React But Maintain Your Equilibrium
Part 2
DOFPro Team

From Part 1
\[ K\ (\mathrm{1\ bar})^\nu P^{-\nu} =\prod (y_i)^{\nu_i} \]
\[ K = \exp \left( - \frac{\Delta G^\circ}{RT}\right) \]
Example
Carbon monoxide and oxygen are fed in equimolar ratios to an isothermal combustor. If the reaction proceeds to equilibrium calculate the fractional conversion of \(\mathrm{CO}\) and \(\mathrm{O_2}\) as the reactor temperature varies from \(300\ \mathrm{K}\) to \(2500\ \mathrm{K}\).
Data
\(\ \ \ \ \mathrm{CO +\frac{1}{2} O_2 = CO_2}\)
\(\ \ \ \ \Delta G^\circ = -281,885 + 85.678 T\ \mathrm{[J/mol]}\)
Solution
\[ \ln K = - \frac{\Delta G^\circ}{RT} = \frac{33,903}{T} - 10.3047 \]
\[ \nu_\mathrm{CO} = -1,\ \ \ \ \nu_{\mathrm{O_2}} = -0.5,\ \ \ \ \nu_{\mathrm{CO_2}} = 1 \]
\[ \nu = -1 - 0.5 + 1 = -0.5 \]
\[ K\ (\mathrm{1\ bar})^\nu P^{-\nu} =\prod (y_i)^{\nu_i} \]
\[ K\ (\mathrm{1\ bar})^{-0.5} P^{0.5} = \frac{y_{\mathrm{CO}_2}}{y_{\mathrm{CO}} y^{0.5}_{\mathrm{O}_2}} \]
Solution (cont.)
\[ y_\mathrm{CO} = \frac{n_\mathrm{CO_{out}}}{n_\mathrm{out}} = \frac{n_\mathrm{CO_{in}} + \nu_\mathrm{CO}\xi}{n_\mathrm{in} + \nu \xi} \]
\[ y_\mathrm{CO_2} = \frac{n_\mathrm{CO_{2_{out}}}}{n_\mathrm{out}} = \frac{n_\mathrm{CO_{2_{in}}} + \nu_\mathrm{CO_2}\xi}{n_\mathrm{in} + \nu \xi} \]
\[ y_\mathrm{O_2} = \frac{n_\mathrm{O_{2_{out}}}}{n_\mathrm{out}} = \frac{n_\mathrm{O_{2_{in}}} + \nu_\mathrm{O_2}\xi}{n_\mathrm{in} + \nu \xi} \]
What is missing?
\(P\) and either \(n_\mathrm{O_{2_{in}}}\) or \(n_\mathrm{CO_{in}}\).
Pick a basis of one mole.
Assume \(P = 1\ \mathrm{bar}\).
Solution (cont.)
\[ \frac{\left(\frac{0+\xi}{2-0.5\xi}\right)}{\left(\frac{1-\xi}{2-0.5\xi}\right)\left(\frac{1-0.5\xi}{2-0.5\xi}\right)^{0.5}} = K(\mathrm{1\ bar})^{-0.5}(\mathrm{1\ bar})^{0.5} \]
\[
\frac{\left( 2 - 0.5 \xi \right)^{0.5}}{\left( 1-0.5 \xi \right)^{0.5}}\frac{\xi}{1-\xi} = K
\]
For \(\xi \rightarrow 0\), \(\xi = K/\sqrt{2}\).
For \(\xi \rightarrow 1\), \(\xi = \frac{K / \sqrt{3}}{1 + K / \sqrt{3}}\) or \(1 - \xi = \frac{1}{1+K/\sqrt{3}}\).
Results
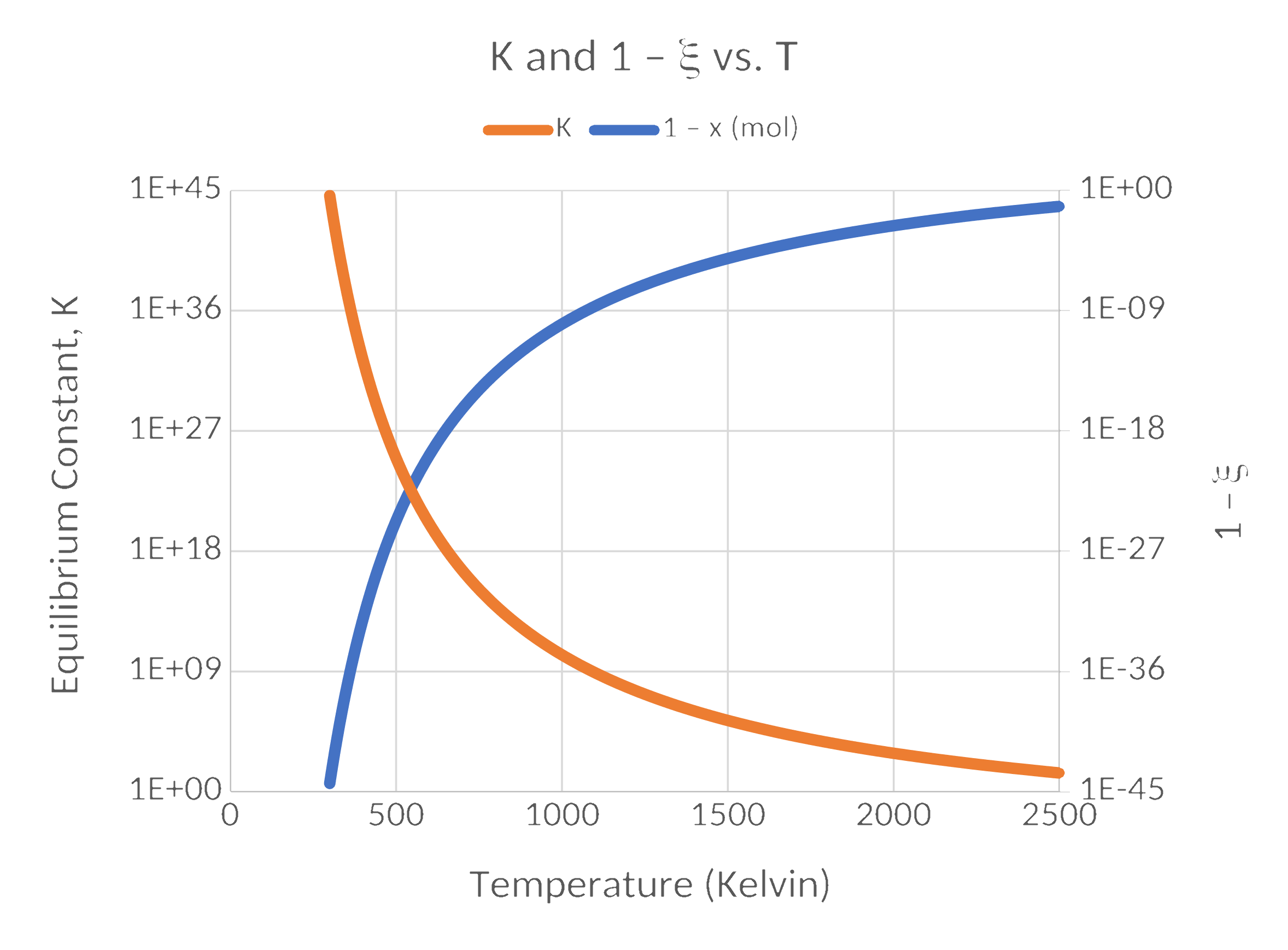
Results (cont.)
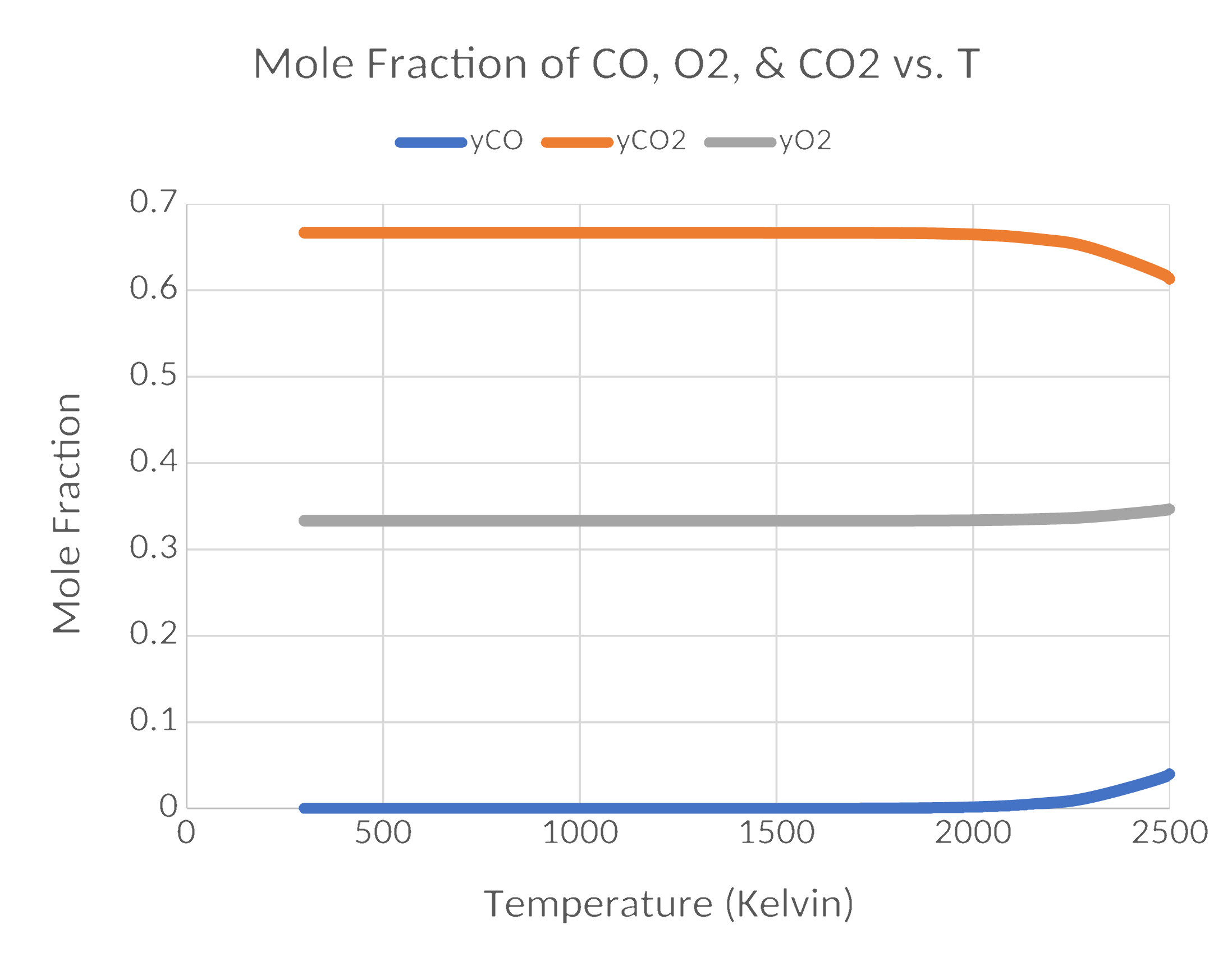
Results (cont.)
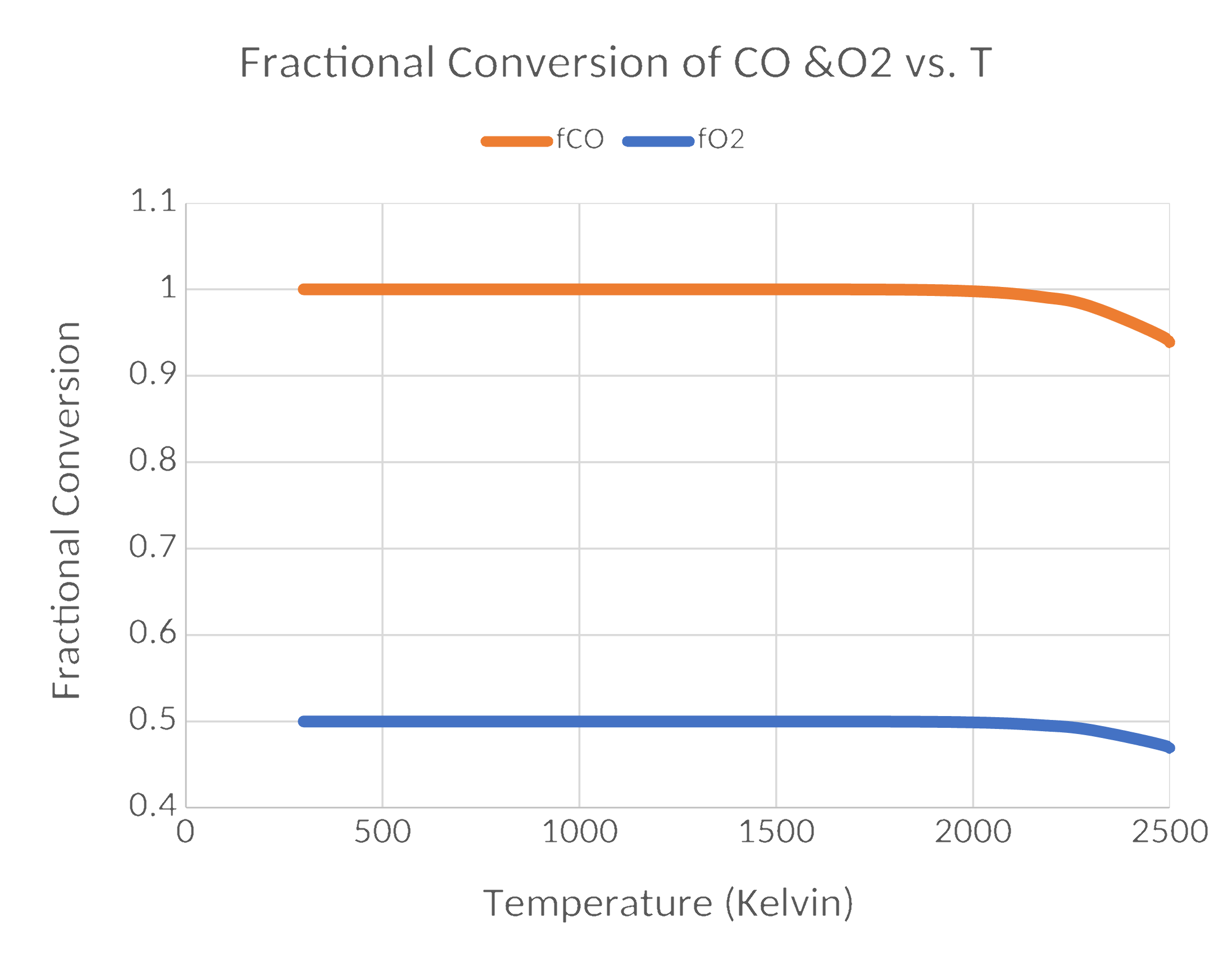
The Takeaways
- For ideal gases the equilibrium extent of reaction can be calculated from the equations for the equilibrium constant, \(K\) in terms of mole fractions and temperatures.
- Equilibrium constants change by orders of magnitude for relatively small changes in temperature.
- Solutions of these types of equations often require finding numerical roots of nonlinear equations.
- Chemical equilibrium predictions without kinetic information can be seriously wrong.
Thanks for watching!
The Full Story companion video is in the link in the upper left. The previous companion video, React But Maintain Your Equilibrium Part 1, in the series, is in the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.

