The Law is a Thief: It Destroys Exergy and Laughs in Your Face Part 1
DOFPro Team

Concepts
- Work is more valuable than heat.
- Heat rejected to (or taken from) the ambient is worthless (or free).
- The same for work performed on or done by the ambient.
- For a given process or system (desired change of state) there is a minimum work required or a maximum work available. The goal is to find it.
Example
Cool 100 mol/s of ethanol from 100°C to 50°C
What is the minimum work required or maximum work obtainable?
- Using cooling water.
- Using a refrigerator.
- Using a series of infinitesimal Carnot engines.
1. Heat Exchanger
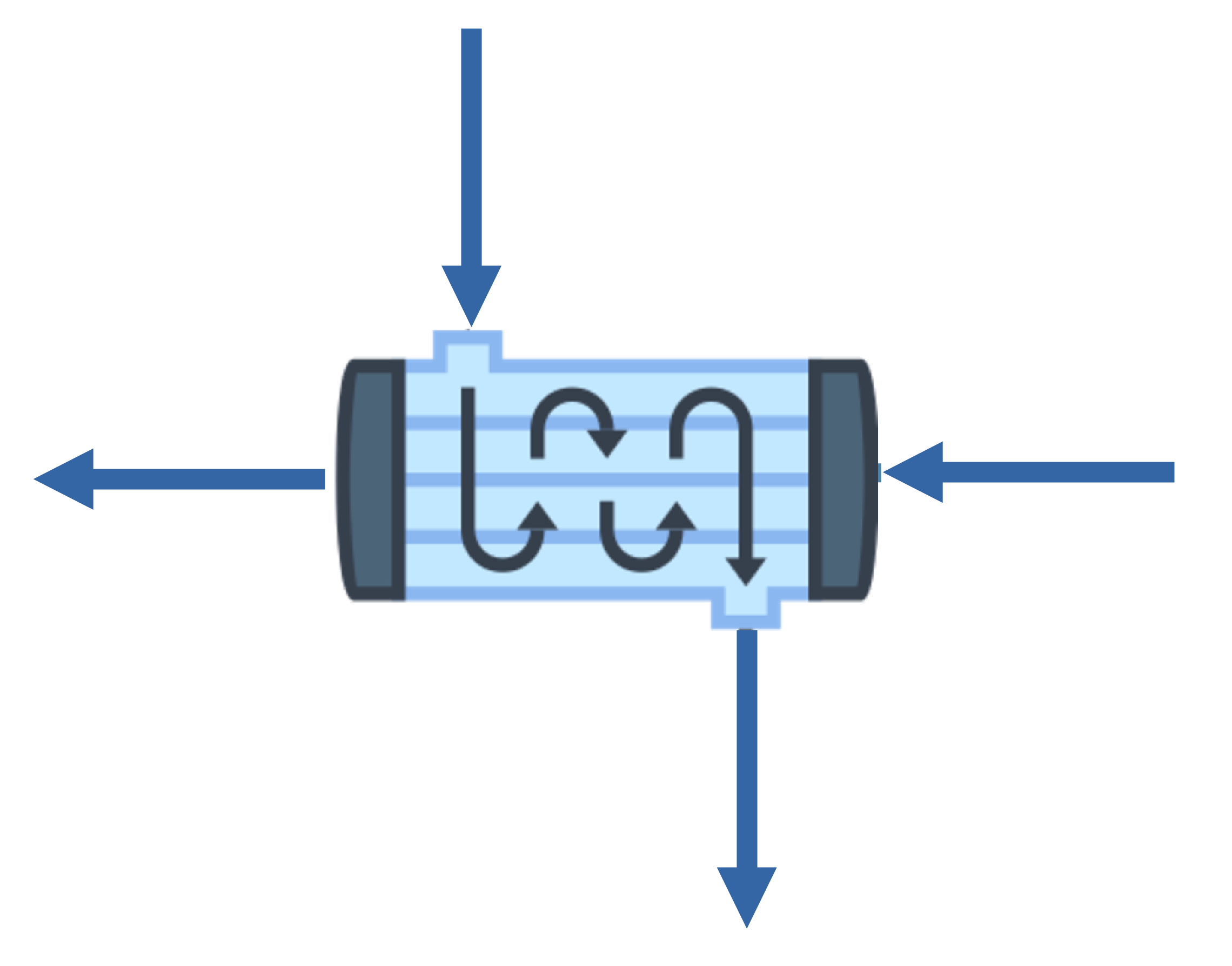
\(\dot{m}\Delta H = \dot{Q} + \dot{W}_s = 0\)
\(\dot{Q} = 0\)
\(\boxed{\dot{W}_s = 0}\)
\(\mathrm{EtOH}\)
\(100\ ^\circ\mathrm{C}\)
\(90\ ^\circ\mathrm{C}\)
\(50\ ^\circ\mathrm{C}\)
\(40\ ^\circ\mathrm{C}\)
\(\mathrm{H_2O}\)
2. Refrigerator
\(\boxed{\dot{W}_s\text{ into system (Oops)}}\)
\(\mathrm{EtOH}\)
\(100\ ^\circ\mathrm{C}\)
\(50\ ^\circ\mathrm{C}\)
\(|\dot{Q}_\mathrm{cold}|\)
\(|\dot{Q}_\sigma|\)
\(|\dot{W}_s|\)
Ambient, \(T_\sigma\)
3. Infinitesimal Carnot Engines
\(\dot{W}_\text{total} = \dot{m}(\hat{Q} - T_\sigma \Delta \hat{S})\)
\[= \dot{m} \left(\int_{100}^{50} C_p dT - T_\sigma \int_{100}^{50} \dfrac{C_p}{T} dT\right)\]
\(= 100[-6722.36 - 300(-19.2917)]\)
\(= -93.49 \text{ kJ/s} = W_\text{max}\)
\(\mathrm{EtOH}\)
\(100\ ^\circ\mathrm{C}\)
\(50\ ^\circ\mathrm{C}\)
\(|\delta\dot{Q}_\mathrm{hot}|\)
\(|\delta\dot{Q}_\sigma|\)
\(|\delta\dot{W}_s|\)
Ambient, \(T_\sigma\)
Derivation
- Derive for an open transient system
- First Law
- Second Law
- Add together
- Solve for the net shaft work
- Assume entropy generation and lost work are zero
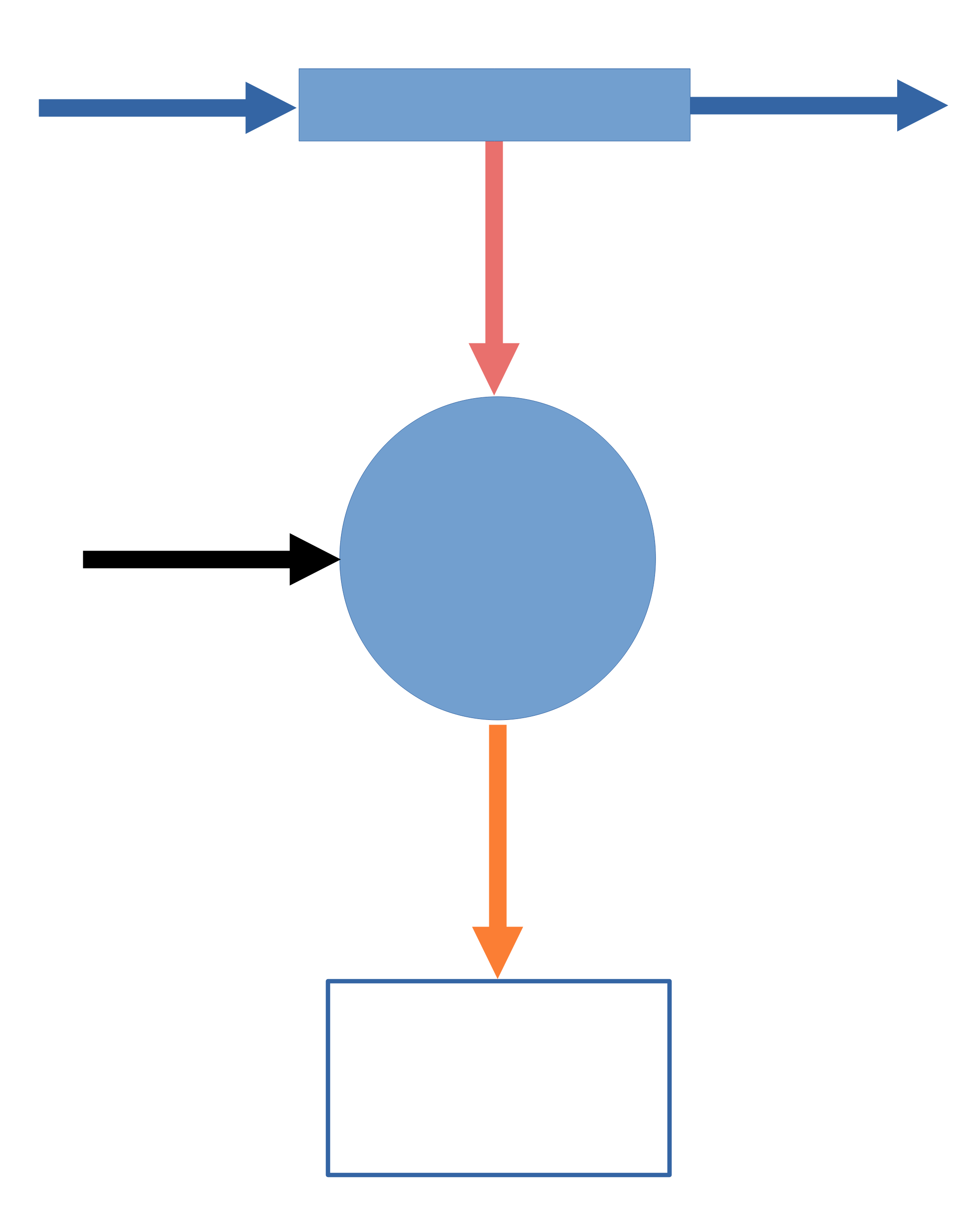
For an open transient system, the First Law is
\(\dfrac{d}{dt}(U + E_k + E_p)_{\text{control volume}} = \sum \dot{Q}_i + \sum \dot{W}_{s_j} - (\Delta \dot{H} + \Delta \dot{E}_k + \Delta \dot{E}_p)_{\text{flow streams}}\)
which on a mass basis is
\(\dfrac{d}{dt}\left[m\left(\hat{U} + \hat{E}_k + \hat{E}_p\right)\right]_\mathrm{cv} = \sum \dot{Q}_i + \sum \dot{W}_{s_i} + \sum\limits_\mathrm{in} \dot{m}_i\left(\hat{H} + \hat{E}_k + \hat{E}_p\right) - \sum\limits_\mathrm{out} \dot{m}_i\left(\hat{H} + \hat{E}_k + \hat{E}_p\right)\)
or on a mole basis is
\(\dfrac{d}{dt}\left[n\left(\hat{U} + \hat{E}_k + \hat{E}_p\right)\right]_\mathrm{cv} = \sum \dot{Q}_i + \sum \dot{W}_{s_i} + \sum\limits_\mathrm{in} \dot{n}_i\left(\hat{H} + \hat{E}_k + \hat{E}_p\right) - \sum\limits_\mathrm{out} \dot{n}_i\left(\hat{H} + \hat{E}_k + \hat{E}_p\right)\)
For an open transient system, the Second Law is
\(\dfrac{dS_\mathrm{cv}}{dt} + \Delta ( \dot{S} )_\mathrm{fs} - \sum_j \dfrac{\dot{Q}_j}{T_j} = \dot{S}_G \geq 0\)
\(\dfrac{d ( m\hat{S} )_\mathrm{cv}}{dt} + \Delta ( \dot{m}\hat{S} )_\mathrm{fs} - \sum_j \dfrac{\dot{Q}_j}{T_j} = \dot{S}_G \geq 0 \quad\) Mass Basis
or \(\dfrac{d ( n\hat{S} )_\mathrm{cv}}{dt} + \Delta ( \dot{n}\hat{S} )_\mathrm{fs} - \sum_j \dfrac{\dot{Q}_j}{T_j} = \dot{S}_G \geq 0 \quad\) Mole Basis
\(\dot{Q}_j\) is heat transferred into system from reservoir \(j\)
\(T_j\) is temperature of reservoir \(j\)
Combine (add the two equations) and solve for \(\sum \dot{W}_{s_i} = \dot{W}_s\)
Ambient \(T\) is \(T_\sigma\).
\(\dot{W}_s = \frac{d}{dt} \left( U + E_k + E_p - T_\sigma S \right)_\mathrm{cv} - \sum \left( 1 - \frac{T_\sigma}{T_i} \right) \dot{Q}_i + \left( \Delta \dot{H} + \Delta \dot{E}_k + \Delta \dot{E}_p - T_\sigma \Delta \dot{S} \right)_\mathrm{fs} + T_\sigma \dot{S}_\mathrm{gen}\)
\(\dot{Q}_\sigma\) is transfer to ambient at \(T_\sigma\) and doesn’t show up in the balance.
What is \(T_\sigma \dot{S}_\mathrm{gen}\)? \(\quad\) \(T_\sigma \dot{S}_\mathrm{gen} = \dot{W}_\mathrm{lost}\)
If \(\dot{S}_\mathrm{gen} = 0 \Rightarrow \dot{W}_\mathrm{lost} = 0\), then \(\quad \dot{W}_{s} = \dot{W}_\mathrm{ideal}\)
\(\dot{W}_\mathrm{ideal}\) is the ideal work or the reversible work. It (or the negative of it) is sometimes called availability, available work or exergy.
The Combined 1st and 2nd Law
Open Transient System
\(\dot{W}_{\text{ideal}} = \dfrac{d}{dt}(U + E_k + E_p - T_\sigma S)_\mathrm{cv} - \sum \left(1 - \dfrac{T_\sigma}{T_i}\right) \dot{Q}_i + (\Delta \dot{H} + \Delta \dot{E}_k + \Delta \dot{E}_p - T_\sigma \Delta \dot{S})_\mathrm{fs}\)
On a mass basis
\(\dot{W}_{\text{ideal}} = \dfrac{d}{dt}\left[m(\hat{U} + \hat{E}_k + \hat{E}_p - T_\sigma \hat{S})\right]_\mathrm{cv} - \sum \left(1 - \dfrac{T_\sigma}{T_i}\right) \dot{Q}_i\)
\[ + \sum_{\text{out}} \dot{m}_i (\hat{H} + \hat{E}_k + \hat{E}_p - T_\sigma \hat{S}) - \sum_{\text{in}} \dot{m}_i (\hat{H} + \hat{E}_k + \hat{E}_p - T_\sigma \hat{S}) \]
Or the same equation on a mole basis with \(n\) and \(\dot{n}\) replacing \(m\) and \(\dot{m}\)
The Combined 1st and 2nd Law (cont.)
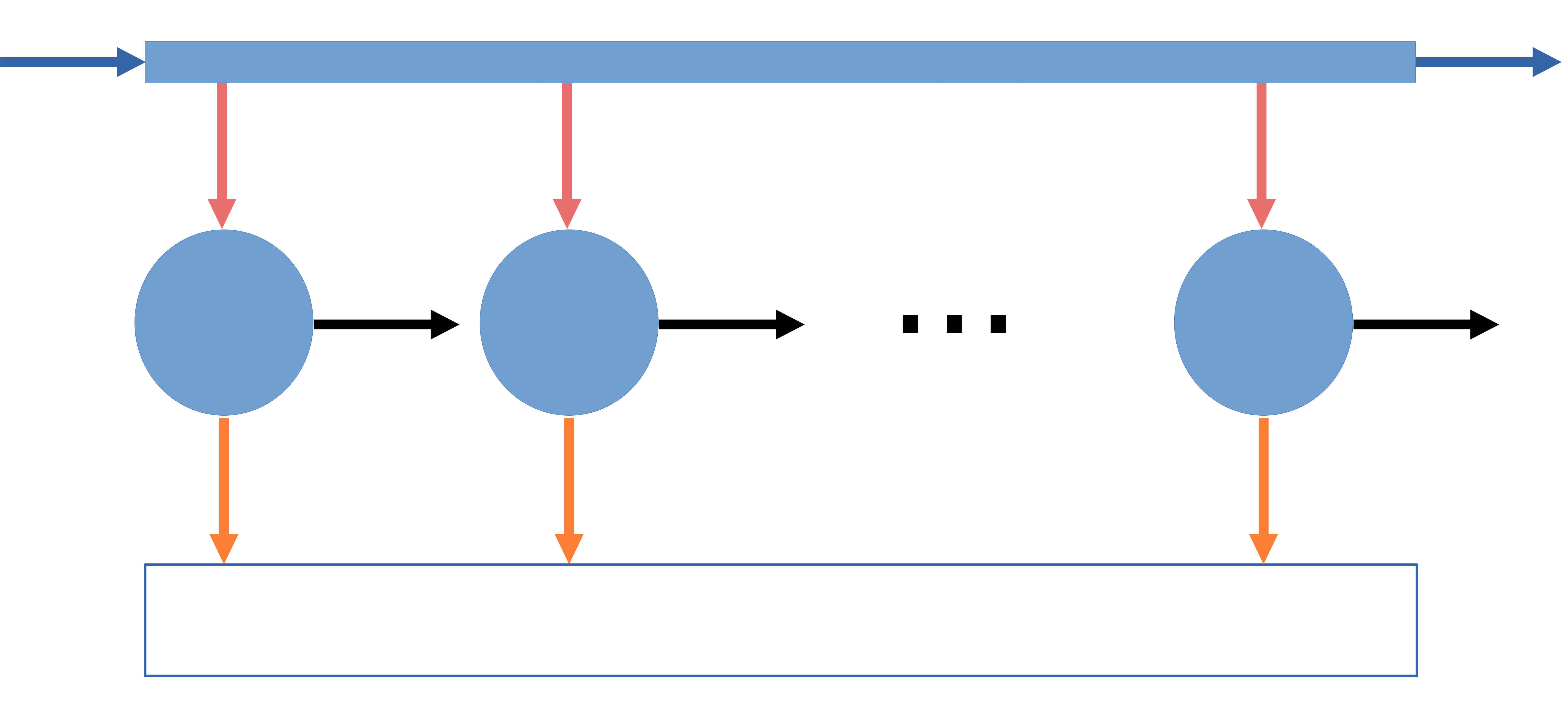
Open Steady-State System
\(\dot{W}_{\text{ideal}} = - \sum \left(1 - \dfrac{T_\sigma}{T_i}\right) \dot{Q}_i + (\Delta \dot{H} + \Delta \dot{E}_k + \Delta \dot{E}_p - T_\sigma \Delta \dot{S})_\mathrm{fs}\)
On a mass basis
\(\dot{W}_{\text{ideal}} = - \sum \left(1 - \dfrac{T_\sigma}{T_i}\right) \dot{Q}_i + \sum\limits_{\text{out}} \dot{m}_i (\hat{H} + \hat{E}_k + \hat{E}_p - T_\sigma \hat{S}) - \sum\limits_{\text{in}} \dot{m}_i (\hat{H} + \hat{E}_k + \hat{E}_p - T_\sigma \hat{S})\)
On a mole basis
\(\dot{W}_{\text{ideal}} = - \sum \left(1 - \dfrac{T_\sigma}{T_i}\right) \dot{Q}_i + \sum\limits_{\text{out}} \dot{n}_i (\hat{H} + \hat{E}_k + \hat{E}_p - T_\sigma \hat{S}) - \sum\limits_{\text{in}} \dot{n}_i (\hat{H} + \hat{E}_k + \hat{E}_p - T_\sigma \hat{S})\)
The Combined 1st and 2nd Law (cont.)
Closed System
\(W_{\text{ideal}} = \Delta U + \Delta E_k + \Delta E_p - T_\sigma \Delta S - \sum \left(1 - \dfrac{T_\sigma}{T_i}\right) Q_i\)
We will rarely encounter closed systems in thermodynamic or 2nd Law analyses.
How do you use it?
Thermodynamic efficiency, \(\eta_t\) or \(\eta_{\tiny \mathrm{II}}\)
For processes that produce work:
\(\eta_t = \frac{\dot{W}_{s}}{\dot{W}_\mathrm{ideal}}\)
For processes that require work:
\(\eta_t = \frac{\dot{W}_\mathrm{ideal}}{\dot{W}_{s}}\)
Also the lost work or irreversibility is
\[\dot{W}_\mathrm{lost} = \dot{W}_{s} - \dot{W}_\mathrm{ideal}\]
Is \(\dot{W}_\mathrm{lost}\) always positive?
\[\dot{W}_\mathrm{lost} = T_\sigma \dot{S}_\mathrm{gen}\]
You should also analyze individual parts of a process and calculate lost work to see where the greatest inefficiencies lie.
Steps
- Solve the original problem. Determine all needed flows, enthalpies, entropies, temperatures, heat flows and work flows.
- Create a table with the information in Step 1. Then for each process or unit operation in the system:
- Solve for the ideal work, \(\dot{W}_\mathrm{ideal}\)
- Solve for the lost work, \(\dot{W}_\mathrm{lost}\)
- Solve for the thermodynamic efficiency, \(\eta_t\)
- Determine which parts of the process contribute the most to the lost work.
The Takeaways
- The combined First and Second Law is literally just the addition of the two laws, setting the entropy generation to zero, and solving for the ideal work, \(\dot{W}_\mathrm{ideal}\)
- The most common version is for open steady-state systems.
- Choosing the correct form of the thermodynamic efficiency requires knowledge of whether the process produces work or requires work.
- The combined First and Second Law helps identify which parts of a process contribute most to lost work or inefficiency.
Thanks for watching!
The previous video in the series is in the link in the upper left. The next video in the series is in the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.
The DOFPro Team

