REAL Gas Mixtures EXPLAINED
Don’t Let This Cost You Points
on Your Exam! Part 2
DOFPro Team

Kay’s Rule Review
- Calculate the pseudocritical temperature.
- Calculate the pseudocritical pressure.
- Calculate the pseudoreduced temperature.
- Calculate the pseudoreduced pressure.
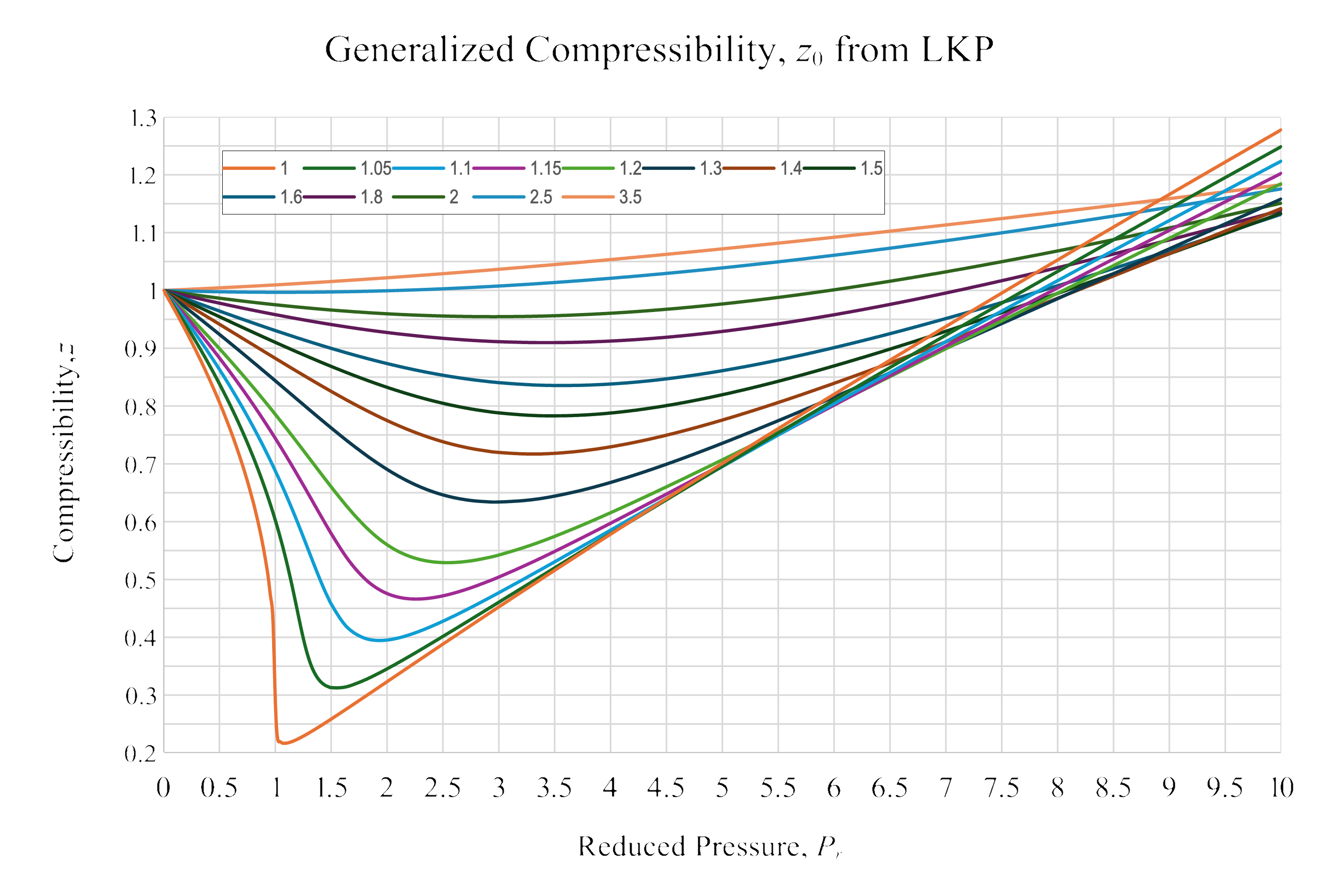
- Use the generalized compressibility charts to calculate \(z_m\).
- Use \(z_m\) to calculate anything else needed.
Kay’s Rule (cont.)
\[ P_r' =\frac{P}{P_c'} \]
\[ T_r' = \frac{T}{T_c'} \]
\[ z_m = z_m(T_r', P_r') \]
DWSIM and Equation of State
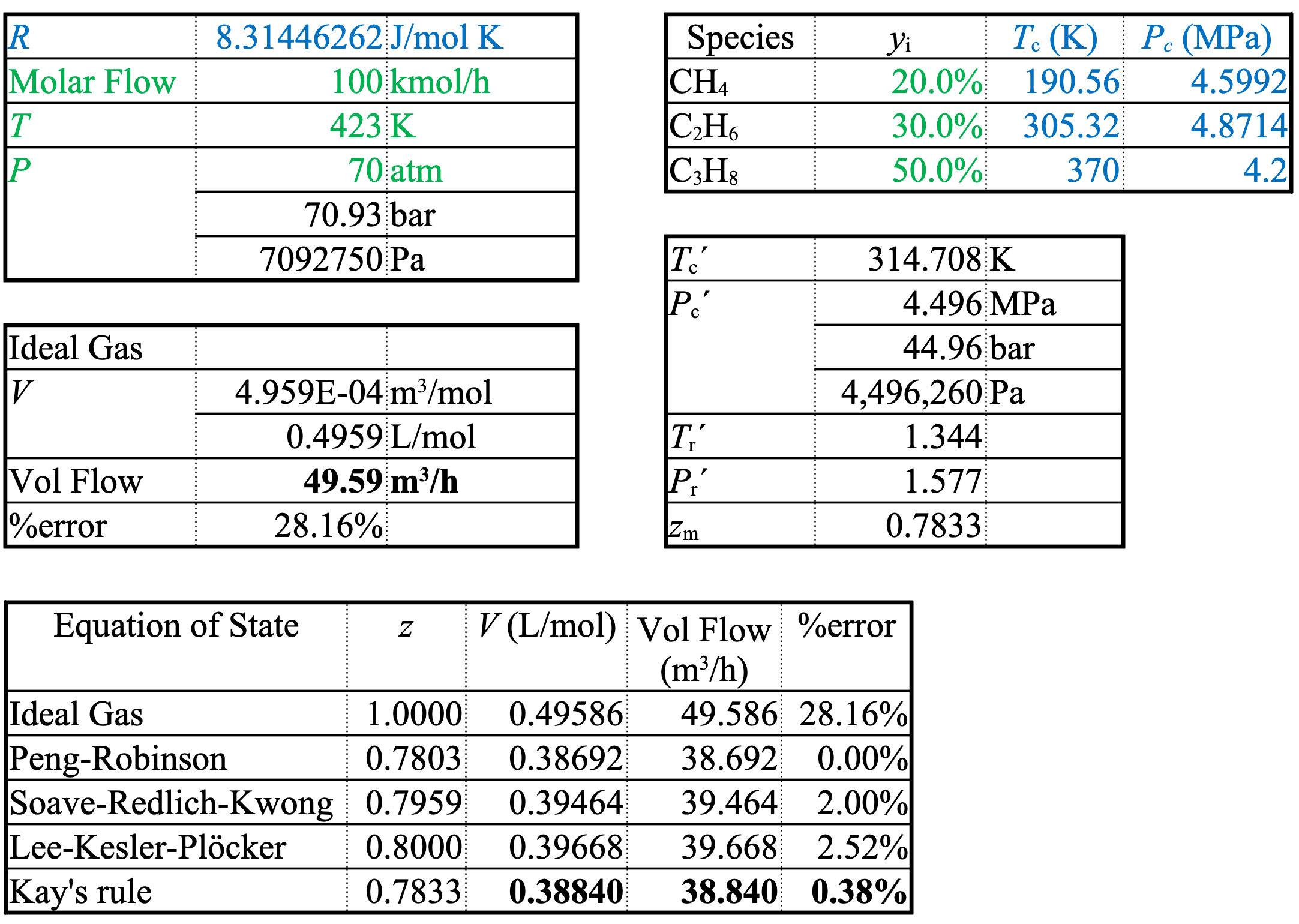
Real Gas Example
A stream of
20% methane,
30% ethane, and
50% propane
at \(T = 423\ \mathrm{K}\) and \(P = 70\ \mathrm{atm}\) flows at \(100.0\ \mathrm{kmol/h}\).
Estimate the volumetric flow rate using Kay’s rule and compare with the ideal gas law and the Peng-Robinson, Soave-Redlich-Kwong, and Lee-Kesler-Plöcker equations of state.
Example (cont.)
\[70\ \mathrm{atm} \boldsymbol{\cdot} 0.101325\ \mathrm{\frac{MPa}{atm}}= 7.093\ \mathrm{MPa} = 7,093,750\ \mathrm{Pa}\]
Ideal Gas
\[ \hat{V}=\frac{RT}{P}=\frac{8.314\mathrm{\frac{J}{mol \cdot K}}\boldsymbol{\cdot}423\ \mathrm{K}}{7.093\times10^{6}\ \mathrm{Pa}}=4.959\times10^{-4}\ \mathrm{\frac{m^{3}}{mol}}=0.4959\ \mathrm{\frac{L}{mol}} \]
\[ \dot{V}=\dot{n}\hat{V}=100\ \mathrm{\frac{kmol}{h}}\boldsymbol{\cdot}\frac{1000\ \mathrm{mol}}{1\ \mathrm{kmol}}\boldsymbol{\cdot}4.959\times10^{-4}\ \mathrm{\frac{m^{3}}{mol}}=49.59\ \mathrm{\frac{m^{3}}{h}} \]
Kay’s Rule Calculation
\[ T_c' = 0.200(4.23) + 0.300(305.32) + 0.500(370) = 314.71\ \mathrm{K} \]
\[ P_c' = 0.200(4.5992) + 0.300(4.8714) + 0.500(4.23) = 4.496\ \mathrm{MPa} \]
\[ T_r' = \frac{T}{T_c'} = \frac{423}{314.7} = 1.344 \]
\[ P_r' = \frac{P}{P_c'} = \frac{7.093}{4.496} = 1.577 \]
Example (cont.)
Using Kay’s rule

\[ P_r' = \frac{P}{P_c'} = \frac{7.093}{4.496} = 1.577 \]
\[ T_r' = \frac{T}{T_c'} = \frac{423}{314.7} = 1.344 \]
\[ z_{m}=0.7833 \]
\[ \dot{V}_{KR}=z_{m}\dot{V}_{IG}=38.84\ \mathrm{\frac{m^{3}}{h}} \]
DWSIM Video
Example Spreadsheet
The Takeaways
- Kay’s rule is the simplest method for calculating properties of mixtures of real gases by hand, and it is very good for aliphatic hydrocarbons, but it is still a fair amount of work
- DWSIM or some other software application is the easiest way to calculate properties of mixtures of real gases using equations of state.
- Life gets much more interesting than the ideal gas law when you have mixtures of real gases.
Thanks for watching!
The previous in the series video is the link in the upper left. The next video in the series is the link the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.