Flame On, Torch!
DOFPro Team

Combustion Reactions
Combustion reactions are rapid oxidation reactions. One usually burns a fuel (typically coal, oil or natural gas) in air (or rarely pure oxygen). The principal atomic species in combustion reactions are:
- carbon,
- hydrogen,
- sulfur, and
- oxygen.
Definitions
Complete combustion
If all of the fuel is completely oxidized, we have complete combustion. For the principal species, the combustion products are \(\mathrm{CO_2}\), \(\mathrm{H_2O}\), and \(\mathrm{SO_2}\).
Partial or incomplete combustion
If not all of the fuel is completely oxidized, or if some is not oxidized at all, we have partial or incomplete combustion. A typical product of incomplete combustion is \(\mathrm{CO}\).
Stack or Flue gas
The combustion products along with any remaining reactants are referred to as stack gas or flue gas. The composition of the flue gas is reported in one of two bases.
Definitions (cont.)
Wet basis
Wet basis includes the water vapor in the flue gas composition.
Dry basis (Orsat analysis)
Dry basis does not include water vapor in the flue gas composition. One apparatus for measuring flue gas composition was named after Orsat. It reports compositions on a dry basis.
Relative, Absolute, Molal, and Percentage Humidity
We discuss humidity and how to calculate it in the Oh, Henry! video. See link in description or on web page.
A Quick Wet- and Dry-Basis Example
The table below shows a gas mixture converted from wet (or total) moles to wet composition, dry moles, and dry composition.
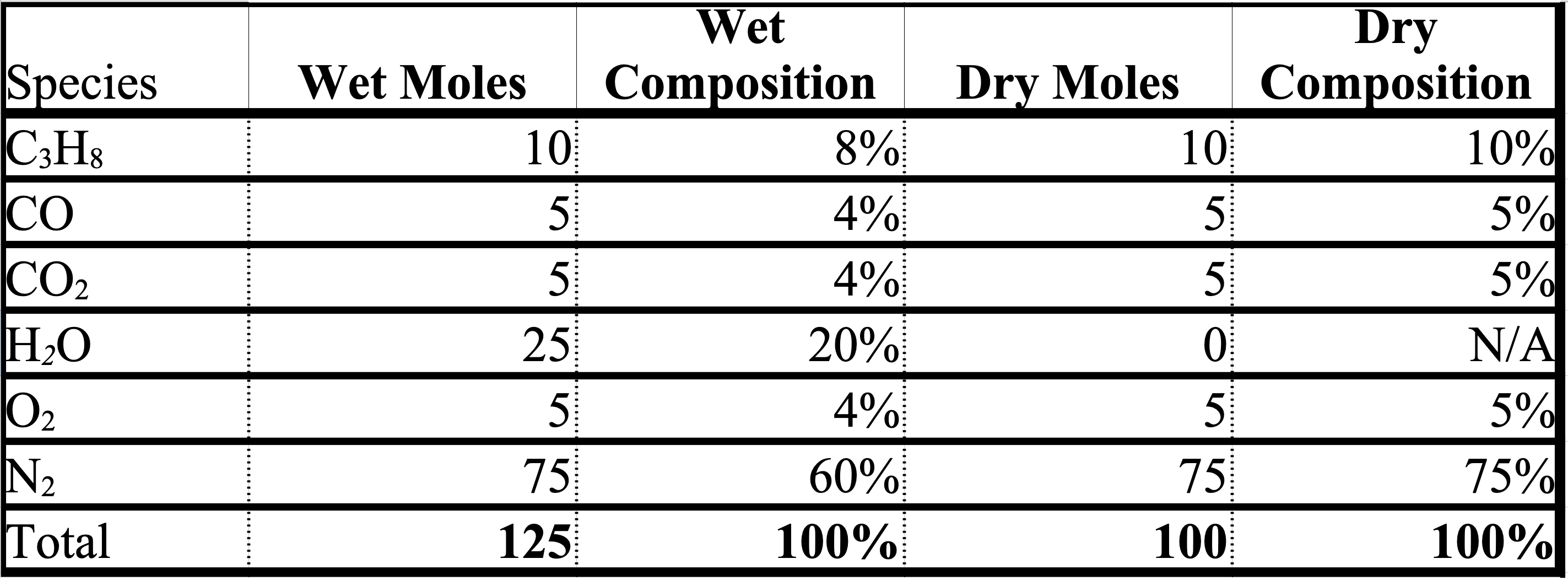
Definitions (cont.)
Air Composition
Dry air contains \(\mathrm{N_2}\), \(\mathrm{O_2}\), \(\mathrm{Ar}\), \(\mathrm{CO_2}\), \(\mathrm{H_2}\), \(\mathrm{He}\), \(\mathrm{Ne}\), \(\mathrm{Kr}\), \(\mathrm{Xe}\) and other trace gases. Search Wikipedia for Atmospheric Chemistry for a detailed list. For DOFPro calculations we will simplify this list to say that dry air is \(79\%\ \mathrm{N_2}\) and \(21\%\ \mathrm{O_2}\). Also, we’ll assume that the value of the \(\mathrm{N_2}\):\(\mathrm{O_2}\) ratio is \(79\):\(21\) or \(3.76\) to three sig figs.
Definitions (cont.)
Theoretical air
Theoretical air is the amount of air containing the stoichiometric oxygen needed for complete combustion.
Excess air
Excess air is calculated in the same manner as any excess reactant. It is the fraction above stoichiometric.
- Burning in excess air does not guarantee complete combustion.
- Burning with less than theoretical air is known as burning rich (as in fuel rich).
- Burning with excess air is known as burning lean.
The Takeaways
- The principal species for combustion reactions are carbon, hydrogen, sulfur, and oxygen.
- The products of complete combustion of the principal species are carbon dioxide, water, and sulfur dioxide.
- Theoretical and excess air are calculated by treating the oxygen in air as a stoichiometric or excess reagent.
- Excess air does not guarantee complete combustion.
- The composition of combustion gases are reported on either a wet or dry basis.
Thanks for watching!
The Full Story companion video is in the link in the upper left. The companion video in the series, A Combustible Mixture!, is in the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.

