Leaving Radiator Springs
in the Dust
Just the Facts
DOFPro Team

Recycle and Bypass
- Some of the basic techniques for process flow diagrams
- Implications on the modeling equations that you use
- Used extensively in chemical engineering
- Used to control or optimize a process as part of a larger system
Bypass
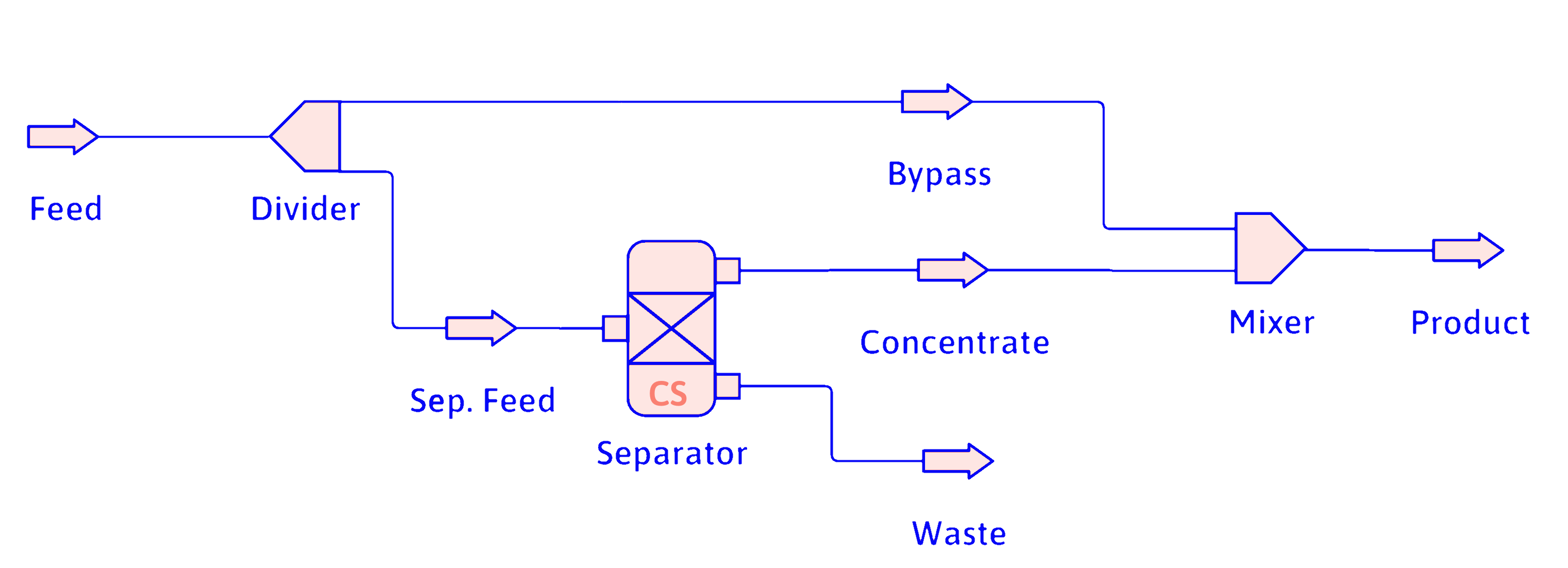
- Less common than recycle
- Common example is fruit-juice concentrator.
Simplifying PFDs
Flowsheets can be simplified by drawing a surrounding box and only dealing with the inputs and outputs. For example, the process flowsheet for the dehydrogenation of propane to propene.
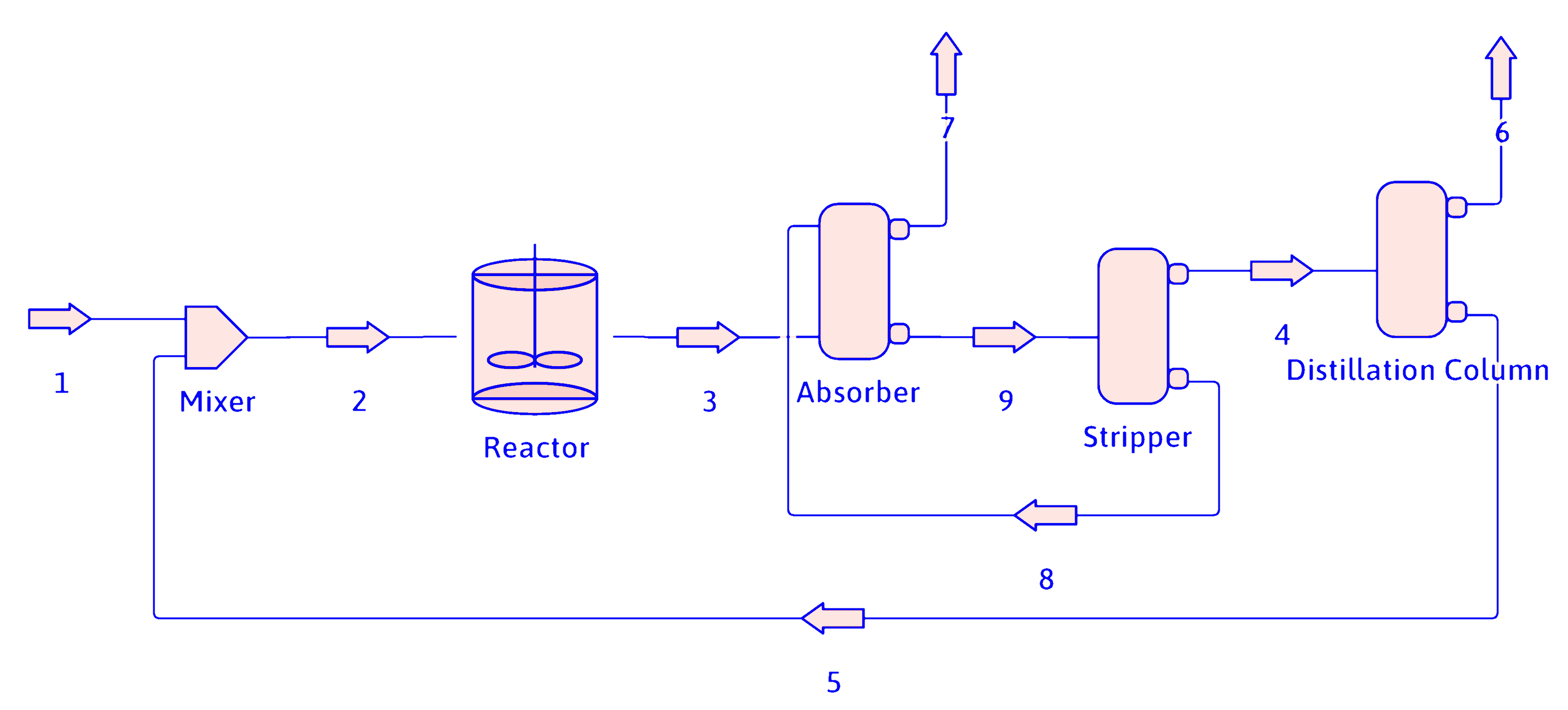
propane
hydrogen
methane
propane
propene
Simplifying PFDs
Flowsheets can be simplified by drawing a surrounding box and only dealing with the inputs and outputs. For example, the process flowsheet for the dehydrogenation of propane to propene.
propane
hydrogen
methane
propane
propene
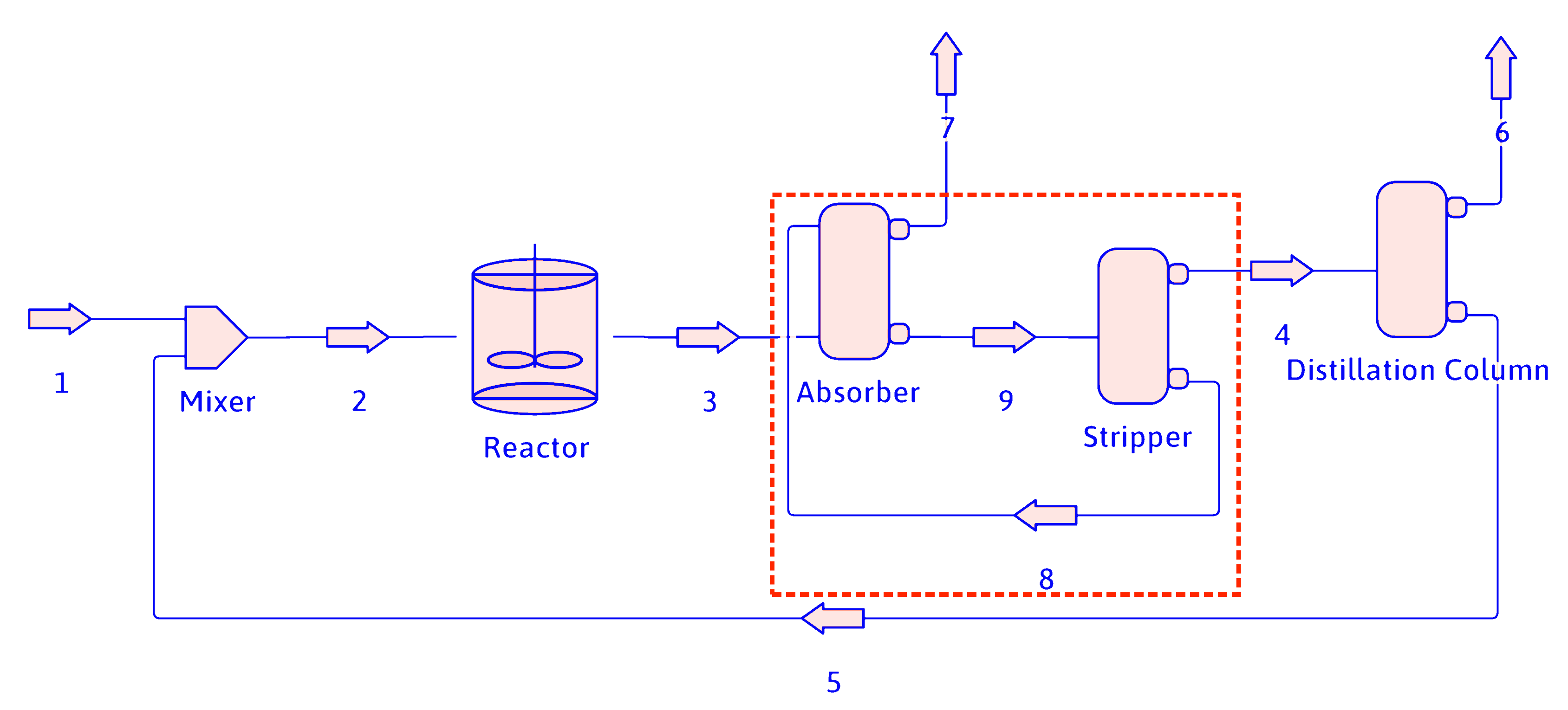
Simplifying PFDs (cont.)
The absorber and stripper can be combined into one separator for calculation purposes.
propane
hydrogen
methane
propane
propene
Simplifying PFDs (cont.)
The absorber and stripper can be combined into one separator for calculation purposes.
propane
hydrogen
methane
propane
propene
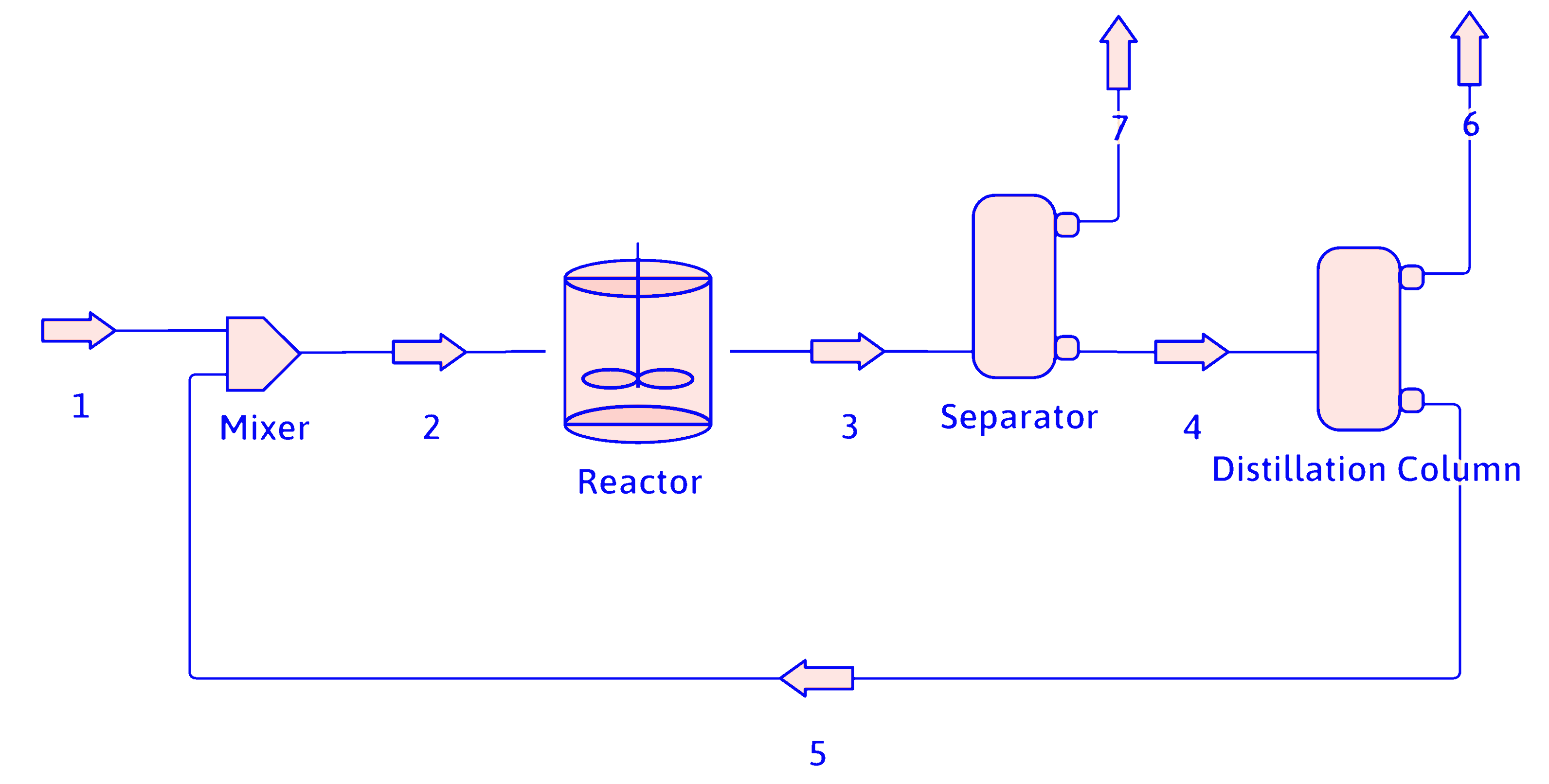
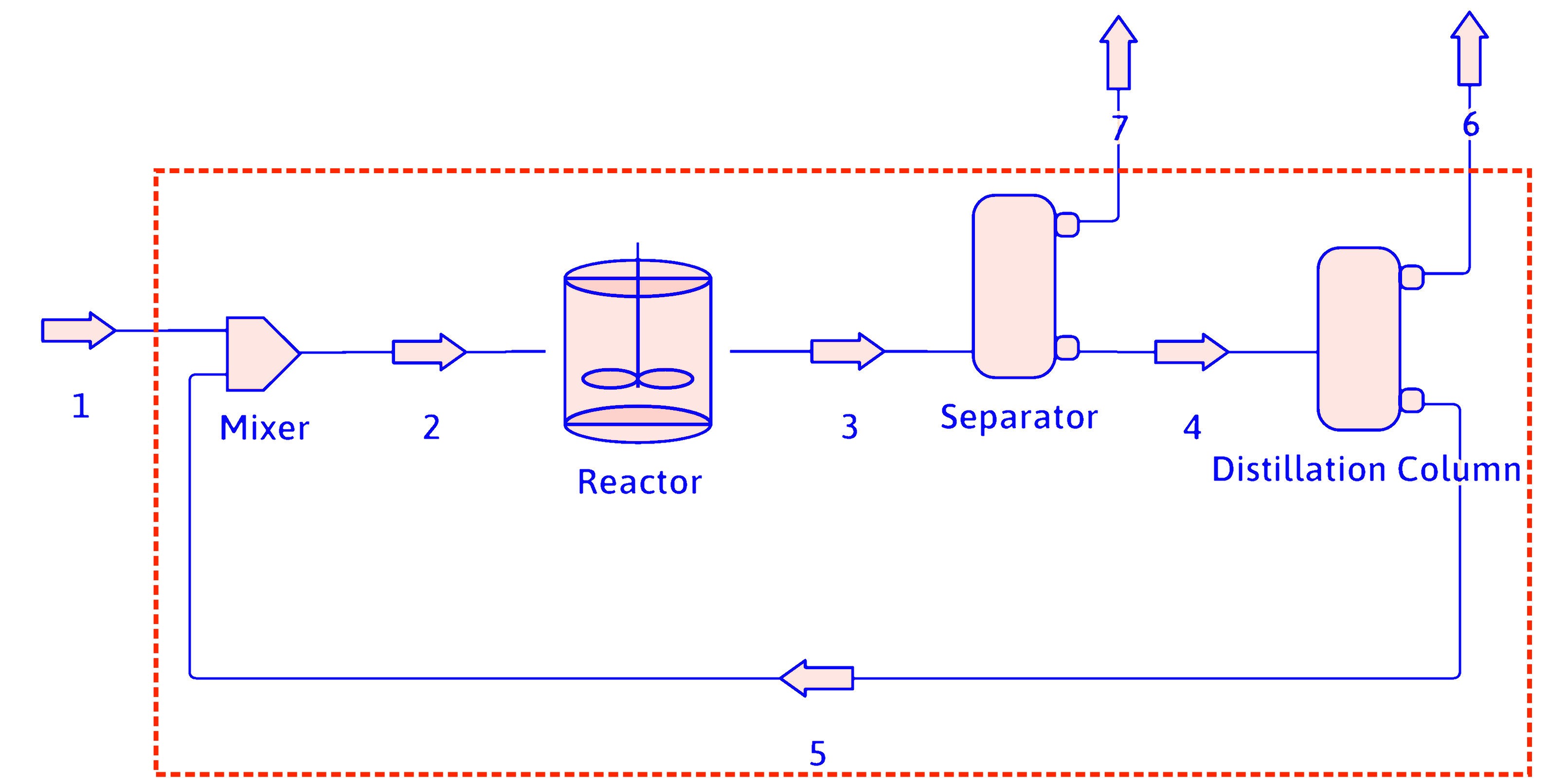
Simplifying PFDs (cont.)
The reactor, separator, distillation column, and recycle loop can be combined into an overall process block for calculation purposes.
propane
hydrogen
methane
propane
propene
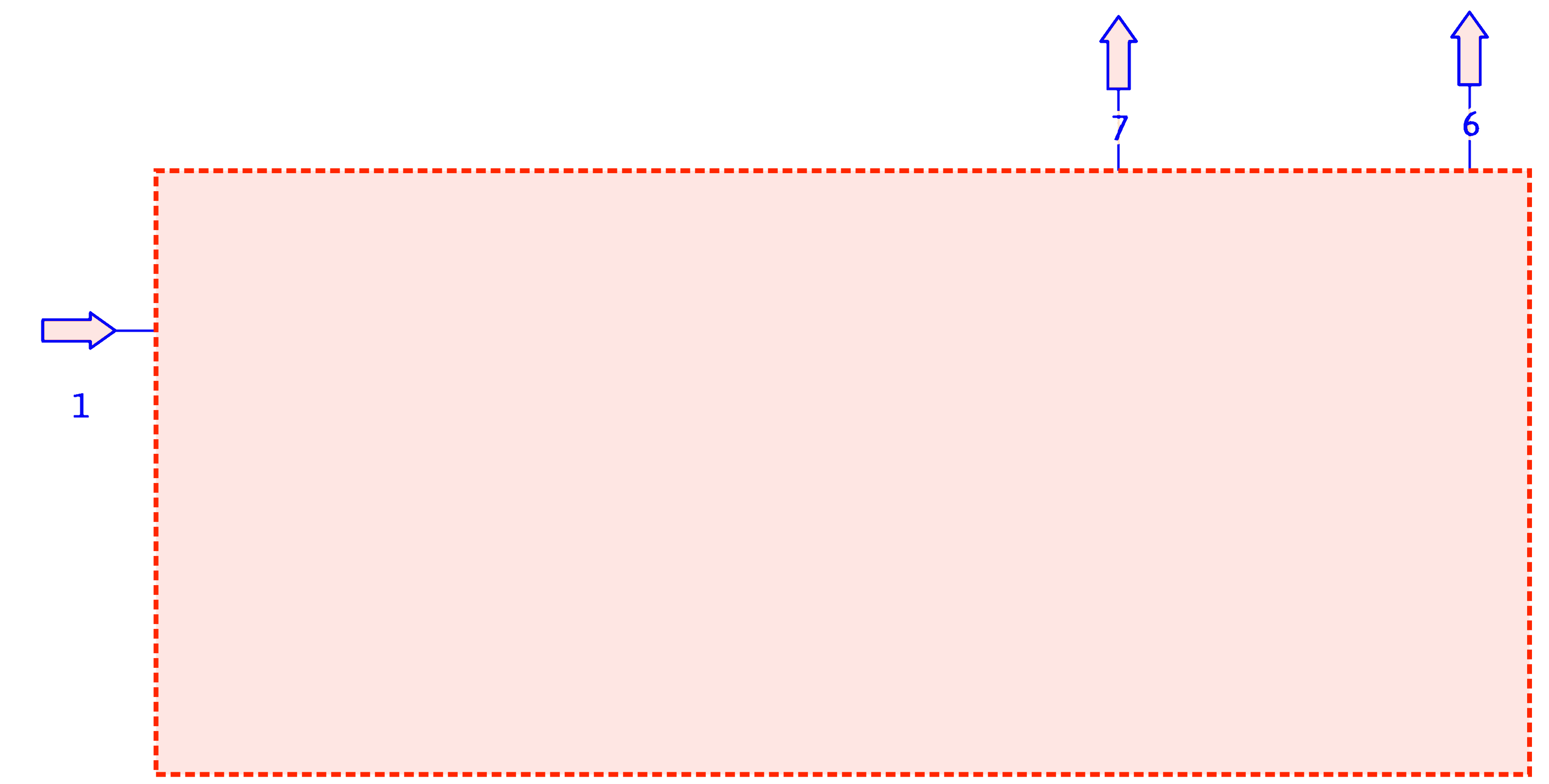
Simplifying PFDs (cont.)
The reactor, separator, distillation column, and recycle loop can be combined into an overall process block for calculation purposes.
\(\enclose{circle}{1}\)
\(\enclose{circle}{7}\)
\(\enclose{circle}{6}\)
propane
hydrogen
methane
propane
propene
Example of Simplifying

propane
hydrogen
methane
propane
propene
Let’s look at the calculations for this slightly more realistic dehydrogenation process.
Product flow rate – \(100\ \mathrm{mol/s}\)
Product stream 6 – \(98\%\) propene
Recycle stream 5 – \(97\%\) propane
\(\mathrm{C_3H_8 - A}\)
\(\mathrm{C_3H_6 - B}\)
\(\mathrm{CH_4 - C}\)
\(\mathrm{H_2 - D}\)
\(\mathrm{C_3H_8 \rightarrow C_3H_6 + H_2}\)
\(\mathrm{C_3H_8 + 2H_2 \rightarrow 3CH_4}\)
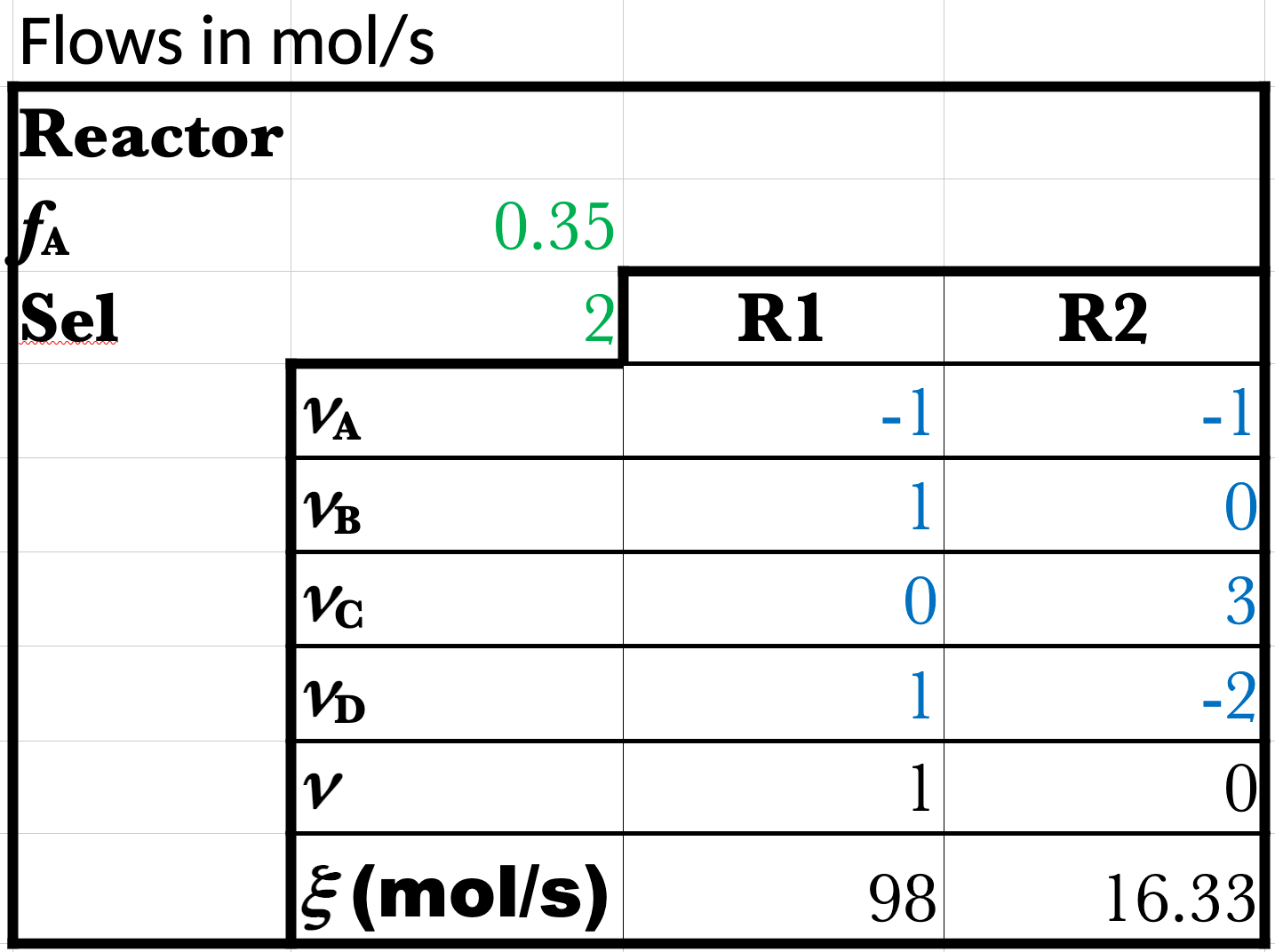
\(f_\mathrm{C_3H_8} = 0.35\) (in reactor)
\(S = \frac{\dot{n}_{\mathrm{C_3H_6}_\text{–gen}}}{\dot{n}_{\mathrm{CH_3}_\text{–gen}}} = 2\) (in reactor)
Example of Simplifying (cont.)

\(\enclose{circle}{1}\)
\(\enclose{circle}{7}\)
\(\enclose{circle}{6}\)
propane
hydrogen
methane
propane
propene
\(\mathrm{C_3H_8 - A}\)
\(\mathrm{C_3H_6 - B}\)
\(\mathrm{CH_4 - C}\)
\(\mathrm{H_2 - D}\)
If there is only one reactor in the process, the extents of reaction overall have to be the same as the extents of reaction in the reactor.
Propene is produced by reaction 1 only, so
\(\dot{\xi}_1 = (0.98)(100\ \mathrm{mol/s}) = 98\ \mathrm{mol/s}\)
\(\dot{n}_\text{B–gen} =\dot{n}_\mathrm{B6} - \dot{n}_\mathrm{B1} = \dot{n}_\mathrm{B6}\)
\(\dot{n}_\text{C–gen} =\dot{n}_\mathrm{C7} - \dot{n}_\mathrm{C1} = \dot{n}_\mathrm{C7}\)
\(S = \frac{\dot{n}_\text{B–gen}}{\dot{n}_\text{C–gen}} = \frac{\dot{n}_\mathrm{B6}}{\dot{n}_\mathrm{C7}} = \frac{\nu_\mathrm{B,1} \dot{\xi}_1}{\nu_\mathrm{C,2} \dot{\xi}_2} = \frac{\dot{\xi}_1}{3 \dot{\xi}_2} = 2\)
\(\dot{\xi}_2 = \frac{\dot{\xi}_1}{6} = \frac{98}{6}\)
Example of Simplifying (cont.)


\(\enclose{circle}{1}\)
\(\enclose{circle}{2}\)
\(\enclose{circle}{3}\)
\(\enclose{circle}{7}\)
\(\enclose{circle}{6}\)
\(\mathrm{A,\ B}\)
\(\mathrm{A,\ B,\ C,\ D}\)
Reactor
propane
hydrogen
methane
propane
propene
\(\mathrm{C_3H_8 - A}\)
\(\mathrm{C_3H_6 - B}\)
\(\mathrm{CH_4 - C}\)
\(\mathrm{H_2 - D}\)
Mole balance on \(\mathrm{A}\)
\(\dot{n}_\mathrm{A6} = \dot{n}_\mathrm{A1} + \nu_\mathrm{A1} \dot{\xi}_1 + \nu_\mathrm{A2} \dot{\xi}_2\)
\(2 = \dot{n}_\mathrm{A1} - 98 - 16.3\)
\(\implies \dot{n}_\mathrm{A1} = 116.3\ \mathrm{mol/s}\)
\(f_\mathrm{A} = 1 - \frac{\dot{n}_\mathrm{A3}}{\dot{n}_\mathrm{A2}}\)
\(\implies \dot{n}_\mathrm{A3} = \dot{n}_\mathrm{A2}(1-f_\mathrm{A})\)
\(\dot{n}_\mathrm{A3} = \dot{n}_\mathrm{A2} + \nu_\mathrm{A1} \dot{\xi}_1 + \nu_\mathrm{A2} \dot{\xi}_2 = \dot{n}_\mathrm{A2} - \dot{\xi}_1 - \dot{\xi}_2\)
\(\dot{n}_\mathrm{A2}(1-f_\mathrm{A}) = \dot{n}_\mathrm{A2} - \dot{\xi}_1 - \dot{\xi}_2\)
\(\dot{n}_\mathrm{A2} = \frac{\dot{\xi}_1 + \dot{\xi}_2}{f_\mathrm{A}} = \frac{98 + \frac{98}{6}}{0.35} = 326.7\)
Example of Simplifying (cont.)

propane
hydrogen
methane
propane
propene
\(\mathrm{C_3H_8 - A}\)
\(\mathrm{C_3H_6 - B}\)
\(\mathrm{CH_4 - C}\)
\(\mathrm{H_2 - D}\)
Material Balance on \(\mathrm{A}\) around each element
M: \(\dot{n}_\mathrm{A2} = 116.3 + \dot{n}_\mathrm{A5}\)
R: \(\dot{n}_\mathrm{A3} = 0.65 \dot{n}_\mathrm{A2}\)
S1: \(\dot{n}_\mathrm{A4} = \dot{n}_\mathrm{A3} - \dot{n}_\mathrm{A7} = \dot{n}_\mathrm{A3}\)
S2: \(2 = \dot{n}_\mathrm{A4} - \dot{n}_\mathrm{A5}\)
Solution
\(\dot{n}_\mathrm{A2} = 326.6\ \mathrm{mol/s}\)
\(\dot{n}_\mathrm{A3} = 212.3\ \mathrm{mol/s}\)
\(\dot{n}_\mathrm{A4} = 212.3\ \mathrm{mol/s}\)
\(\dot{n}_\mathrm{A5} = 210.3\ \mathrm{mol/s}\)
Recycle \(97\%\ \mathrm{A}\)
\(\therefore\ \dot{n}_5 = \frac{210.3}{0.97} = 216.8\ \mathrm{mol/s}\)
Example of Simplifying Spreadsheet

propane
hydrogen
methane
propane
propene
\(\mathrm{C_3H_8 - A}\)
\(\mathrm{C_3H_6 - B}\)
\(\mathrm{CH_4 - C}\)
\(\mathrm{H_2 - D}\)
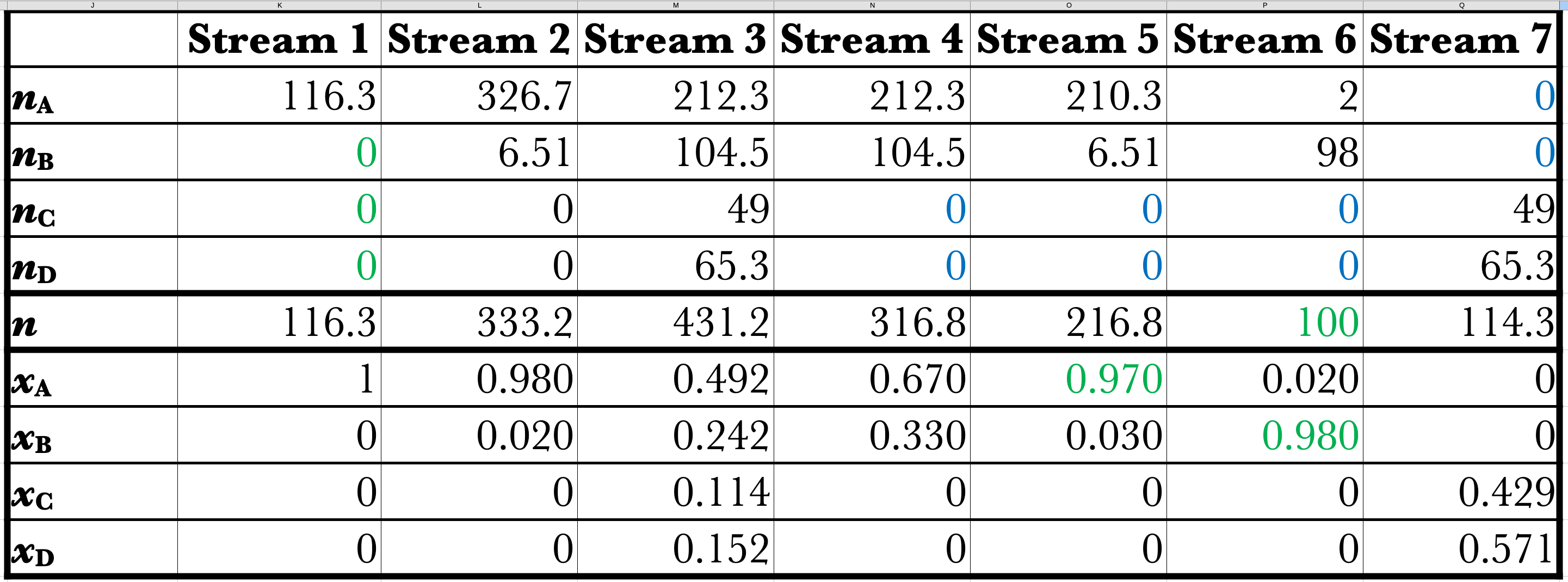
The Takeaways
- Bypass is less common than recycle, but can be analyzed the same way.
- Often the quickest way to analyze a PFD is to do overall balances and partial balances first.
- If there is only one reactor (with possibly multiple reactions) in a process, the extents of reaction are the same for the overall process and for the reactor.
- If there is only one reactor (with possibly multiple reactions) in a process with recycle, the overall fractional conversion is almost certainly much higher than the fractional conversion in the reactor.
Thanks for watching!
The Full Story companion video is in the link in the upper left. The companion video in the series, The Symbolic Meaning of Recycling, is in the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.

