
Can You Tell an Azeotrope from a Peritectic? Get Your Phase Diagrams Right! Part 1
DOFPro Team
Introduction
Can You Tell an Azeotrope from a Peritectic?
Get Your Phase Diagrams Right! Part 1 and Part 2
- Introduction to
- Phase separation
- Phase equilibria
- i.e., where split fractions come from
Moonshiners

National Photo Company Collection, Public domain, via Wikimedia Commons
Oil Producers

User:Luigi Chiesa, CC BY 3.0, via Wikimedia Commons
Makers of High-Strength Alloys

Djhé, CC BY 4.0, via Wikimedia Commons
Gibbs Phase Rule
\[\boxed{DF = 2 + c - \Pi}\]
A Degree of Freedom, \(DF\), is a coordinate or variable.
A Component, \(c\), is a chemical species, e.g. \(\mathrm{Al_2O_3}\).
A Phase, \(\Pi\), is a physical entity with a uniform composition, temperature, pressure and specific volume. In a given container, you can have multiple solid phases and multiple liquid phases, but only one vapor or gas phase.
Cooking Oil and Vinegar
Phase Diagrams for Single Components
How do you calculate phase equilibria?
What does phase equilibrium mean?
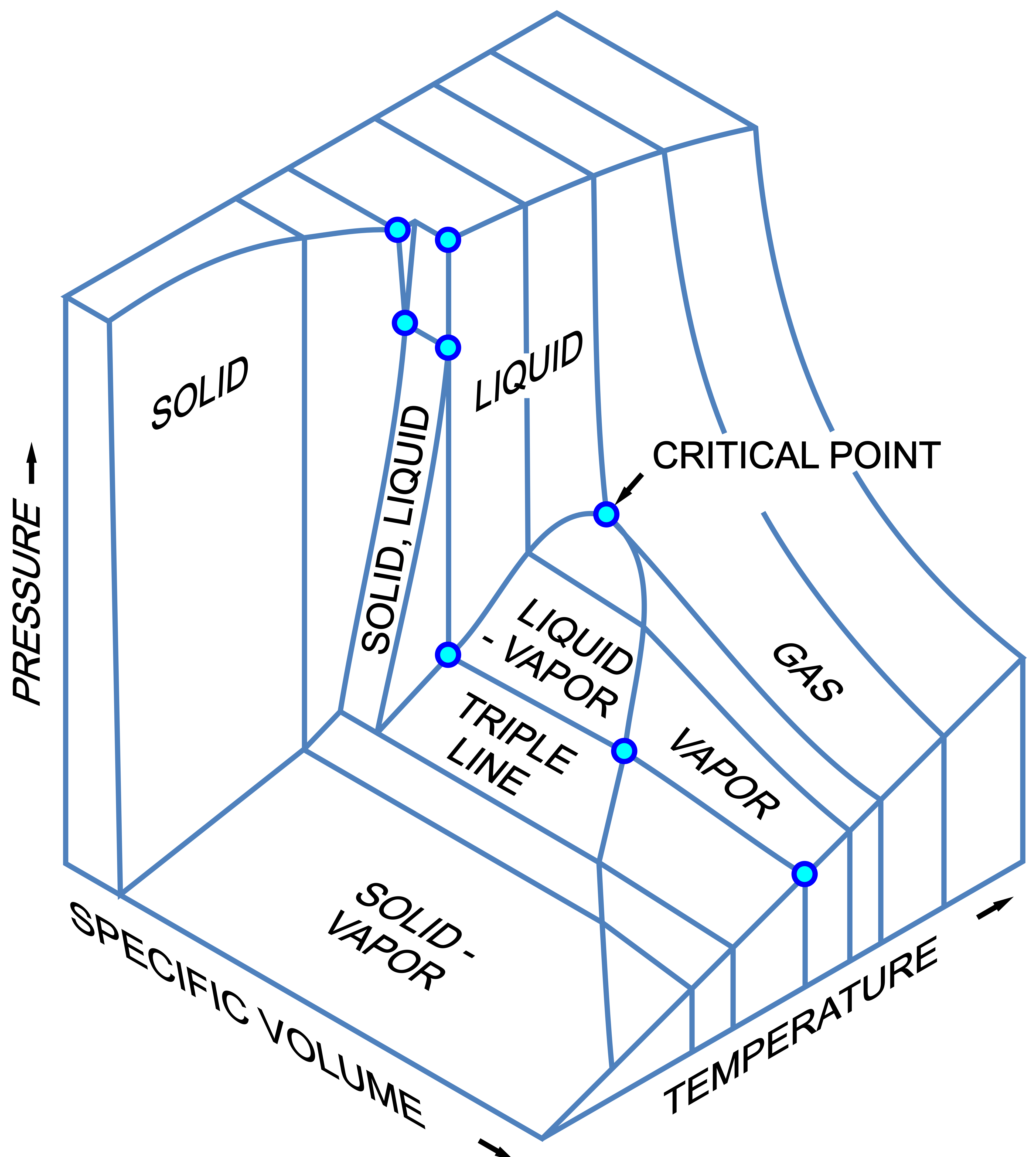
Д.Ильин: vectorization,
CC0, via Wikimedia Commons
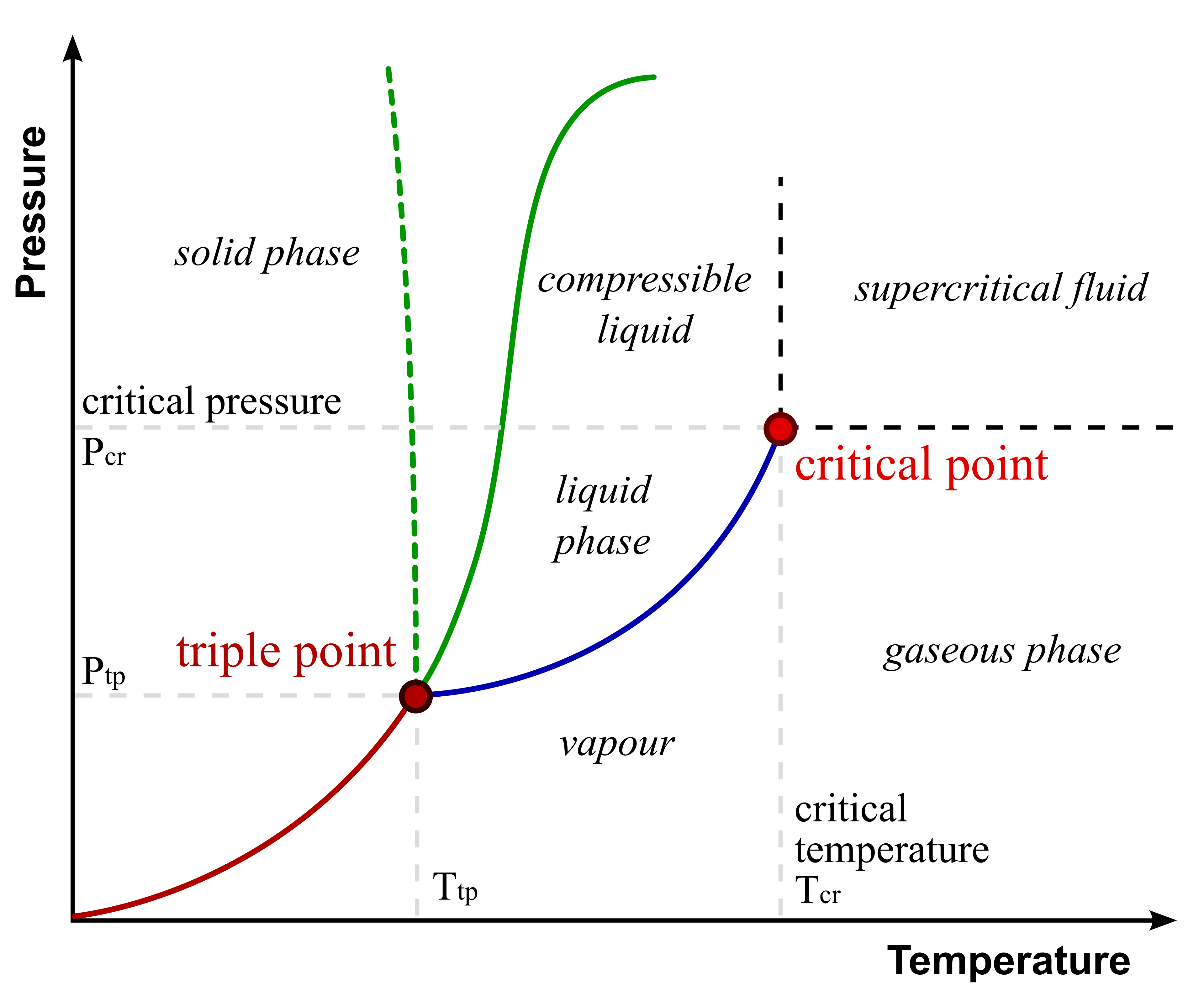
Matthieumarechal, CC BY-SA 3.0,
via Wikimedia Commons
\[DF = 2 + c - \Pi = 3 - \Pi\]
\(1\text{-}\phi\)
\(\ \ \bullet\)
\(2\text{-}\phi\)
\(\ \ \bullet\)
\(1\text{-}\phi\)
\(\ \ \bullet\)
\(2\text{-}\phi\)
\(\ \ \bullet\)
\(1\text{-}\phi\)
\(\ \ \bullet\)
Water/Steam/Ice Phase Diagram
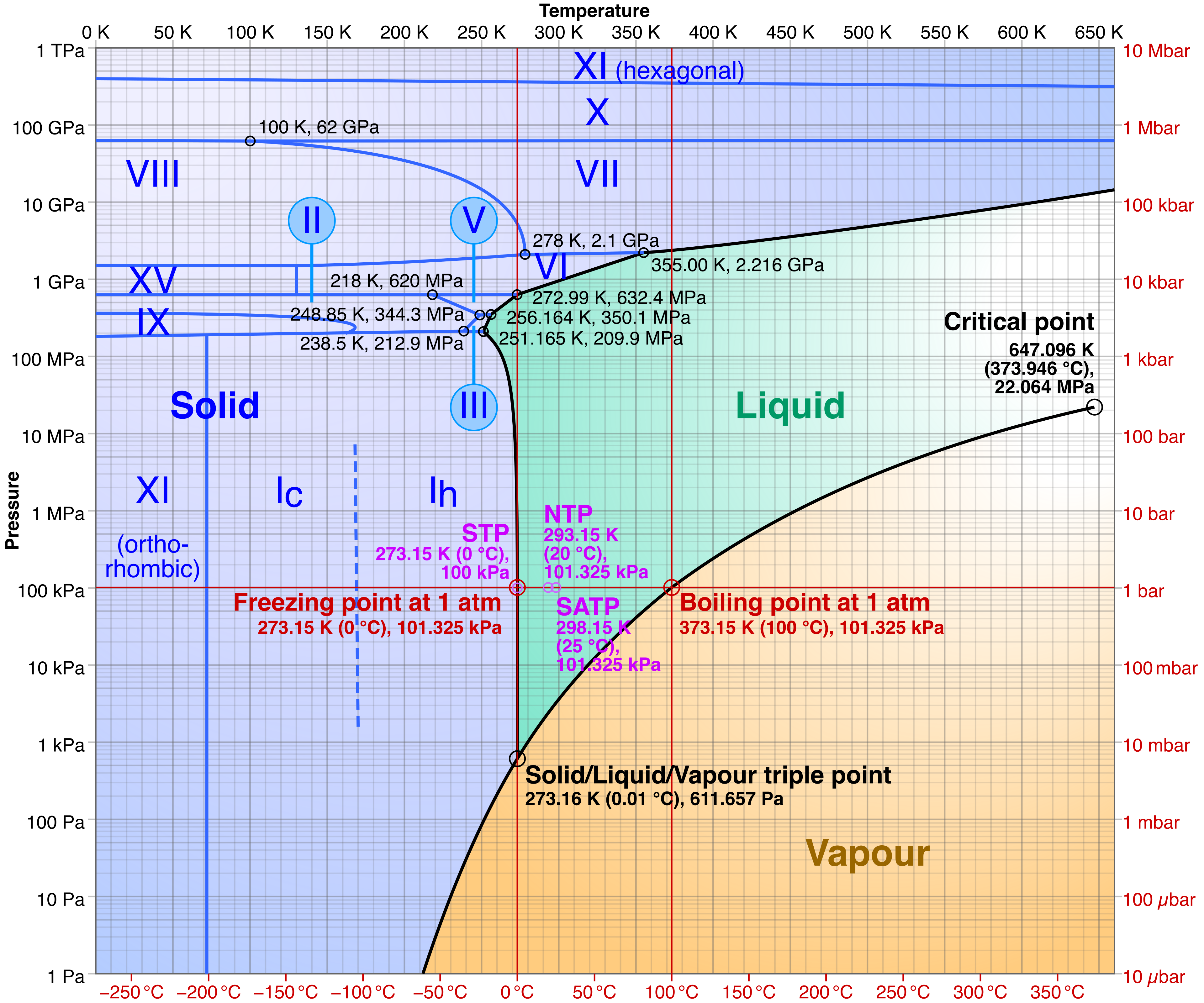
Cmglee, CC BY-SA 3.0,
via Wikimedia Commons
Al-Ni Phase Diagram
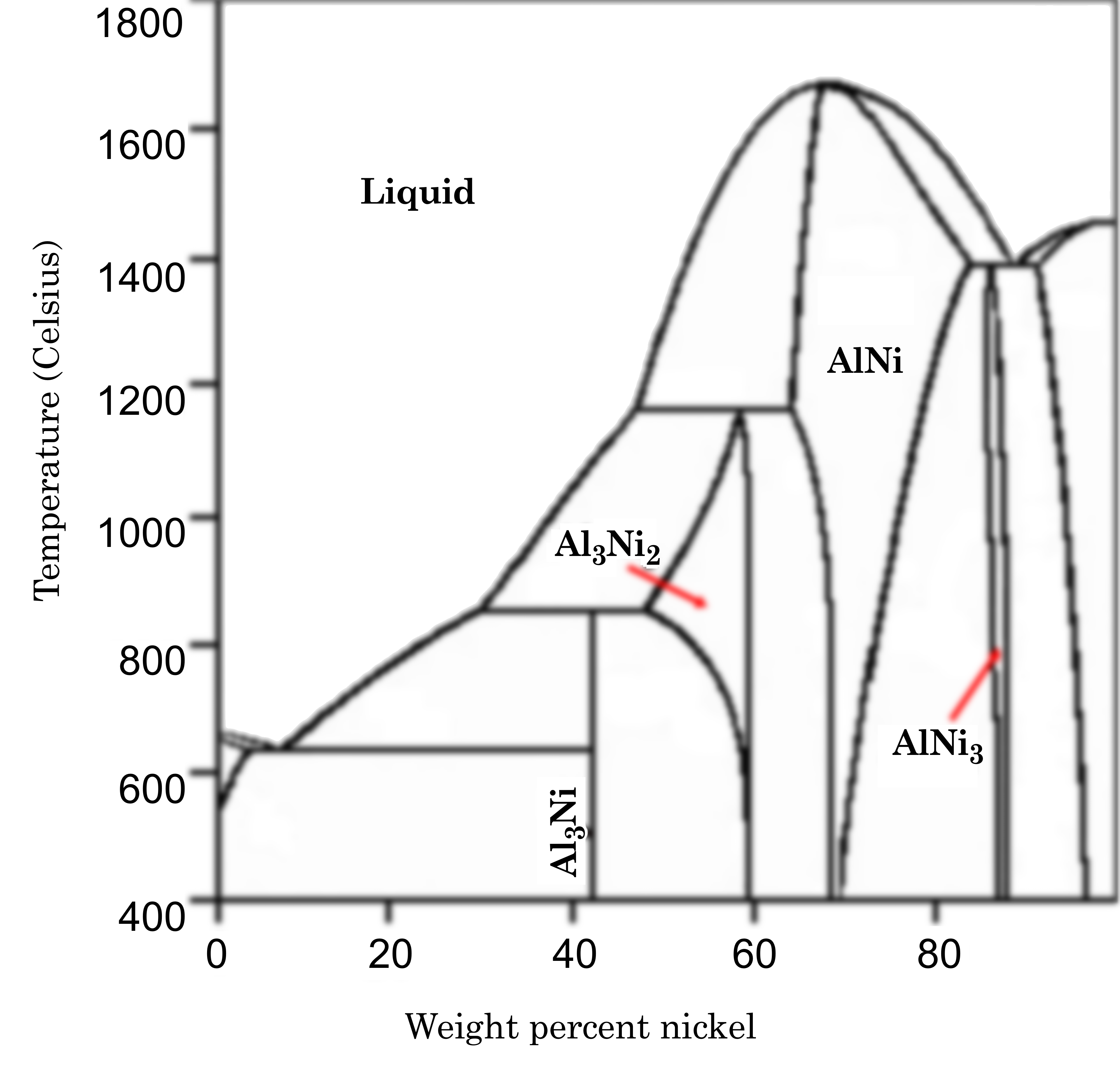
User Rune.welsh on en.wikipedia, CC BY-SA 3.0, via Wikimedia Commons
\(DF = 2(-1,\ P=\mathrm{const.})+c - \Pi = 3 - \Pi\)
\(1\text{-}\phi\)
\(\ \ \bullet\)
\(2\text{-}\phi\)
\(\ \ \bullet\)
\(\ \ \bullet\)
\(1\text{-}\phi\)
\(2\text{-}\phi\)
\(\ \ \bullet\)
\(1\text{-}\phi\)
\(\ \ \bullet\)
\(3\text{-}\phi\)
\(\ \ \bullet\)
\(\bullet\)
\(\bullet\)
Fe-O Phase Diagram
 Joanna Kośmider, Public domain, via Wikimedia Commons
Joanna Kośmider, Public domain, via Wikimedia Commons
\(DF = 3 - \Pi\)
\(1\text{-}\phi\)
\(\ \ \bullet\)
\(2\text{-}\phi\)
\(\ \ \bullet\)
\(1\text{-}\phi\)
\(\ \ \bullet\)
\(2\text{-}\phi\)
\(\ \ \bullet\)
\(1\text{-}\phi\)
\(\ \ \bullet\)
\(\ \ \bullet\)
\(3\text{-}\phi\)
\(\bullet\)
\(\bullet\)
MgSO4–H2O Phase Diagram
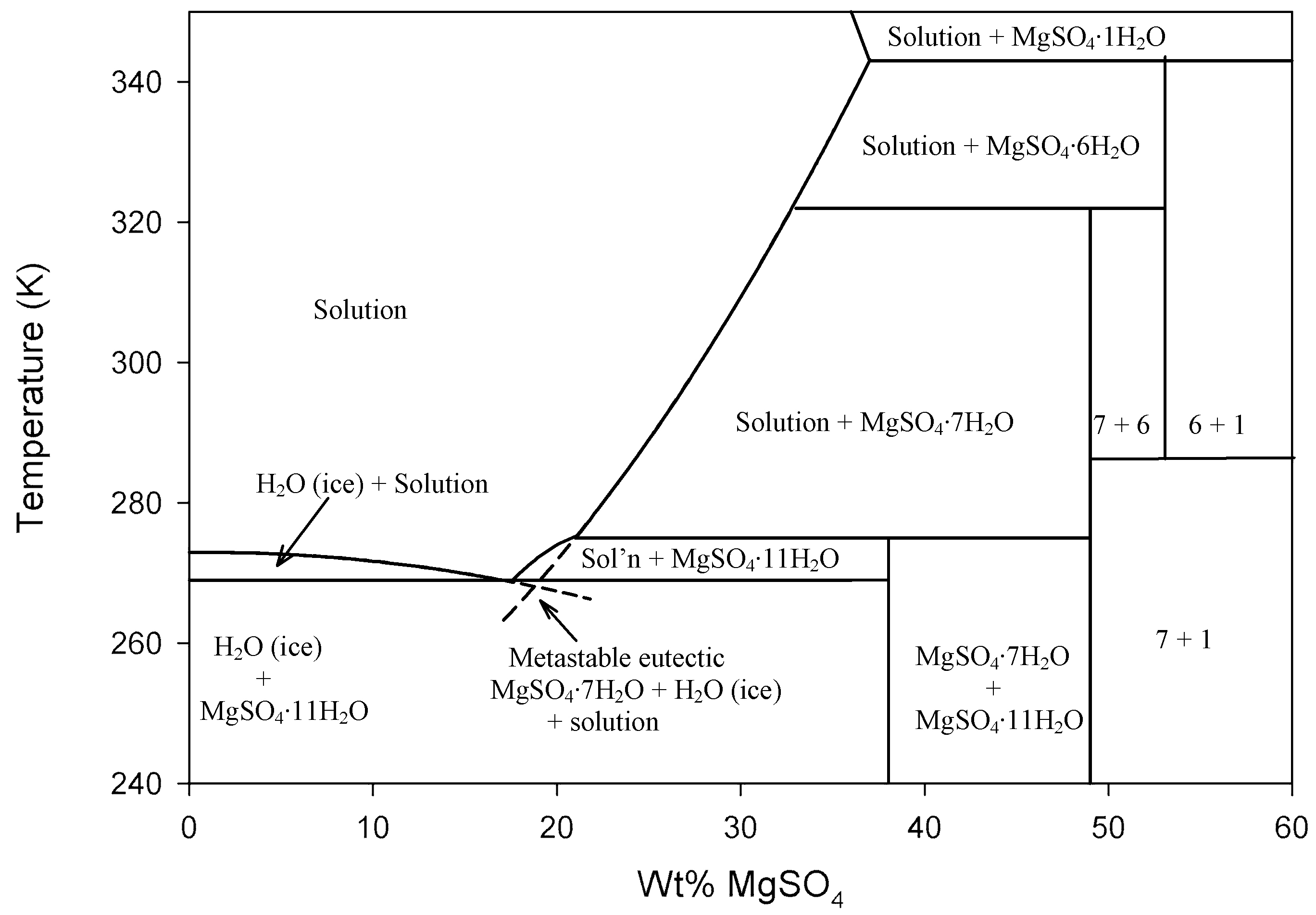
Ronald C. Peterson and Ruiyao Wang, Attribution, via Wikimedia Commons
\(1\text{-}\phi\)
\(\ \ \bullet\)
\(2\text{-}\phi\)
\(\ \ \bullet\)
\(1\text{-}\phi\)
\(\ \ \bullet\)
\(\ \ \bullet\)
\(2\text{-}\phi\)
\(1\text{-}\phi\)
\(\ \ \bullet\)
The Takeaways
- The Gibbs phase rule can be used to determine if an area on a phase diagram is a one-, two-, or three-phase region.
- The number of phases increments or decrements by one when you cross into a new region on a binary phase diagram
- The three-phase regions on a binary phase diagram are the eutectic, eutectoid, peritectic, peritectoid, and monotectic
Thanks for watching!
The previous video in the series is in the link in the upper left. The next video in the series is in the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.
The DOFPro Team

