Gotta Keep ’Em Separated
Just the Facts
DOFPro Team

Gotta Keep ’Em Separated
- The first in a series
- The most common unit-operations material balances
- Discusses the mixer, the divider, and the separator.
- For the mixer, the divider, and the separator both mass and moles are conserved.
- When You Saw It, What Was the Extent of Your Reaction? Parts 1, 2, and 3
- Chemical reactor extent of reaction and fractional conversion models
- Moles are not conserved in chemical reactions
Common Processing Units
- Mixer
- Divider
- Separator
- Reactor (Next three videos)
For open steady-state systems it is useful to use the following definitions and constitutive relationships:
Mixer
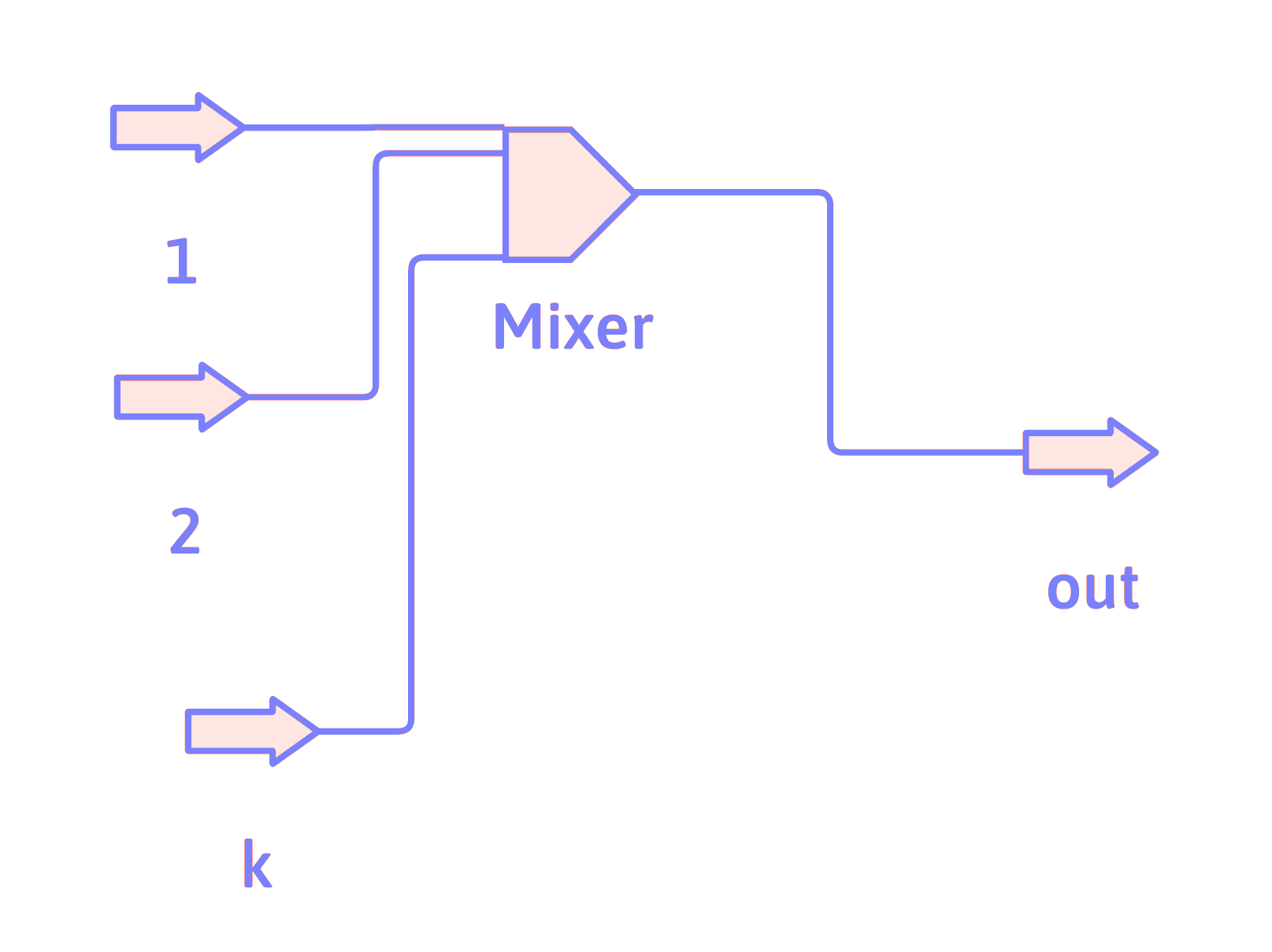
For each component, \(i\),
\[ \dot{n}_{i_\mathrm{out}} = \dot{n}_{i_1} + \dot{n}_{i_2} + \cdots +\dot{n}_{i_k} \]
Overall
\[ \dot{n}_\mathrm{out} = \dot{n}_1 + \dot{n}_2 + \cdots +\dot{n}_k \]
where
\[ \dot{n} = \sum_\mathrm{All} \dot{n}_i \]
Mixer Usage
A mixer combines (or mixes) the input streams together, so that the output stream is just the sum of all of the input streams.
The mass equations for a mixer are the same as the mole equations, with the \(n\)’s or \(\dot{n}\)’s replaced by \(m\)’s or \(\dot{m}\)’s.
\[ \dot{m}_{i_\mathrm{out}} = \dot{m}_{i_1} + \dot{n}_{i_2} + \cdots +\dot{m}_{i_k} \]
Overall
\[ \dot{m}_\mathrm{out} = \dot{m}_1 + \dot{m}_2 + \cdots +\dot{m}_k \]
where
\[ \dot{m} = \sum_\mathrm{All} \dot{m}_i \]
Divider
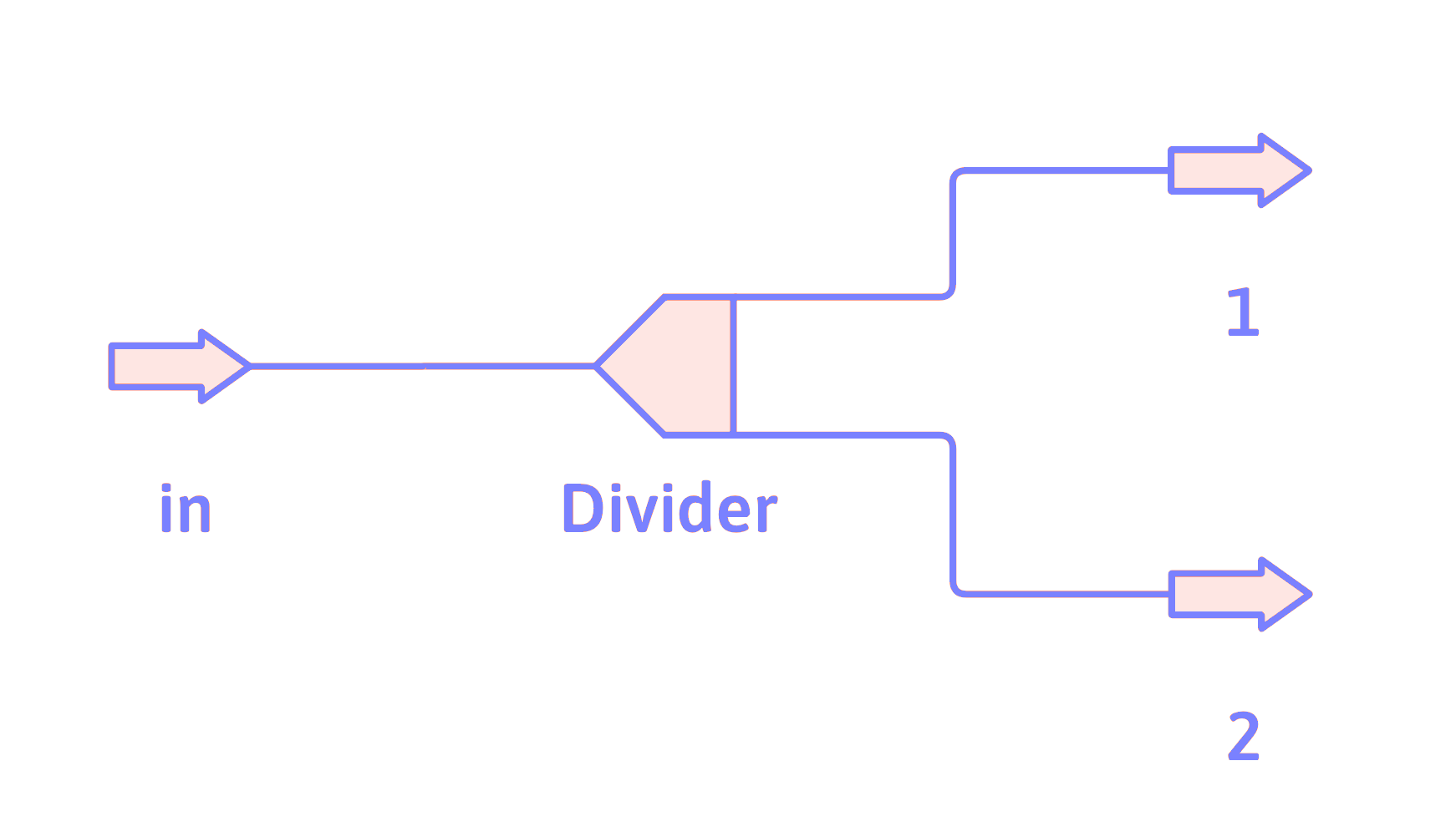
For each component, \(i\),
\[ \dot{n}_{i_\mathrm{in}} = \dot{n}_{i_1} + \dot{n}_{i_2} \]
Overall
\[ \dot{n}_\mathrm{in} = \dot{n}_1 + \dot{n}_2 \]
where
\[ \dot{n} = \sum_\mathrm{All} \dot{n}_i \]
Also
\[ x_{i_1} = x_{i_2} = x_{i_\mathrm{in}} \]
where the mole fraction, \(x_i \equiv \frac{n_i}{n_\mathrm{Total}} = \frac{\dot{n}_i}{\dot{n}}\) .
Divider Usage
A divider is used when a single stream needs to be divided up into two or more streams. The mass equations for a divider are the same as the mole equations, with the \(n\)’s or
\(\dot{n}\)’s replaced by \(m\)’s or \(\dot{m}\)’s.
For each component, \(i\),
\[ \dot{m}_{i_\mathrm{in}} = \dot{m}_{i_1} + \dot{m}_{i_2} \]
Overall
\[ \dot{m}_\mathrm{in} = \dot{m}_1 + \dot{m}_2 \]
where
\[ \dot{m} = \sum_\mathrm{All} \dot{m}_i \]
Also
\[ x_{i_1} = x_{i_2} = x_{i_\mathrm{in}} \]
where the mass fraction, \(x_i \equiv \frac{m_i}{m_\mathrm{Total}} = \frac{\dot{m}_i}{\dot{m}}\) .
Separator
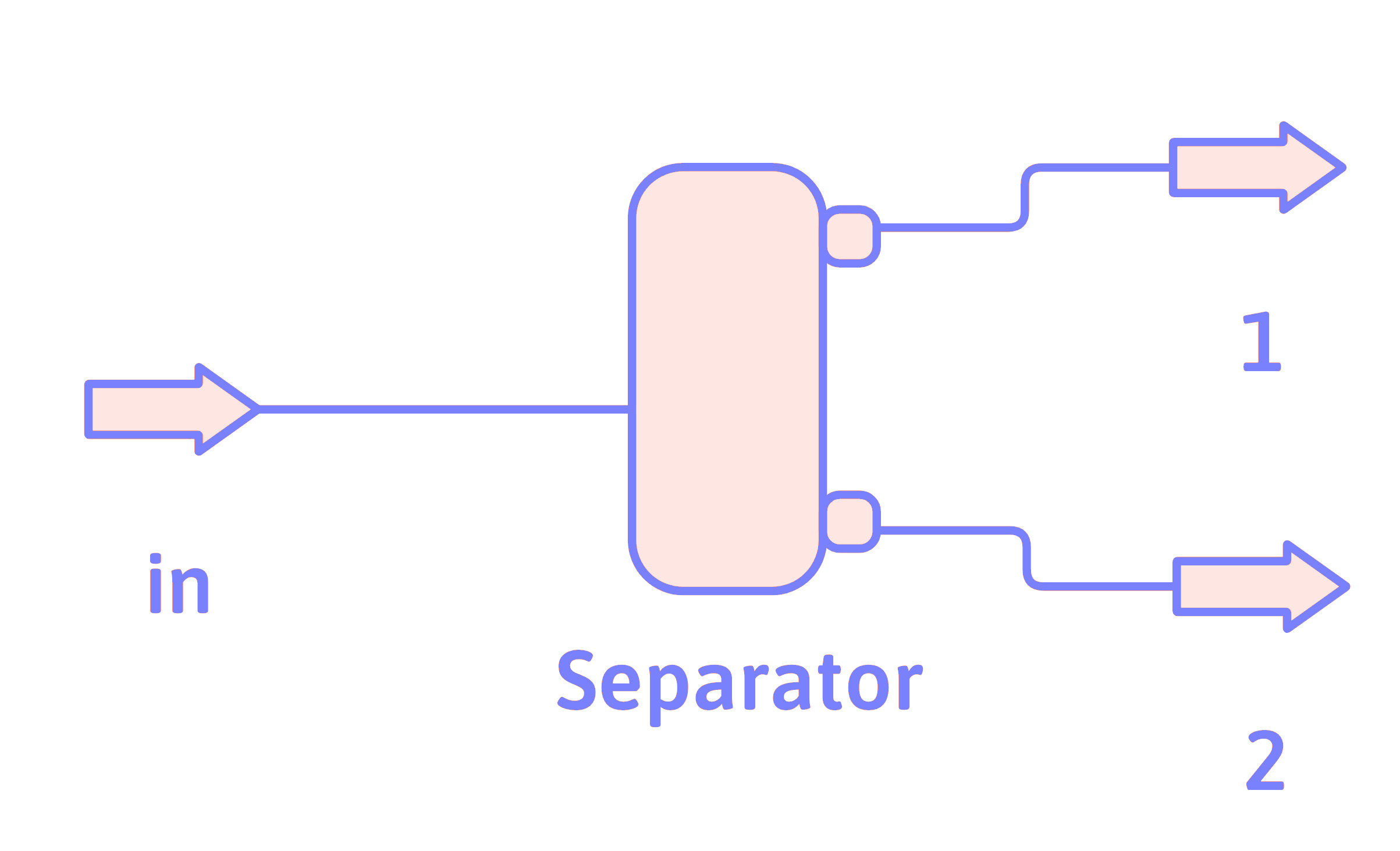
You often specify the split fraction,
\[ t_{is} \equiv \frac{\dot{n}_{i_s}}{\dot{n}_{i_\mathrm{in}}} \]
\(t_{i1}\) is the split fraction of species \(i\) with respect to Stream 1.
\[ \dot{n}_{i_1} = t_{i1} \dot{n}_{i_\mathrm{in}} \]
\[ \dot{n}_{i_2} = (1-t_{i1}) \dot{n}_{i_\mathrm{in}} \]
If the split fraction is the same for all species, then the separator is just a divider. The split fractions must be different for the different species to effect a separation.
The Takeaways
- In a mixer the molar or mass flow in the output stream is the sum of the molar or mass input flows.
- In a divider the molar or mass flow in the input stream is the sum of the molar or mass output flows. Also, the composition of each stream is the same.
- In a separator the molar or mass flow of a given species in an output stream is the split fraction for that species and stream times the molar or mass flow of that species in the input stream.
Thanks for watching!
The Full Story companion video is in the link in the upper left. The next video in the series, When You Saw It, What Was the Extent of Your Reaction? Part 1, is in the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.

