Recycling Before
Recycling Was Cool
DOFPro Team

Recycle
- One of the basic techniques in process flow diagrams
- The implications on the equations
- Used extensively in chemical engineering
- Used to control or optimize a process
Recycle
- No reactions go to completion.
- No separators have perfect separation.
- Wasted reagents are wasted money.
- Recycle (like feedback) can improve performance.
No Recycle
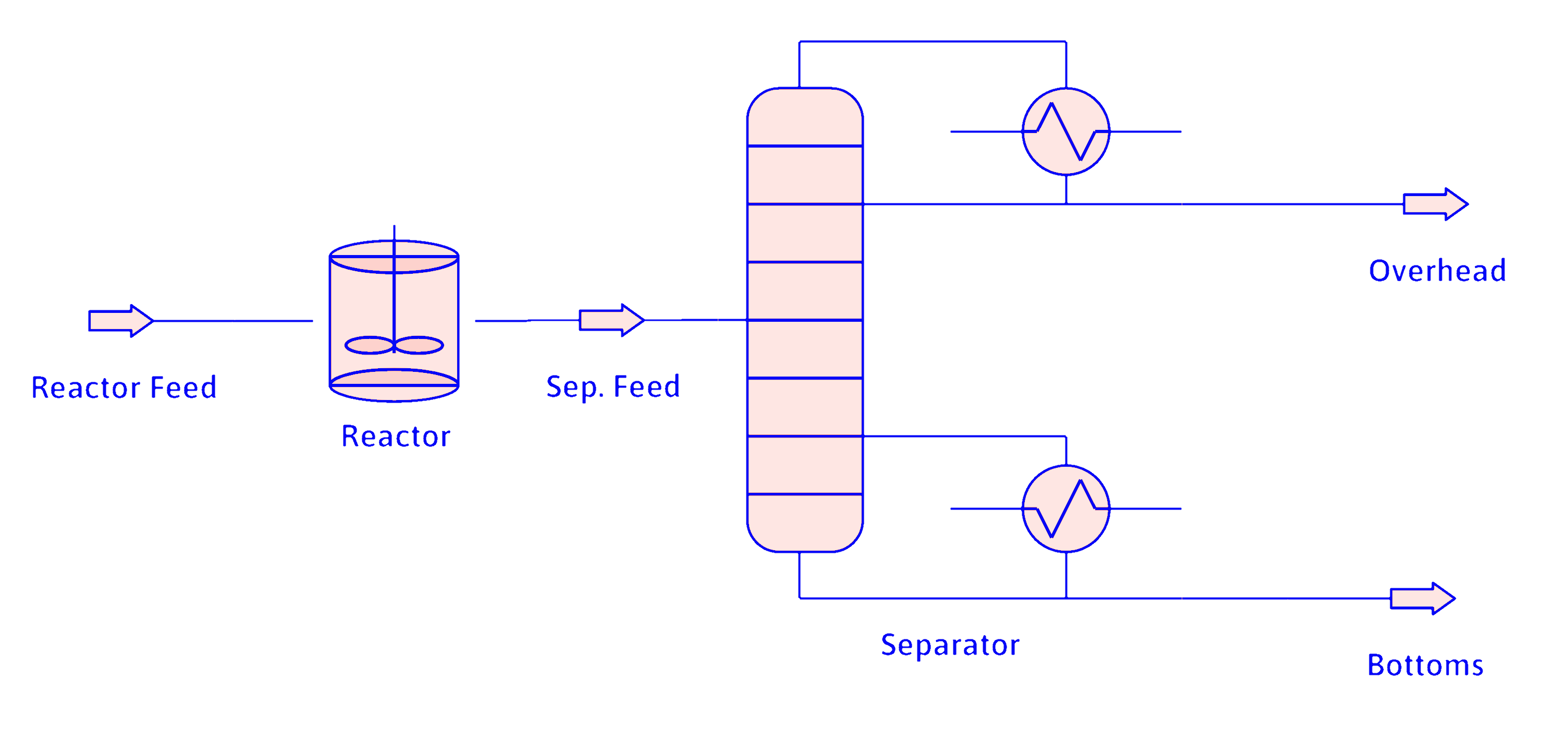
Recycle
Recycle Comparison – No Recycle

\(\enclose{circle}{1}\)
\(\enclose{circle}{2}\)
\(\enclose{circle}{4}\)
\(\enclose{circle}{3}\)
\(\mathrm{A}\)
\(\begin{array}{c}\mathrm{A}\\\mathrm{B}\\\mathrm{C}\end{array}\)
\(\begin{array}{c}\mathrm{A}\\\mathrm{B}\\\mathrm{C}\end{array}\)
\(\begin{array}{c}\mathrm{A}\\\mathrm{B}\end{array}\)
\[ \mathrm{C_3H_8 \rightarrow C_3H_6 + H_2} \]
\[ \mathrm{A \rightarrow B + C} \]
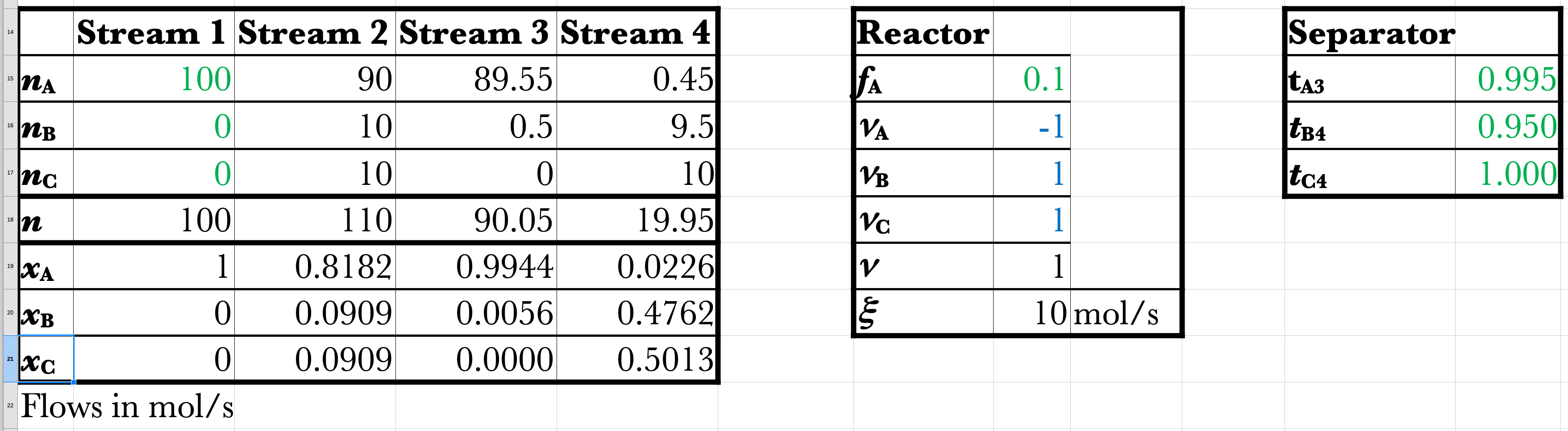
Given: Feed \(\mathrm{100\ mol/s\ A}\)
Reactor: \(f_\mathrm{A}=0.10\ (10\%)\)
Separator: \(t_\mathrm{A3} = 0.995,\ t_\mathrm{B4} = 0.950,\ t_\mathrm{C4} = 1\).
No Recycle (cont.)
Material Balances
Reactor
\[ \dot{\xi} = f_\mathrm{A} \dot{n}_\mathrm{A1} /(-\nu_\mathrm{A}) \]
\[ \dot{\xi} = (0.10)(100) = 10\ \mathrm{mol/s} \]
\[ \dot{n}_{i2} = \dot{n}_{i1} + \nu_i \dot{\xi} \]
\[ \dot{n}_\mathrm{A2} = 100 - 10 = 90\ \mathrm{mol/s} \]
\[ \dot{n}_\mathrm{B2} = 0 + 10 = 10\ \mathrm{mol/s} \]
\[ \dot{n}_\mathrm{C2} = 0 + 10 = 10\ \mathrm{mol/s} \]
Separator
\[ \dot{n}_{i3} = t_{i3} \dot{n}_{i2},\ \ \ \ \ t_{i4} = 1 - t_{i3} \]
\[ \dot{n}_\mathrm{A3}=(0.995)(90) = 89.55\ \mathrm{mol/s} \]
\[ \dot{n}_\mathrm{B3}=(0.05)(10) = 0.5\ \mathrm{mol/s} \]
\[ \dot{n}_\mathrm{C3}=(0)(10) = 0\ \mathrm{mol/s} \]
\[ \dot{n}_\mathrm{A4} = \dot{n}_\mathrm{A2} - \dot{n}_\mathrm{A3} = 0.45\ \mathrm{mol/s} \]
\[ \dot{n}_\mathrm{B4} = \dot{n}_\mathrm{B2} - \dot{n}_\mathrm{B3} = 9.5\ \mathrm{mol/s} \]
\[ \dot{n}_\mathrm{C4} = \dot{n}_\mathrm{C2} - \dot{n}_\mathrm{C3} = 10\ \mathrm{mol/s} \]
No Recycle Spreadsheet

\(\enclose{circle}{1}\)
\(\enclose{circle}{2}\)
\(\enclose{circle}{4}\)
\(\enclose{circle}{3}\)
\(\mathrm{A}\)
\(\begin{array}{c}\mathrm{A}\\\mathrm{B}\\\mathrm{C}\end{array}\)
\(\begin{array}{c}\mathrm{A}\\\mathrm{B}\\\mathrm{C}\end{array}\)
\(\begin{array}{c}\mathrm{A}\\\mathrm{B}\end{array}\)
\[ \mathrm{C_3H_8 \rightarrow C_3H_6 + H_2} \]
\[ \mathrm{A \rightarrow B + C} \]
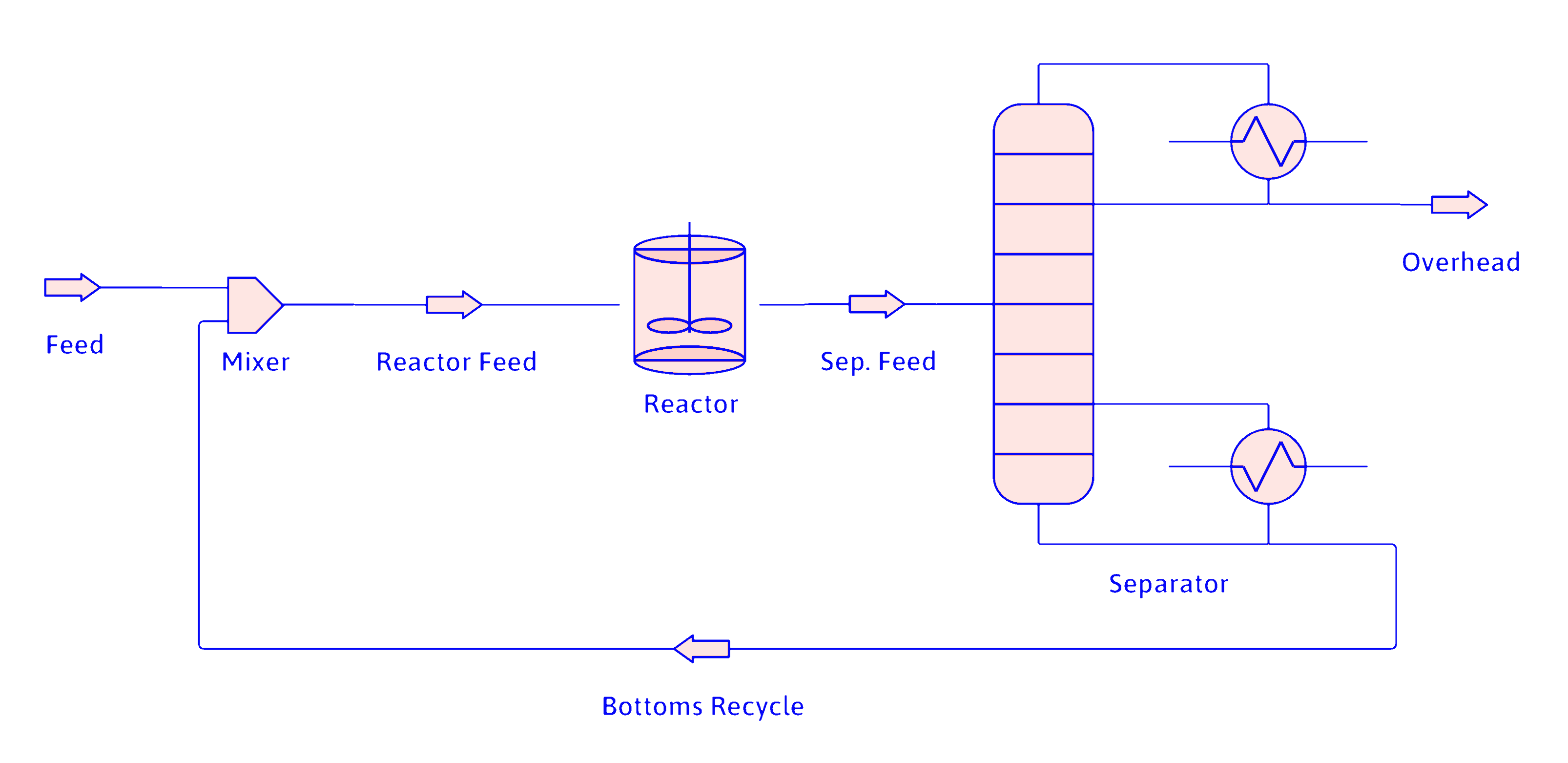
Same System with Recycle

\(\enclose{circle}{1}\)
\(\enclose{circle}{2}\)
\(\enclose{circle}{4}\)
\(\enclose{circle}{3}\)
\(\enclose{circle}{5}\)
\(\mathrm{A}\)
\(\begin{array}{c}\mathrm{A}\\\mathrm{B}\\\mathrm{C}\end{array}\)
Reactor
\(\dot{\xi} = 0.10 \dot{n}_\mathrm{A5}\ \mathrm{mol/s}\)
\(\dot{n}_\mathrm{A2} = \dot{n}_\mathrm{A5} - 0.1 \dot{n}_\mathrm{A5} = 0.90 \dot{n}_\mathrm{A5}\)
Separator
\(\dot{n}_\mathrm{A3} = 0.995 \dot{n}_\mathrm{A2}\)
\(\dot{n}_\mathrm{A4} = 0.005 \dot{n}_\mathrm{A2}\)
Mixer
\(\dot{n}_\mathrm{A5} = 100 + \dot{n}_\mathrm{A3}\)
Material balance for component A
Four equations in four unknowns, \(\dot{n}_\mathrm{A2}\), \(\dot{n}_\mathrm{A3}\), \(\dot{n}_\mathrm{A4}\), and \(\dot{n}_\mathrm{A5}\).
Same System with Recycle (cont.)
Successive substitution on \(\mathrm{A}\) for solution
\(\dot{n}_\mathrm{A2} = 0.90 \dot{n}_\mathrm{A5}\)
\(\dot{n}_\mathrm{A3} = 0.995 \dot{n}_\mathrm{A2} = 0.995(0.90 \dot{n}_\mathrm{A5})\)
\(\dot{n}_\mathrm{A5} = 100 + \dot{n}_\mathrm{A3}= 100 + 0.995(0.90 \dot{n}_\mathrm{A5})\)
\(\dot{n}_\mathrm{A5}[1-0.995(0.90)] = 100\)
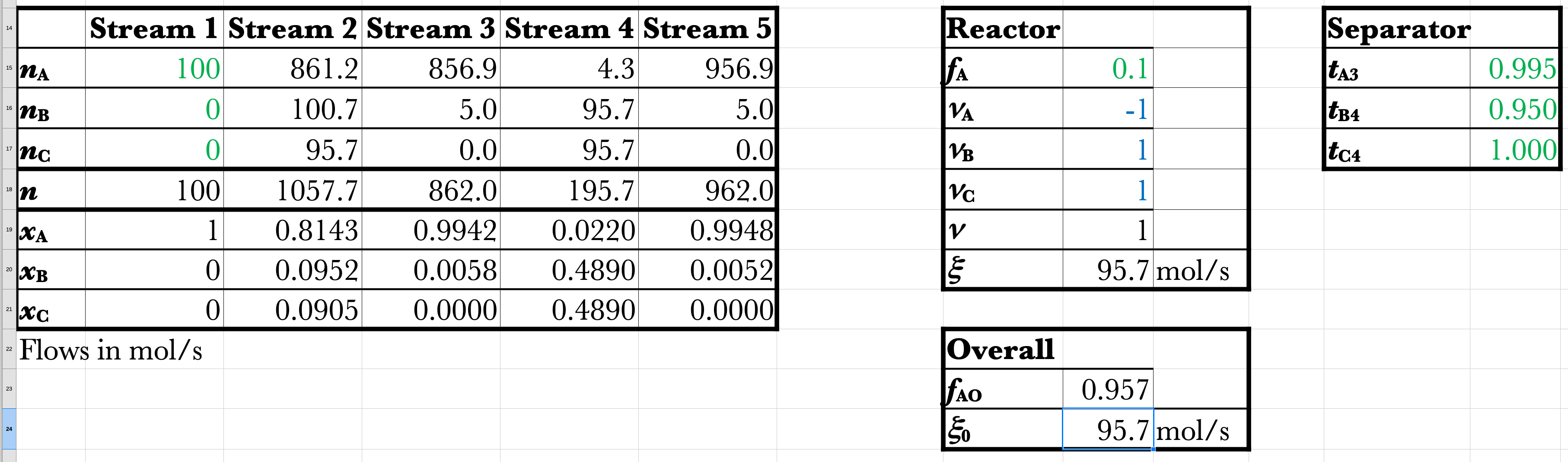
\(\dot{n}_\mathrm{A5} = \frac{100}{[1-0.995(0.90)]} = 956.9\)
Same System with Recycle (cont.)
Use similar approach for B and C
Solution:
\[ \dot{n}_\mathrm{A2} = 861\ \mathrm{mol/s} \]
\[ \dot{n}_\mathrm{A3} = 857\ \mathrm{mol/s} \]
\[ \dot{n}_\mathrm{A4} = 4.3\ \mathrm{mol/s} \]
\[ \dot{n}_\mathrm{A5} = 957\ \mathrm{mol/s} \]
\[ \dot{n}_\mathrm{B2} = 101\ \mathrm{mol/s} \]
\[ \dot{n}_\mathrm{B3} = 5.0\ \mathrm{mol/s} \]
\[ \dot{n}_\mathrm{B4} = 96\ \mathrm{mol/s} \]
\[ \dot{n}_\mathrm{B5} = 5.0\ \mathrm{mol/s} \]
\[ \dot{n}_\mathrm{C2} = 96\ \mathrm{mol/s} \]
\[ \dot{n}_\mathrm{C3} = 0\ \mathrm{mol/s} \]
\[ \dot{n}_\mathrm{C4} = 96\ \mathrm{mol/s} \]
\[ \dot{n}_\mathrm{C5} = 0\ \mathrm{mol/s} \]
Recycle Example (cont.)
What did recycle buy us?
Overall fractional conversion \(= \frac{\dot{n}_\mathrm{A1} - \dot{n}_\mathrm{A4}}{\dot{n}_\mathrm{A1}}\)
\[ f_\mathrm{A} = \frac{100-4.3}{100} = 0.957\ \mathrm{vs.\ 0.10} \]
Overall extent of reaction (same as extent of reaction in the reactor, unlike fractional conversion)
\[ \dot{\xi}_\mathrm{overall} = 100-4.3 = 95.7\ \mathrm{mol/s\ vs.\ 10\ mol/s} \]
What did it cost?
\[ \dot{n}_\mathrm{2} = 1058\ \mathrm{mol/s\ vs.\ 110\ mol/s} \]
\[ \dot{n}_\mathrm{3} = 862\ \mathrm{mol/s\ vs.\ 90\ mol/s} \]
Recycle Example Spreadsheet

\(\enclose{circle}{1}\)
\(\enclose{circle}{2}\)
\(\enclose{circle}{4}\)
\(\enclose{circle}{3}\)
\(\enclose{circle}{5}\)
\(\mathrm{A}\)
\(\begin{array}{c}\mathrm{A}\\\mathrm{B}\\\mathrm{C}\end{array}\)
\[ \mathrm{C_3H_8 \rightarrow C_3H_6 + H_2} \]
\[ \mathrm{A \rightarrow B + C} \]
Purge
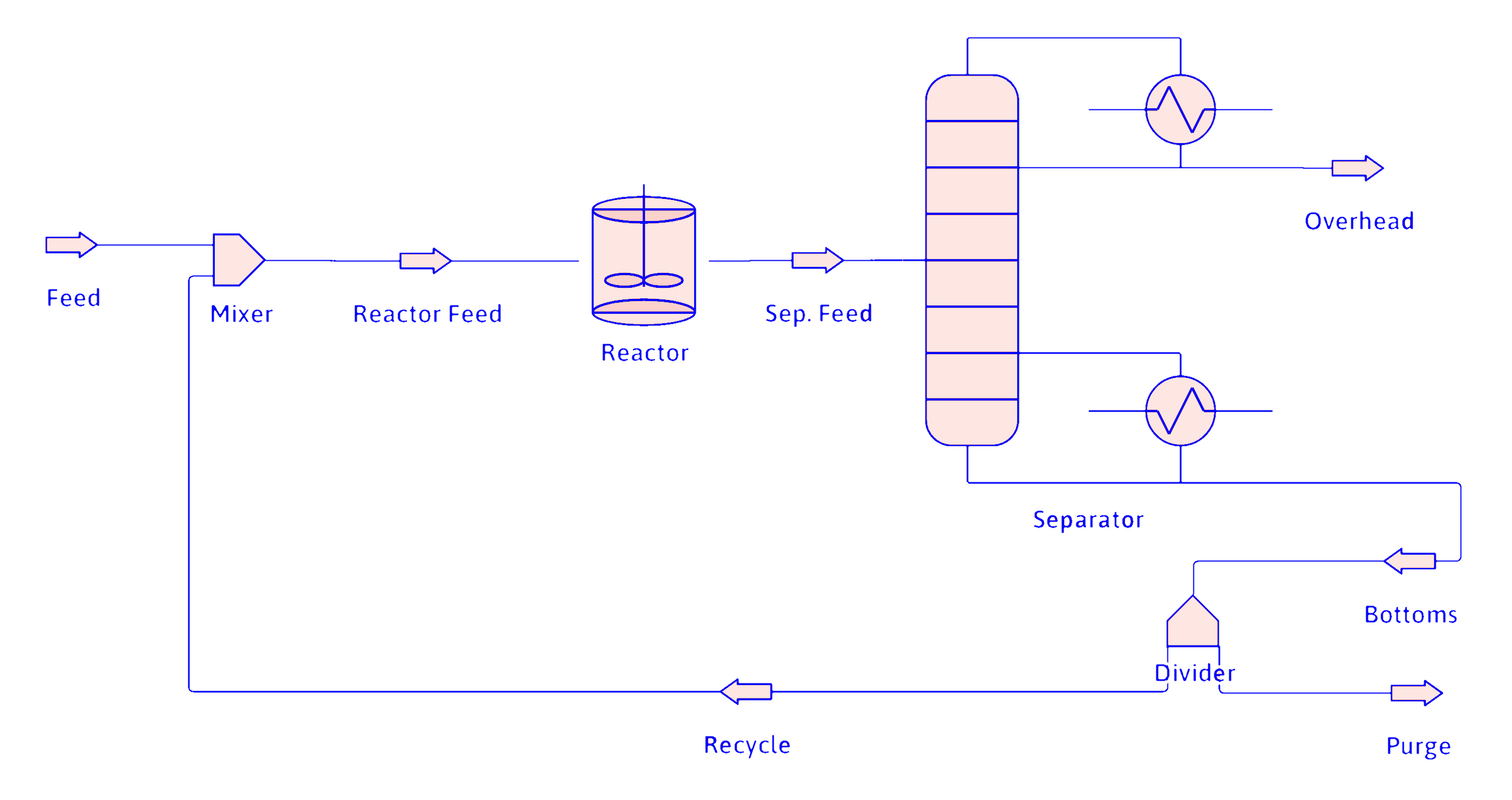
- Often necessary to remove trace species in the feed
- Used with recycle when the separator has a split fraction of zero on a trace species
- Less expensive than an additional separator
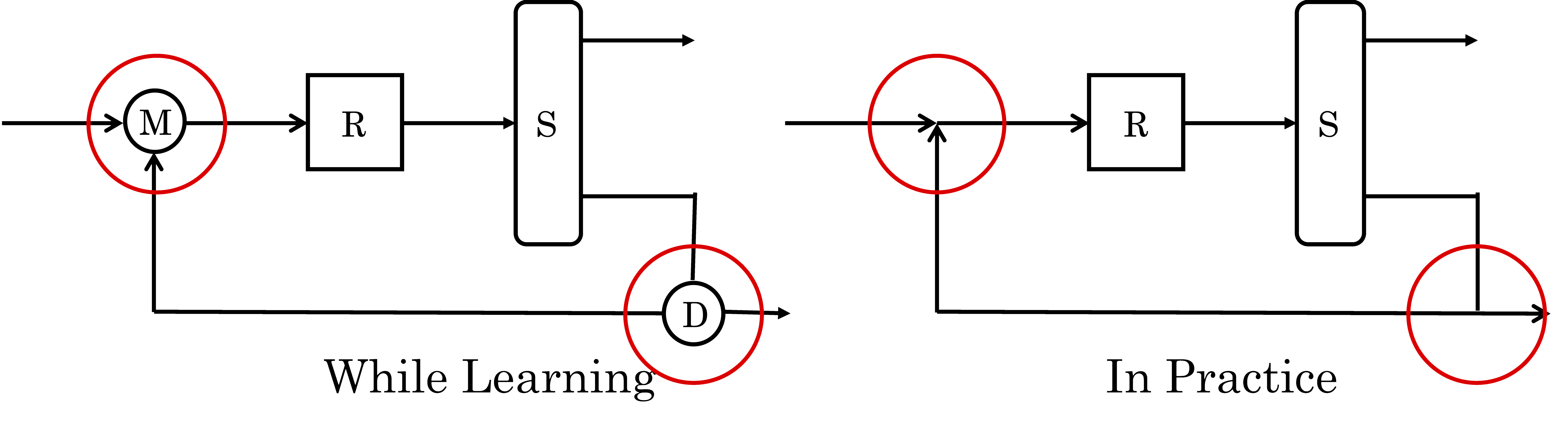
Actual vs. Instructional PFD
Mixers with only two inputs and dividers with only two outputs are almost never shown on actual PFDs. They are represented by piping tees, instead.
The Takeaways
- Recycle is used extensively in the chemical process industry to improve overall efficiency and cut down on waste.
- Analyzing a recycle loop almost always requires a symbolic solution around the loop before the equations can be solved.
- A purge line is often required in a recycle loop to prevent the buildup of trace components.
- Mixers and dividers are often represented by tee-junctions in PFDs.
Thanks for watching!
The Full Story companion video is in the link in the upper left. The companion video in the series, Leaving Radiator Springs In the Dust, is in the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.

