]()
Goal is pressure increase of gas
Inputs – \(\dot{m}\) or \(\dot{n}\), \(T\) and \(P\)
Outputs – \(\dot{m}\) or \(\dot{n}\), \(P\)
Have to input work
Turbine or Expander

CC BY 3.0, via Wikimedia Commons

National Archives and Records Administration, Public domain, via Wikimedia Commons
Turbine or Expander
A turbine or expander takes a high-temperature and high-pressure steam or gas mixture and sends it through a series of blades, called stator blades and rotor blades, and expands it to create power or shaft work.
1st Law
Steam
\[\Delta \dot{H} = \dot{W}_s = \dot{m} \Delta \hat{H} = \dot{m} (\hat{H}_\mathrm{out} - \hat{H}_\mathrm{in})\]
Ideal Gas
\[\Delta \dot{H} = \dot{W}_s = \dot{n} \Delta \hat{H} = \dot{n} \int\limits_{T_\mathrm{in}}^{T_\mathrm{out}} C_p dT = \dot{n} C_p (T_\mathrm{out} - T_\mathrm{in})\]
Turbine or Expander
A real turbine has a turbine efficiency, that compares its performance with that of an isentropic turbine.
\[\eta_\mathrm{turbine} = \frac{\dot{W}_{s\text{-actual}}}{\dot{W}_{s\text{-isentropic}}} = \frac{\Delta \hat{H}_{\text{actual}}}{\Delta \hat{H}_{\text{isentropic}}}\]
Calculating the isentropic values
\[\Delta \hat{S} = 0\]
Steam
\[\hat{S}_\mathrm{out} = \hat{S}_\mathrm{in}\]
\[x = \dfrac{\hat{S}_\mathrm{in}-\hat{S}_l}{\hat{S}_v-\hat{S}_l}\]
To calculate the outlet enthalpy
\[\hat{H}_\mathrm{out} = x \hat{H}_v + (1 - x) \hat{H}_l\]
\[\Delta \hat{H}_\mathrm{isentropic} = \hat{H}_\mathrm{out} - \hat{H}_\mathrm{in}\]
Ideal Gas
\[\Delta \hat{S} = 0 = C_p \ln \frac{T_\mathrm{out}}{T_\mathrm{in}} - R \ln \frac{P_\mathrm{out}}{P_\mathrm{in}}\]
\[C_p \ln \frac{T_\mathrm{out}}{T_\mathrm{in}} = R \ln \frac{P_\mathrm{out}}{P_\mathrm{in}}\ \ \ \ \text{or}\ \ \ \ \frac{T_\mathrm{out}}{T_\mathrm{in}} = \left(\frac{P_\mathrm{out}}{P_\mathrm{in}}\right)^\frac{R}{C_p}\]
Substitute in for \(\Delta T\).
\[ \Delta \hat{H}_\mathrm{isentropic} = C_p \Delta T = C_p (T_\mathrm{out} - T_\mathrm{in}) = C_p T_\mathrm{in} \left[\left(\frac{P_\mathrm{out}}{P_\mathrm{in}}\right)^\frac{R}{C_p} - 1\right] \]
Calculating actual properties
Steam
\[\eta_\mathrm{turbine} = \frac{\Delta \hat{H}_{\text{actual}}}{\Delta \hat{H}_{\text{isentropic}}}\]
\[\hat{H}_{\text{out-actual}} = \eta_\mathrm{turbine} \Delta \hat{H}_{\text{isentropic}} + \hat{H}_{\text{in}}\]
Calculating actual quality and entropy for steam
\[x_\mathrm{actual} = \dfrac{\hat{H}_\text{out-actual}-\hat{H}_l}{\hat{H}_v-\hat{H}_l}\]
\[\hat{S}_\text{out-actual}=x_\mathrm{actual}\hat{S}_v + (1 - x_\mathrm{actual}) \hat{S}_l\]
Calculating actual properties
Ideal Gas
\[\eta_\mathrm{turbine} = \frac{\Delta \hat{H}_{\text{actual}}}{\Delta \hat{H}_{\text{isentropic}}}\]
\[\Delta \hat{H}_{\text{actual}} = \eta_\mathrm{turbine} \Delta \hat{H}_{\text{isentropic}}\]
\[\Delta \hat{H}_{\text{actual}} = C_p \Delta T_\text{actual}\]
\[T_\text{out-actual} = \frac{\Delta \hat{H}_{\text{actual}} }{C_p} + T_\mathrm{in}\]
\[\Delta \hat{S}_{\text{actual}} = C_p \ln \frac{T_\text{out-actual}}{T_\text{in}} - R \ln \frac{P_\text{out}}{P_\text{in}}\]
Compressor
Ideal Gas only in introductory videos. Two-phase compressors are rare.

NASA, Public domain, via Wikimedia Commons

Asurnipal, CC BY-SA 4.0 https://creativecommons.org/licenses/by-sa/4.0, via Wikimedia Commons
Adiabatic Compressor
A real adiabatic compressor has a compressor efficiency, that compares its performance with that of an isentropic compressor.
\[\eta_\mathrm{compressor} = \frac{\dot{W}_{s\text{-isentropic}}}{\dot{W}_{s\text{-actual}}} = \frac{\Delta \hat{H}_{\text{isentropic}}}{\Delta \hat{H}_{\text{actual}}}\]
1st Law
\[\Delta \dot{H}_\mathrm{isentropic} = \dot{W}_{s\text{-isentropic}} = \dot{n} \int\limits_{T_\mathrm{in}}^{T_\mathrm{out}} C_p dT = \dot{n} C_p \Delta T\]
2nd Law
\[\Delta \hat{S}_\mathrm{air} = \int\limits_{T_\mathrm{in}}^{T_\mathrm{out}} \frac{C_p}{T} dT - R \ln \frac{P_\mathrm{out}}{P_\mathrm{in}}= C_p \ln \frac{T_\mathrm{out}}{T_\mathrm{in}} - R \ln \frac{P_\mathrm{out}}{P_\mathrm{in}}\]
Minimum work for \(\Delta \hat{S} = 0\)
\[C_p \ln \frac{T_\mathrm{out}}{T_\mathrm{in}} = R \ln \frac{P_\mathrm{out}}{P_\mathrm{in}}\ \ \ \ \text{or}\ \ \ \ \frac{T_\mathrm{out}}{T_\mathrm{in}} = \left(\frac{P_\mathrm{out}}{P_\mathrm{in}}\right)^\frac{R}{C_p}\]
Plug \(T_\mathrm{out}\) back into 1st Law.
\[\dot{W}_{s\text{-isentropic}} = \dot{n} C_p \Delta T\]
Calculate work and final enthalpy from efficiency
\[\dot{W}_{s\text{-actual}} = \frac{\dot{W}_{s\text{-isentropic}}}{\eta_\mathrm{compressor}}\ \ \ \ \text{and}\ \ \ \ \Delta \hat{H}_{\text{actual}} = \frac{\Delta \hat{H}_{\text{isentropic}}}{\eta_\mathrm{compressor}}\]
If necessary, you can calculate actual outlet \(T\) and entropy change.
Isothermal Compressor
A cooling-water jacket is often used to attempt to maintain the compressor at a constant temperature. An isothermal compressor uses much less power than an isentropic compressor, at the cost of a cooling system. A real isothermal compressor has a compressor efficiency, that compares its performance with that of an isothermal compressor.
\[\eta_\mathrm{compressor} = \frac{\dot{W}_{s\text{-isothermal}}}{\dot{W}_{s\text{-actual}}}\]
1st Law
\[\Delta \dot{H}_\text{iso} = 0\ \ \ \ \text{and}\ \ \ \ \dot{Q} = - \dot{W}_{s\text{-iso}} = \dot{n} RT \ln \frac{P_1}{P_2}\]
There is no need for the 2nd Law. Use the efficiency to calculate the actual work
\[\dot{W}_{s\text{-actual}} = \frac{\dot{W}_{s\text{-isothermal}}}{\eta_\mathrm{compressor}}\]
If necessary, you can calculate actual enthalpy change, outlet \(T\), and entropy change.
The Takeaways
- Adiabatic Turbines and compressors have efficiencies that compare the real or actual case with the isentropic case.
- Isothermal compressors have efficiencies that compare the real or actual case with the isothermal case.
- We have shown how to calculate turbines and expanders for both steam and ideal gas.
- We have shown how to calculate compressors for ideal gas only.
Thanks for watching!
The previous video in the series is in the link in the upper left. The next video in the series is in the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.
The DOFPro Team

