
Can You Tell an Azeotrope from a Peritectic? Get Your Phase Diagrams Right! Part 2
DOFPro Team
Introduction
Can You Tell an Azeotrope from a Peritectic?
Get Your Phase Diagrams Right! Part 1 and Part 2
- Introduction to
- Phase separation
- Phase equilibria
- i.e., where split fractions come from
- Part 1
- Single-component and binary phase diagrams
- Part 2
- Ternary phase diagrams
- The Lever Rule.
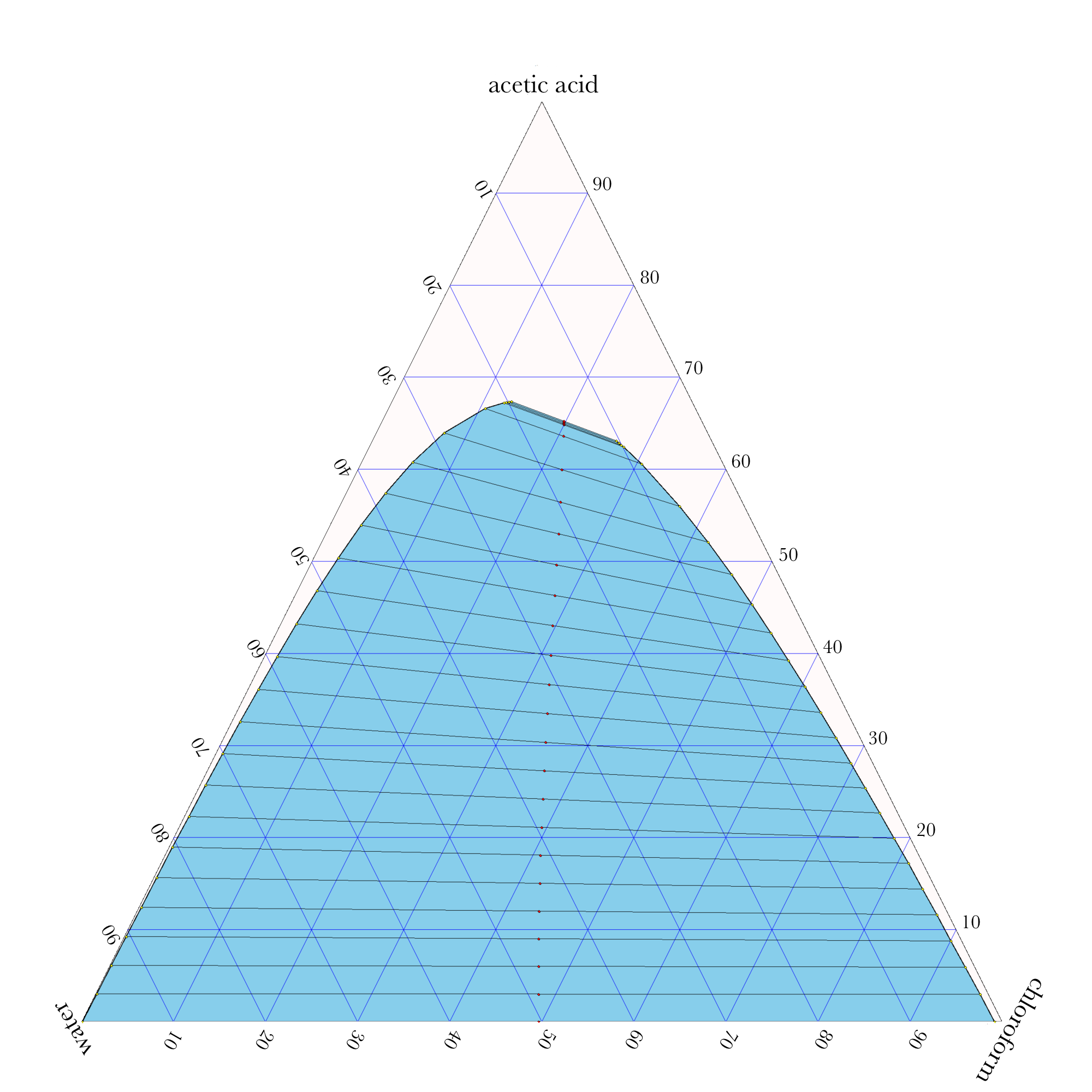
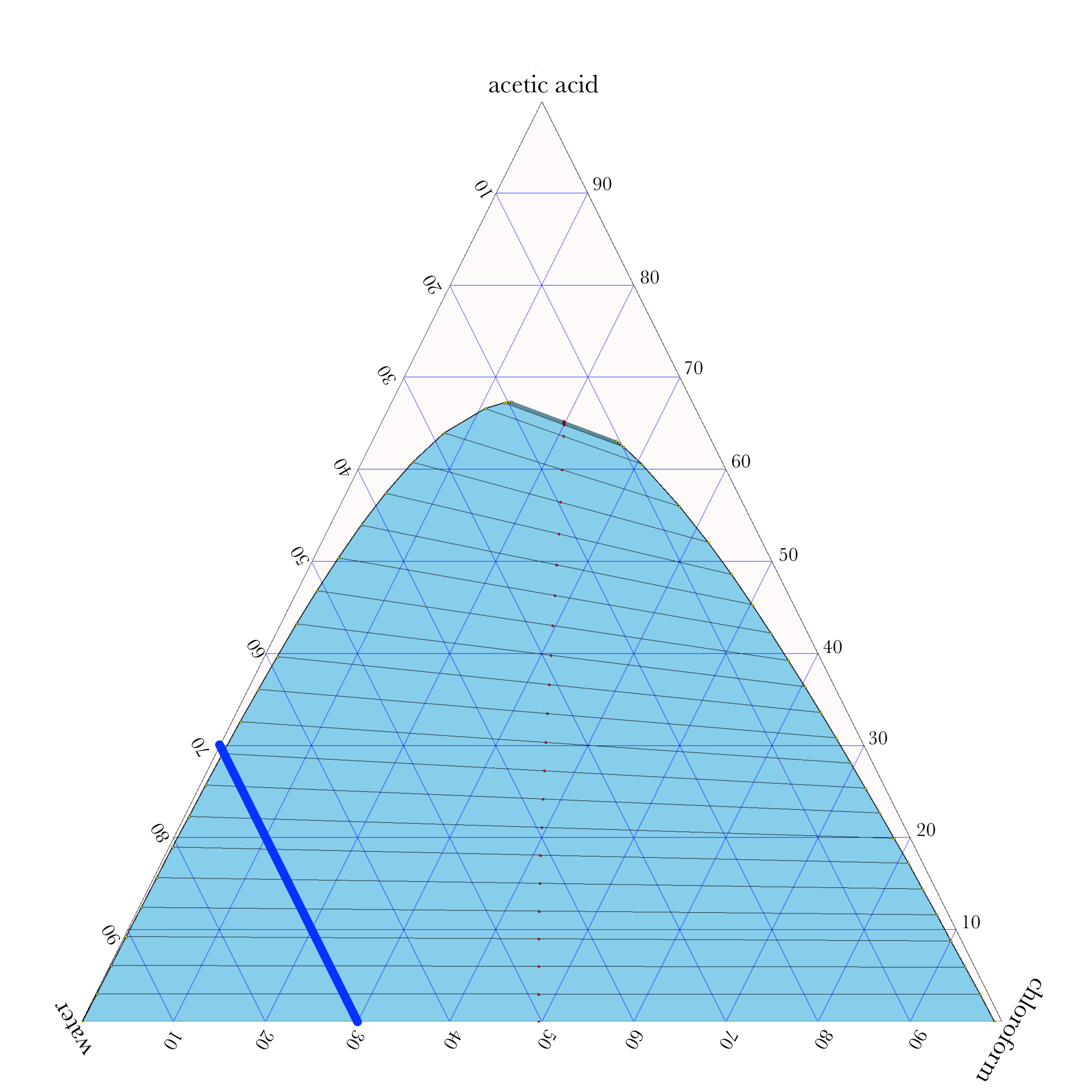
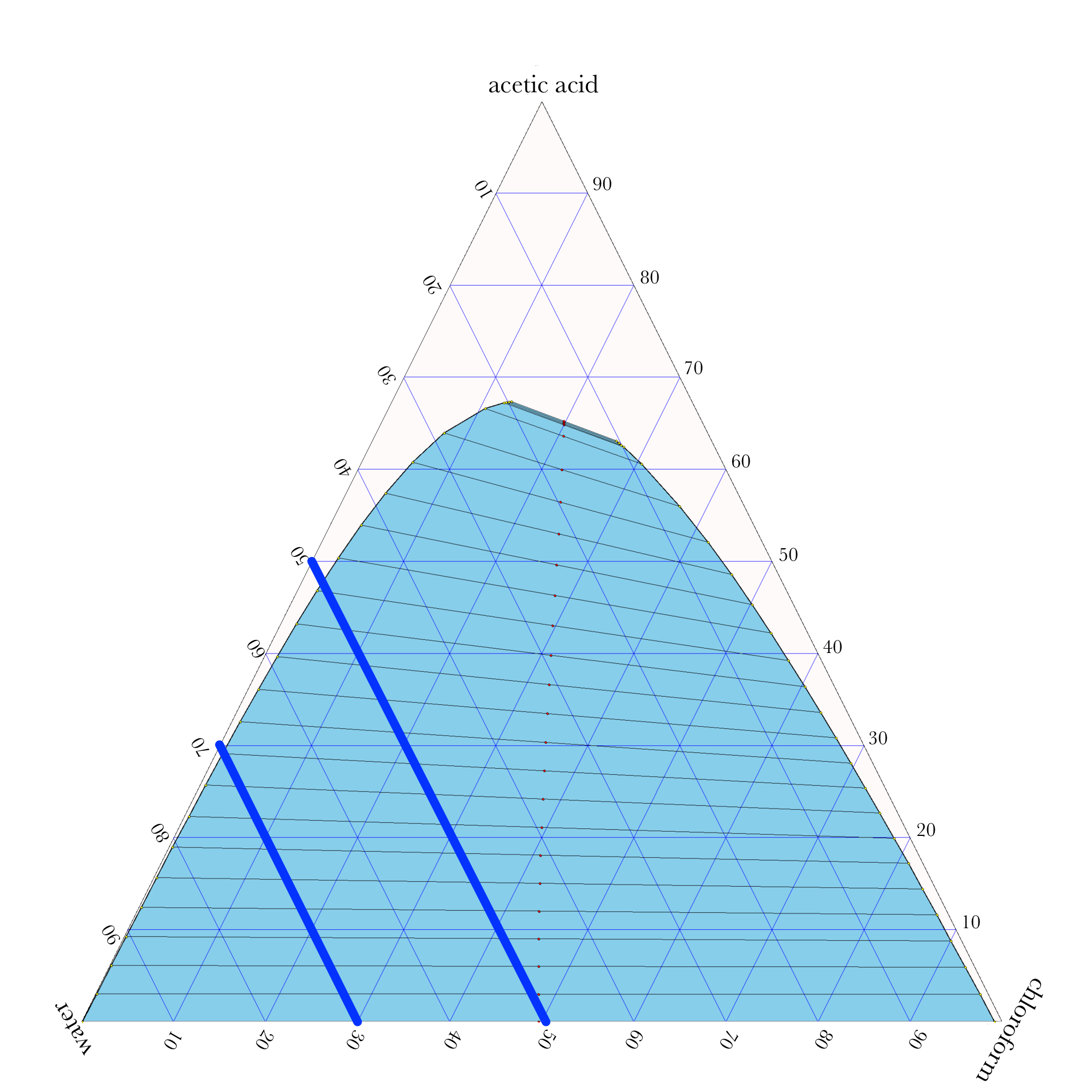
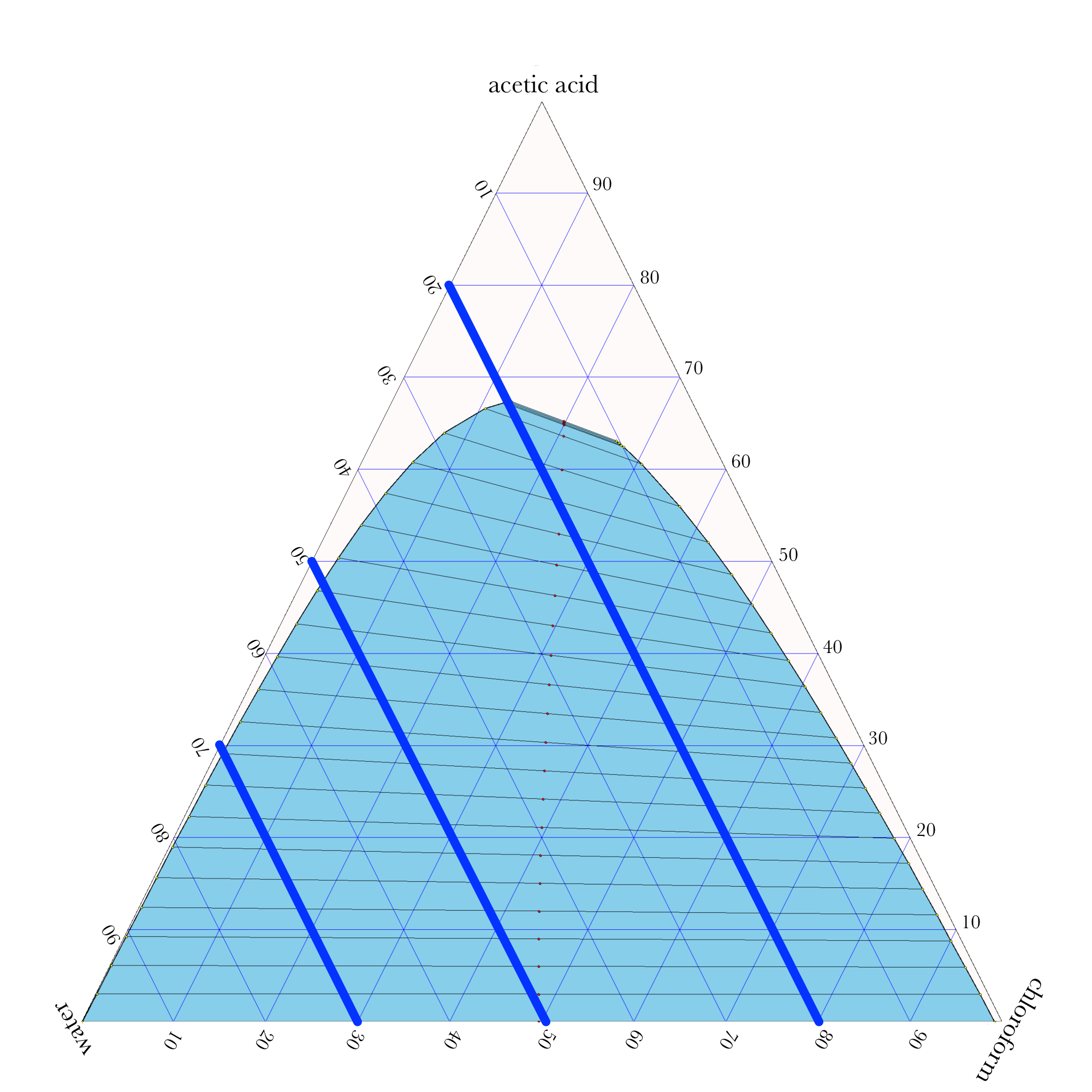
Liquid-Liquid Extraction
Liquid-Liquid Extraction
Liquid-Liquid Extraction
Liquid-Liquid Extraction
Liquid-Liquid Extraction
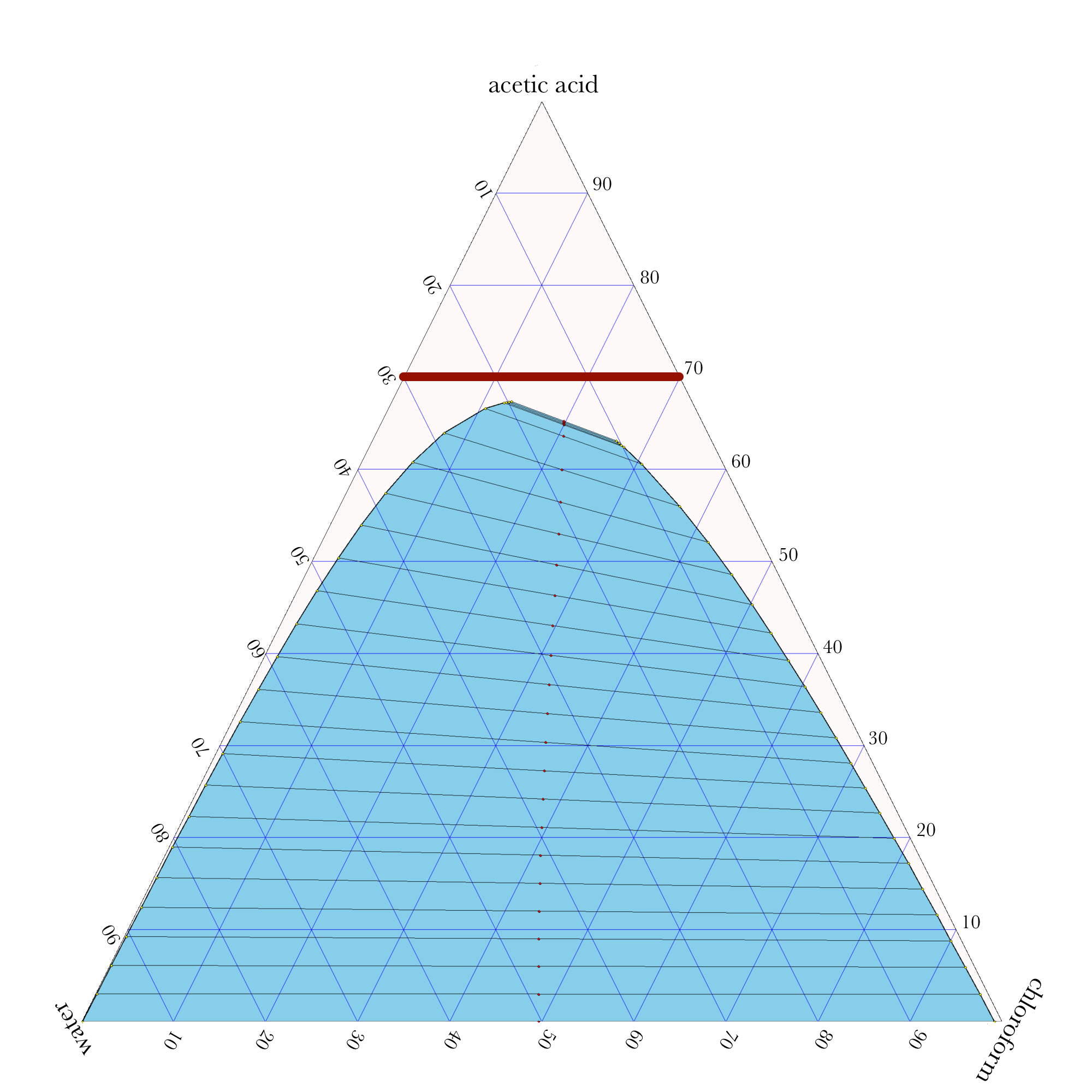
Liquid-Liquid Extraction
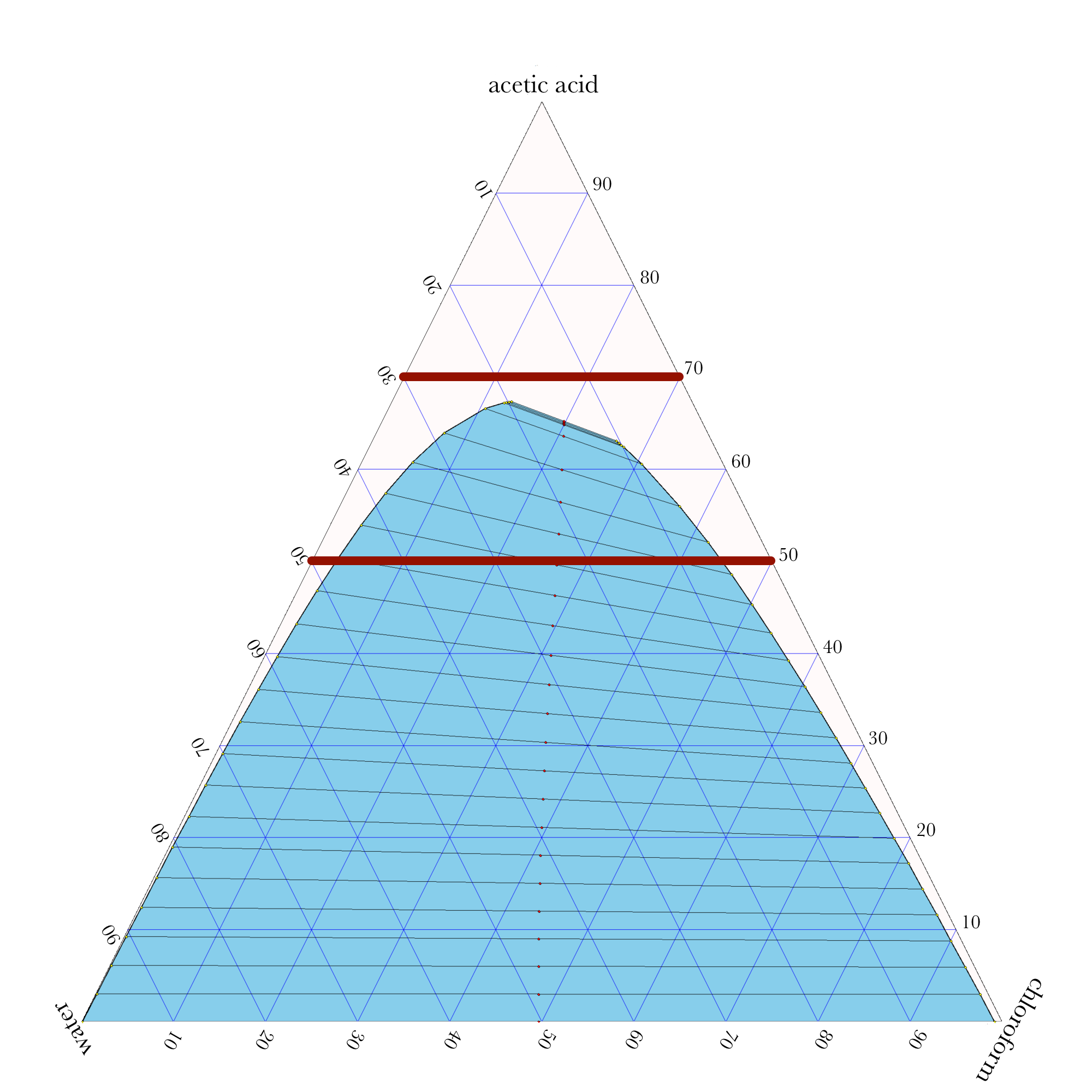
Liquid-Liquid Extraction
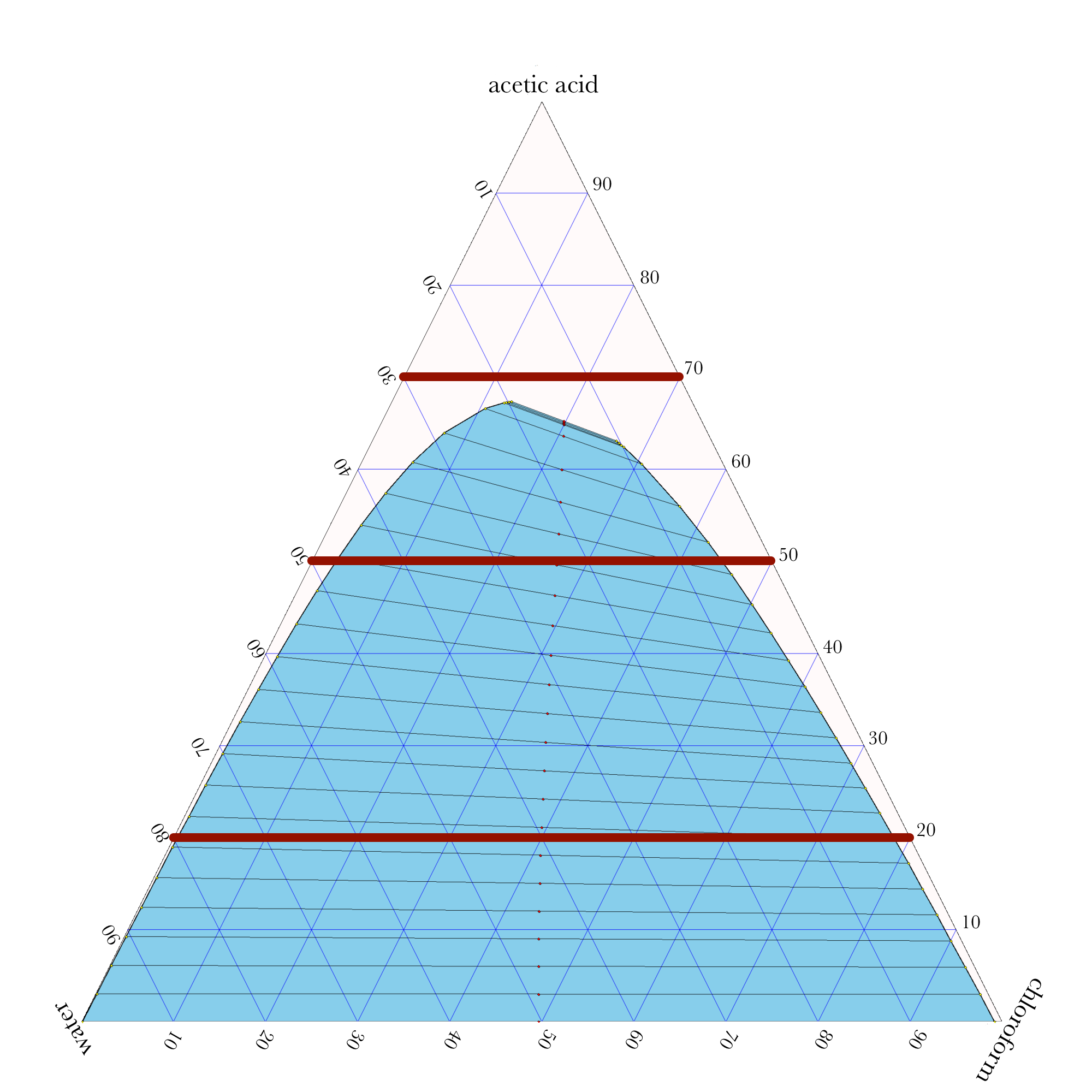
Liquid-Liquid Extraction
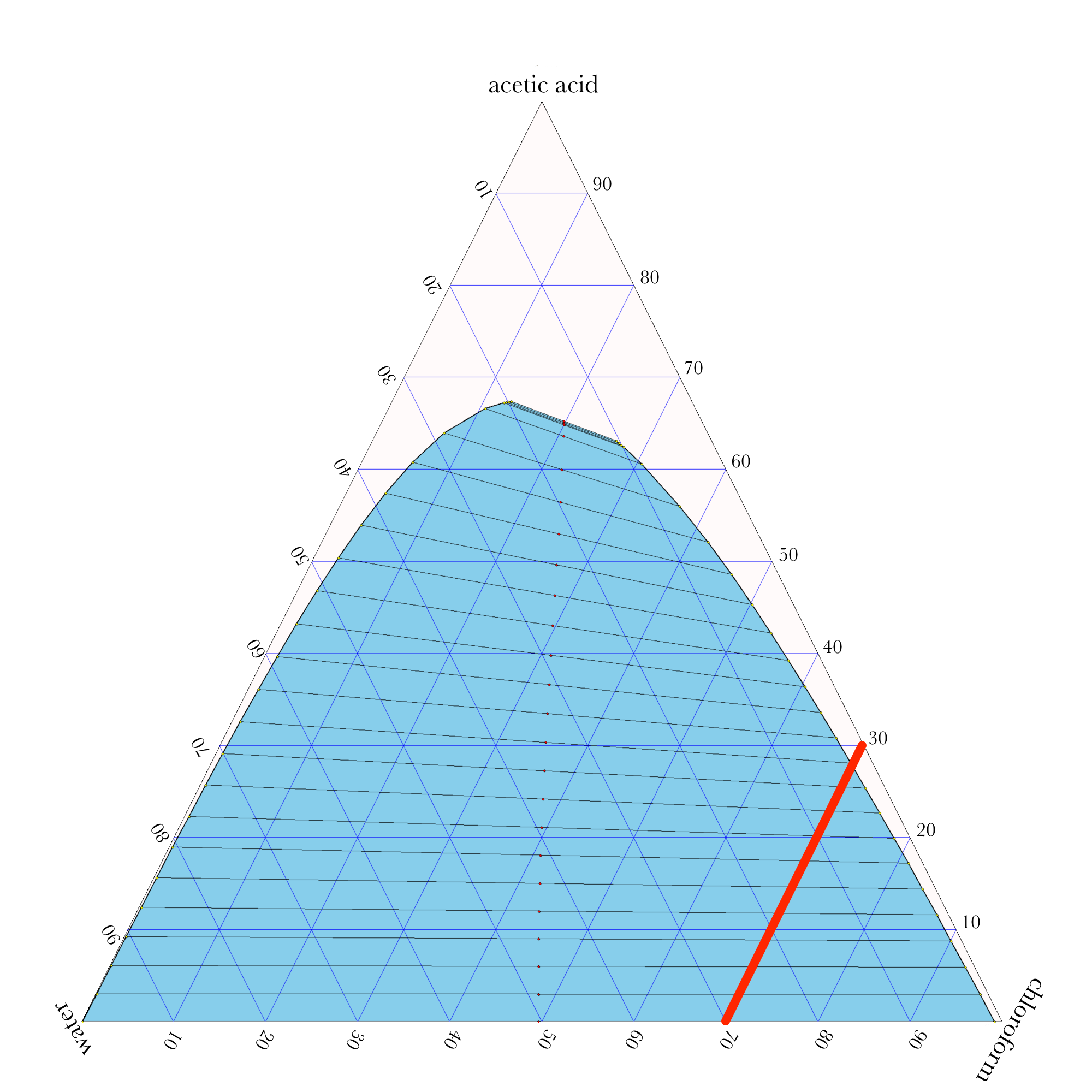
Liquid-Liquid Extraction
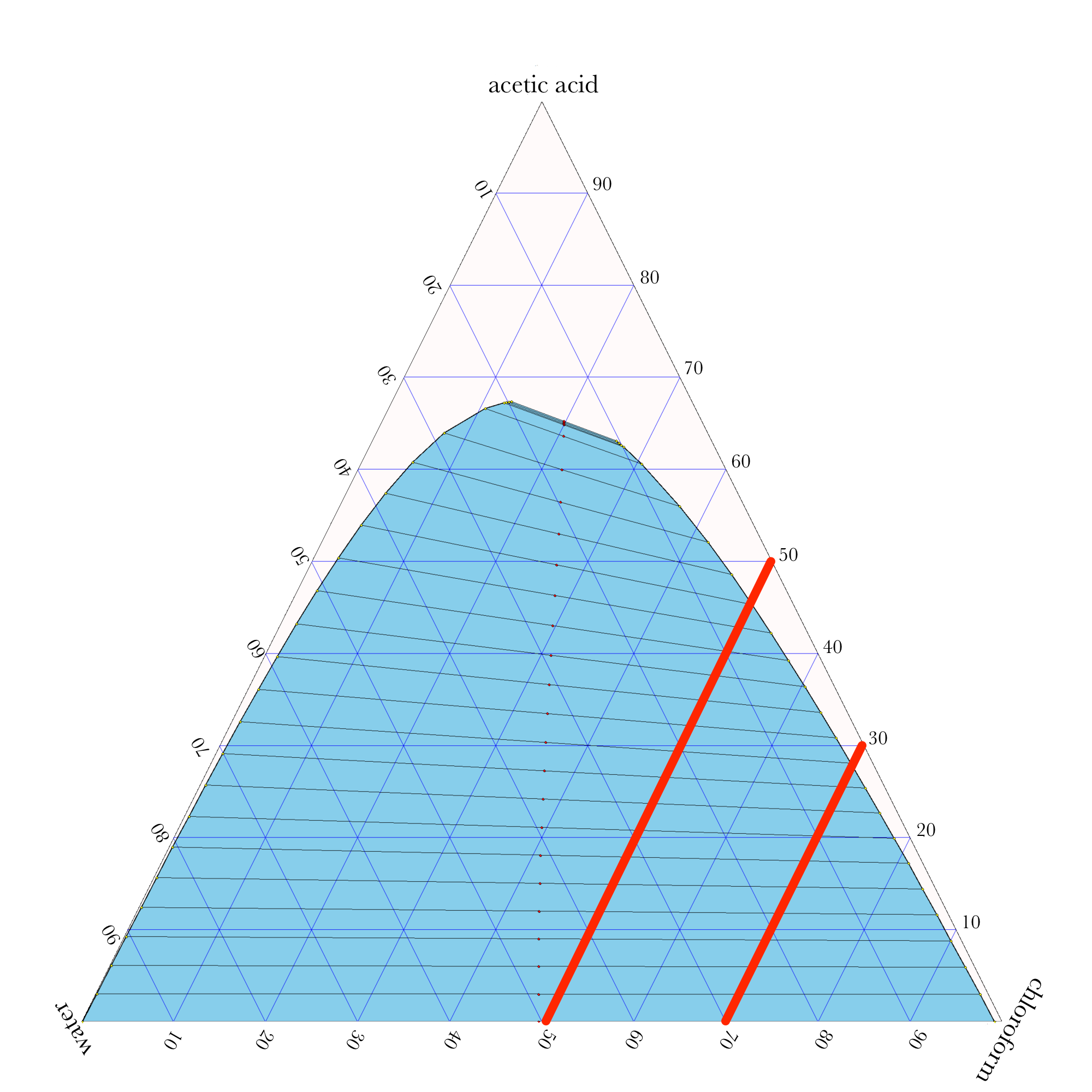
Liquid-Liquid Extraction
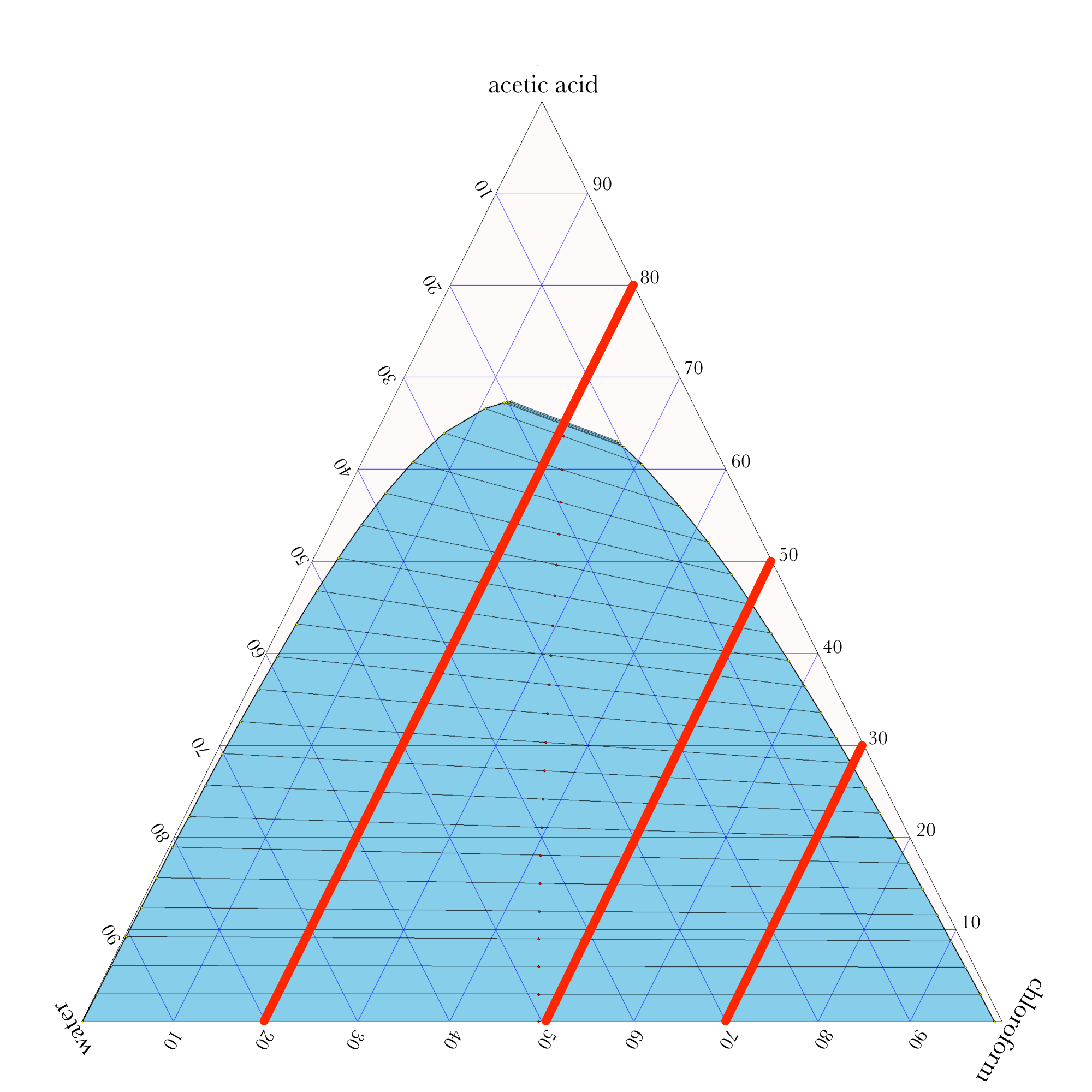
Liquid-Liquid Extraction

Liquid-Liquid Extraction
Liquid-Liquid Extraction
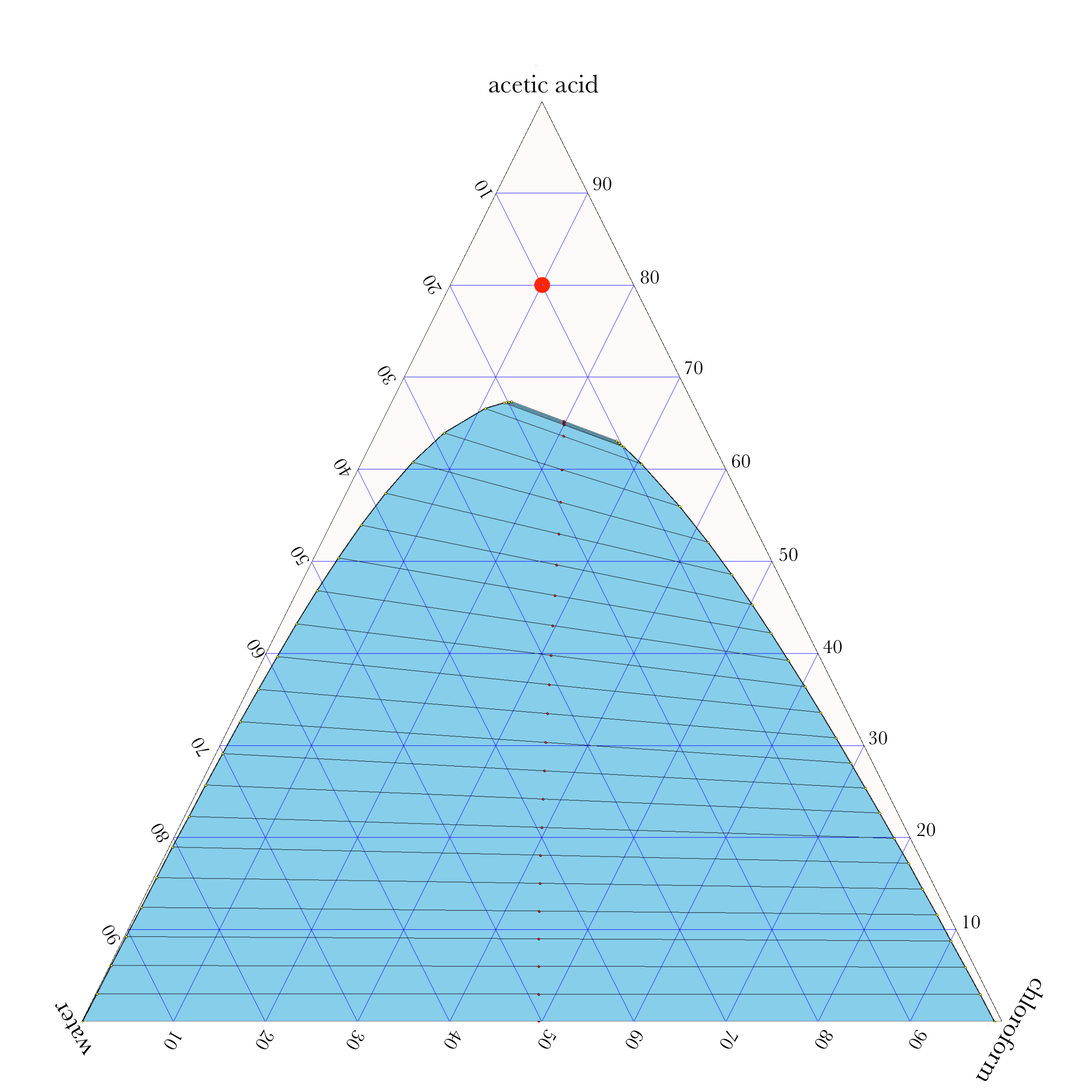
Liquid-Liquid Extraction
Liquid-Liquid Extraction
Liquid-Liquid Extraction
\(\phi_1\)
Liquid-Liquid Extraction
\(\phi_1\)
\(\phi_2\)
Txy and Pxy Binary Phase Diagrams
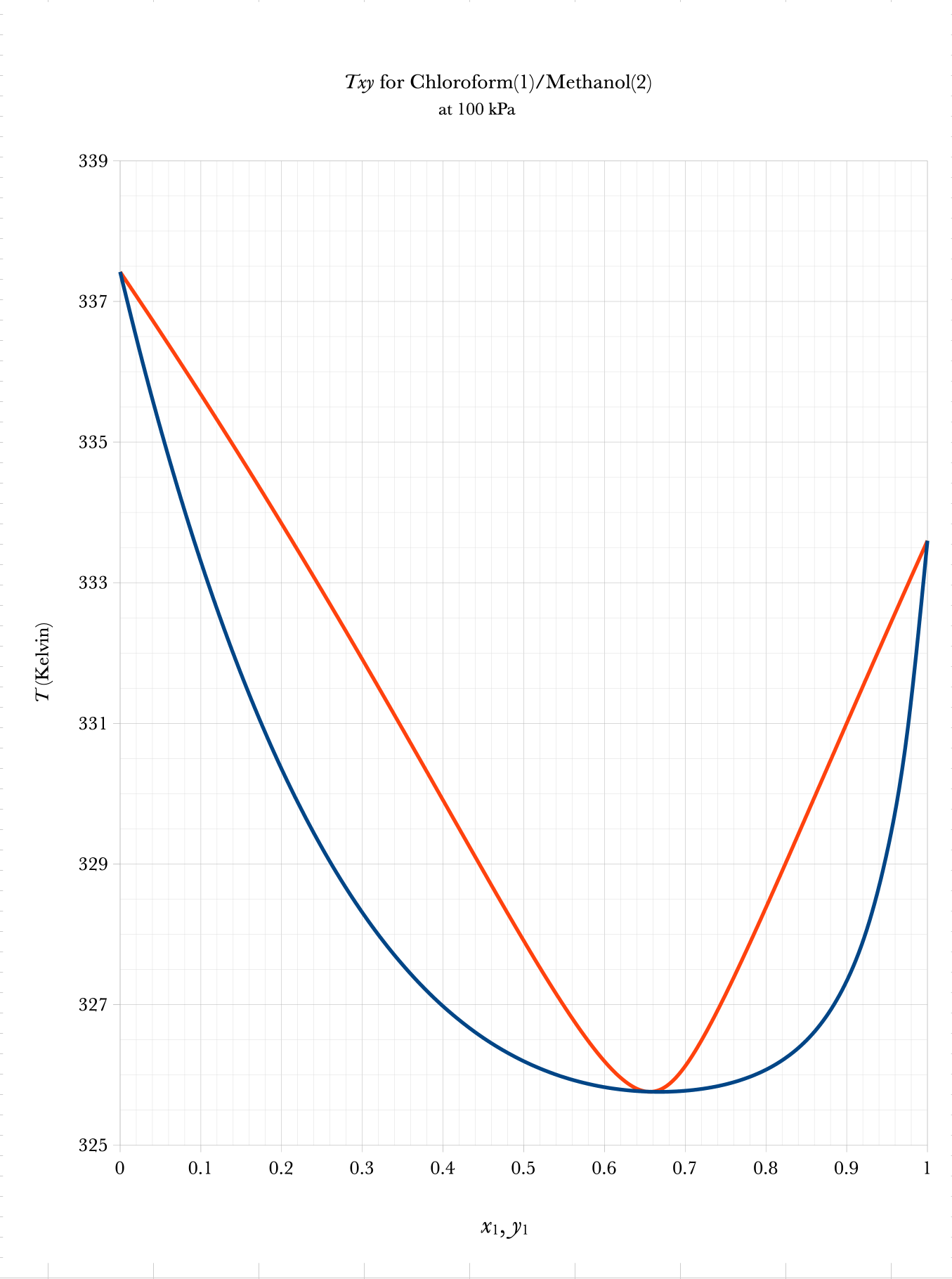
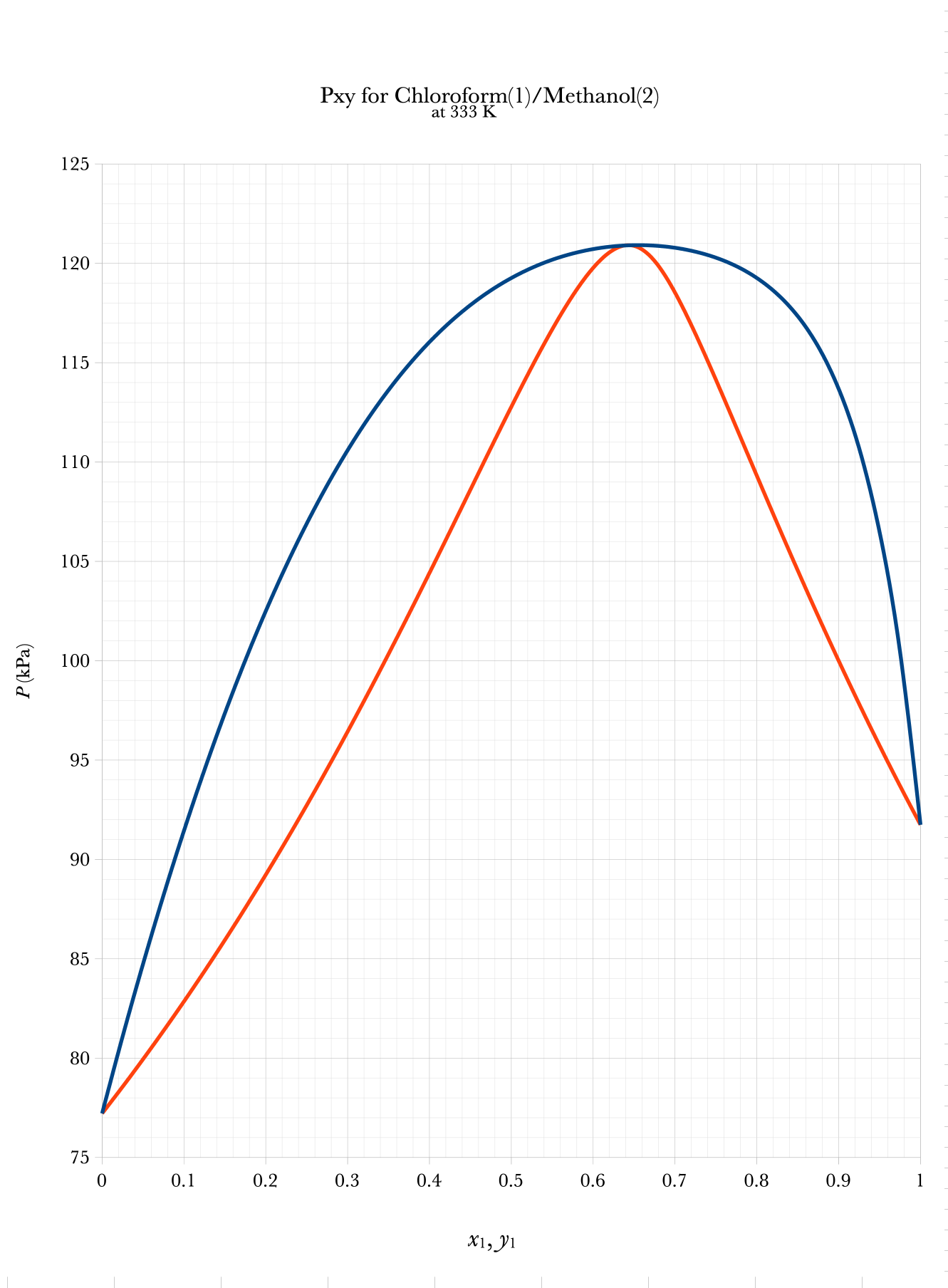
\(\mathrm{liquid}\)
\(\mathrm{liquid}\)
\(\mathrm{vapor}\)
\(\mathrm{vapor}\)
\(2\text{–}\phi\)
\(2\text{–}\phi\)
Lever Rule
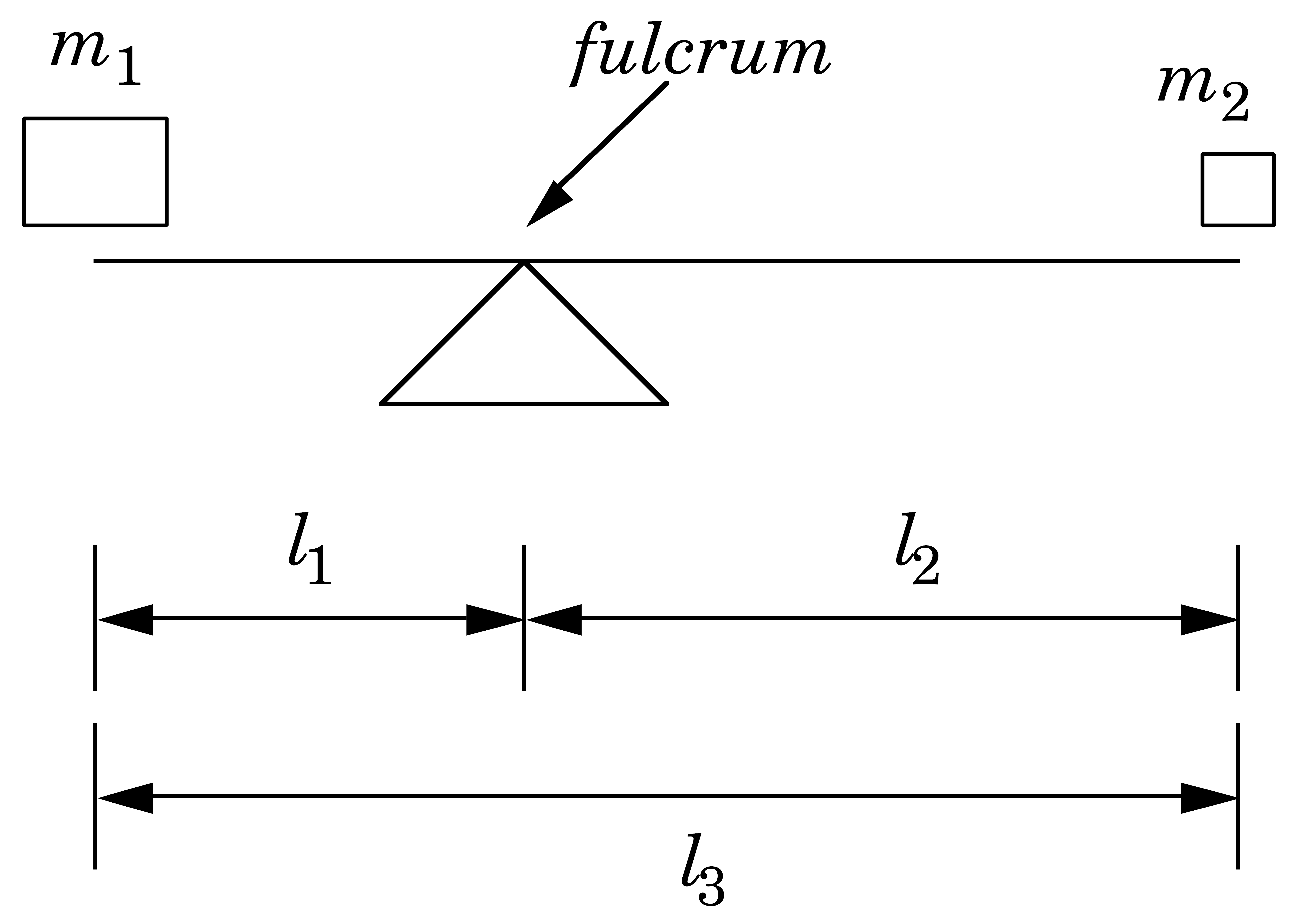
The Lever Rule for Phase Diagrams

\(\text{Phase }\alpha\)
\(\text{Phase }\beta\)
\(2\text{–}\phi\)
\(x_\mathrm{O}\)
The Lever Rule for Phase Diagrams
\(\text{Phase }\alpha\)
\(\text{Phase }\beta\)
\(2\text{–}\phi\)
\(x_\mathrm{O}\)
The Lever Rule for Phase Diagrams
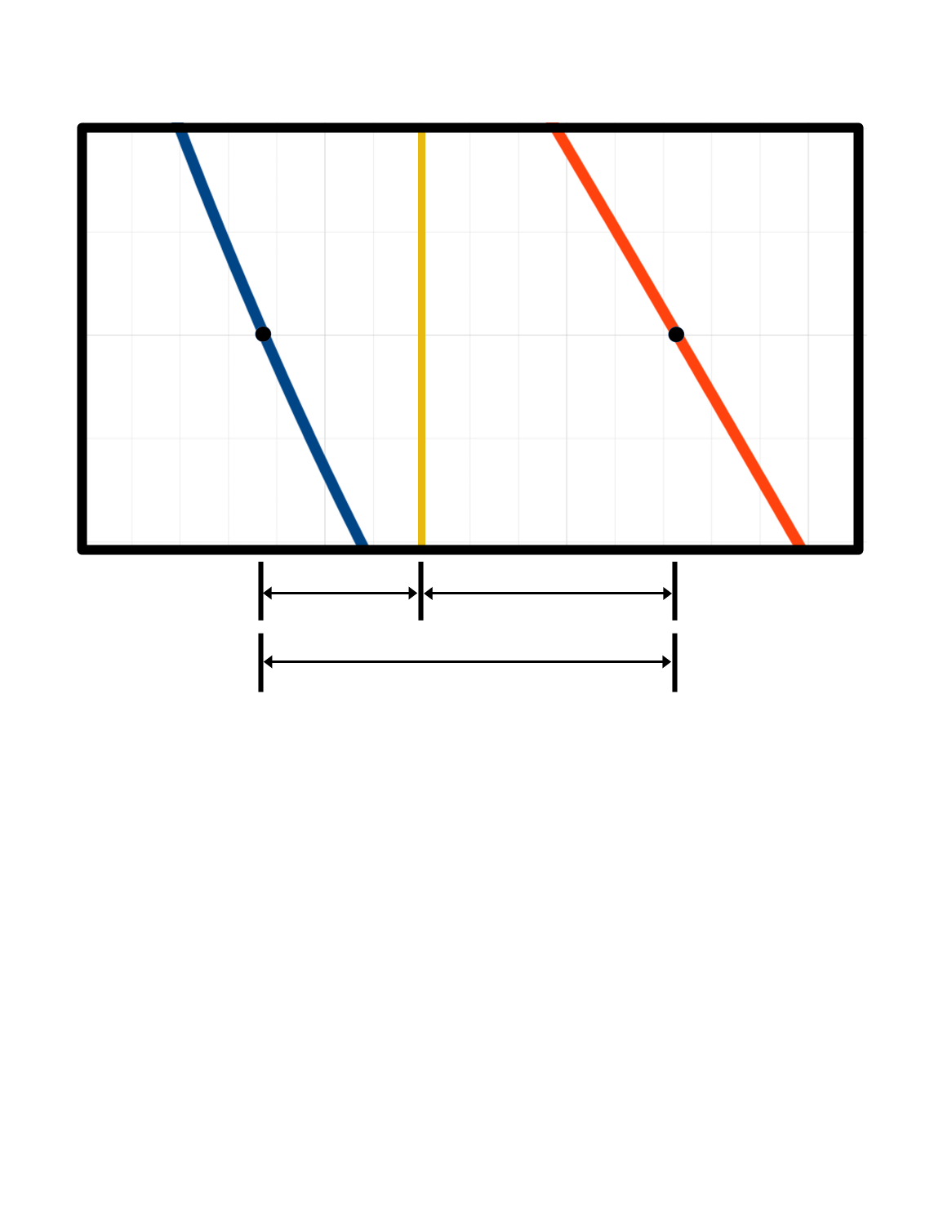
\(\text{Phase }\alpha\)
\(\text{Phase }\beta\)
\(2\text{–}\phi\)
\(x_\mathrm{O}\)
\(x_\alpha\)
\(x_\beta\)
\(l_1 = x_\mathrm{O} - x_\alpha\)
\(l_2 = x_\beta - x_\mathrm{O}\)
\(l_3 = x_\beta - x_\alpha\)
If
\(\ \ \ \ n_\mathrm{total} = n_\alpha + n_\beta\)
Then
\(\ \ \ \ n_\alpha l_1 = n_\beta l_2\)
\(\ \ \ \ \dfrac{n_\alpha}{n_\mathrm{total}} = \dfrac{l_2}{l_3}\)
\(\ \ \ \ \dfrac{n_\beta}{n_\mathrm{total}} = \dfrac{l_1}{l_3}\)
If \(\alpha\) is a liquid and \(\beta\) is a vapor, then
\(\mathcal{V} \equiv \dfrac{n_\beta}{n_\mathrm{total}} = \dfrac{l_1}{l_3}\) and \(\mathcal{L} \equiv \dfrac{n_\alpha}{n_\mathrm{total}} = \dfrac{l_2}{l_3}\)
Example
User Rune.welsh on en.wikipedia, CC BY-SA 3.0,
via Wikimedia Commons
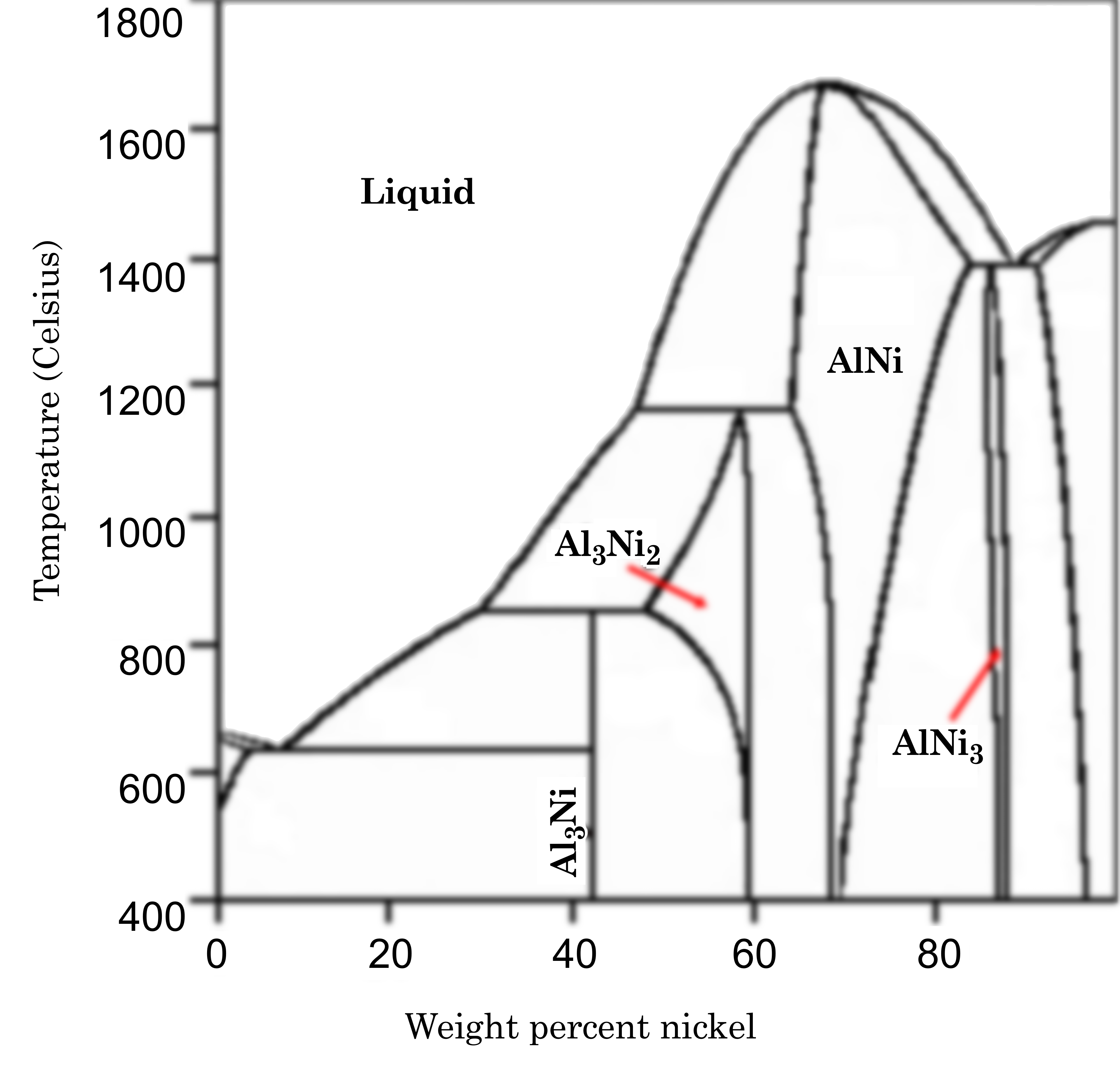
We have an equilibrium mixture of aluminum and nickel at \(1000\ ^\circ\mathrm{C}\). \(50\%\) of the overall mixture is nickel. Give the compositions and relative amounts of all phases present.
\(50\ \mathrm{mole}\% = \frac{50(58.6934)}{50(58.6934)+ 50(26.9815)} = 68.5\ \mathrm{mass}\%\)
Example Solution

User Rune.welsh on en.wikipedia, CC BY-SA 3.0,
via Wikimedia Commons
Atomic percent: 100% AlNi
Weight percent: Calc in 2-\(\phi\)
between Liquid and Al3Ni2
\(x_\mathrm{Liquid} = 36\%\)
\(x_\mathrm{Al_3Ni_2} = 57\%\)
\(\dfrac{n_\mathrm{Al_3Ni_2}}{n_\mathrm{O}} = \dfrac{x_\mathrm{Al_3Ni_2} - x_\mathrm{O}}{x_\mathrm{Al_3Ni2} - x_\mathrm{Liquid}} = \dfrac{57 - 50}{57 - 36} = 33.3\%\)
\(\dfrac{n_\mathrm{Liquid}}{n_\mathrm{O}} = 1 - \dfrac{n_\mathrm{Al_3Ni_2}}{n_\mathrm{O}} = 100\% - 33.3\% = 66.7\%\)
The Takeaways
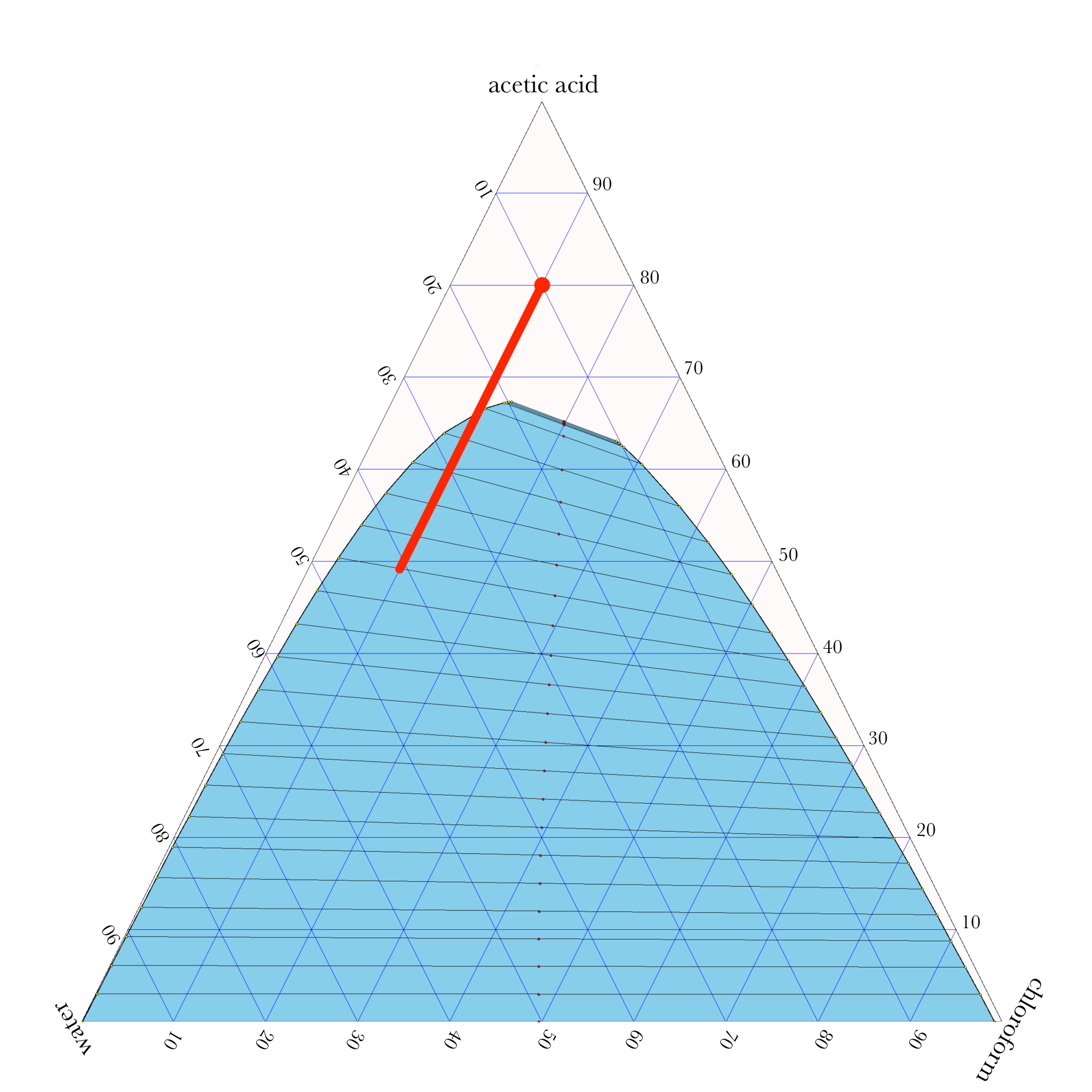
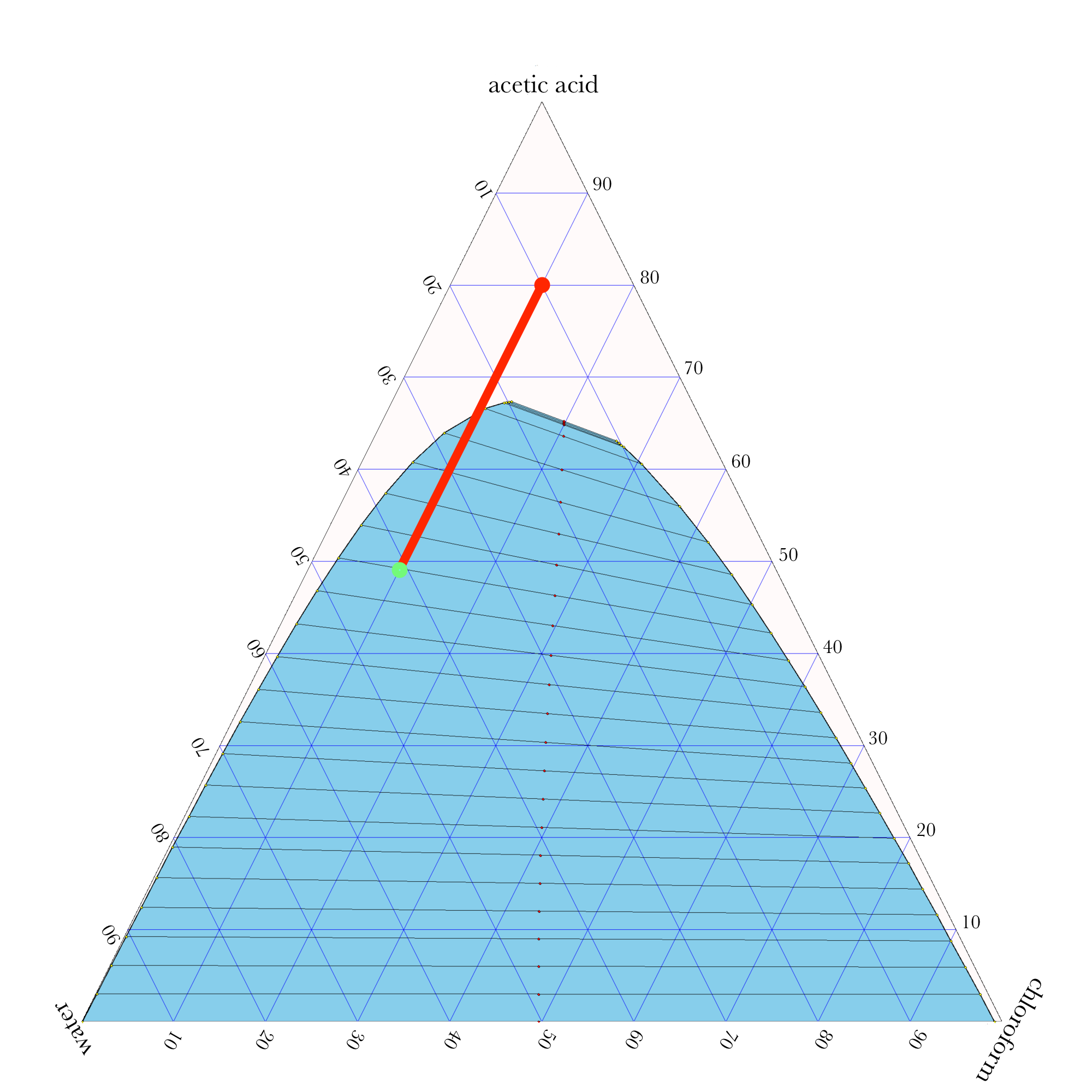
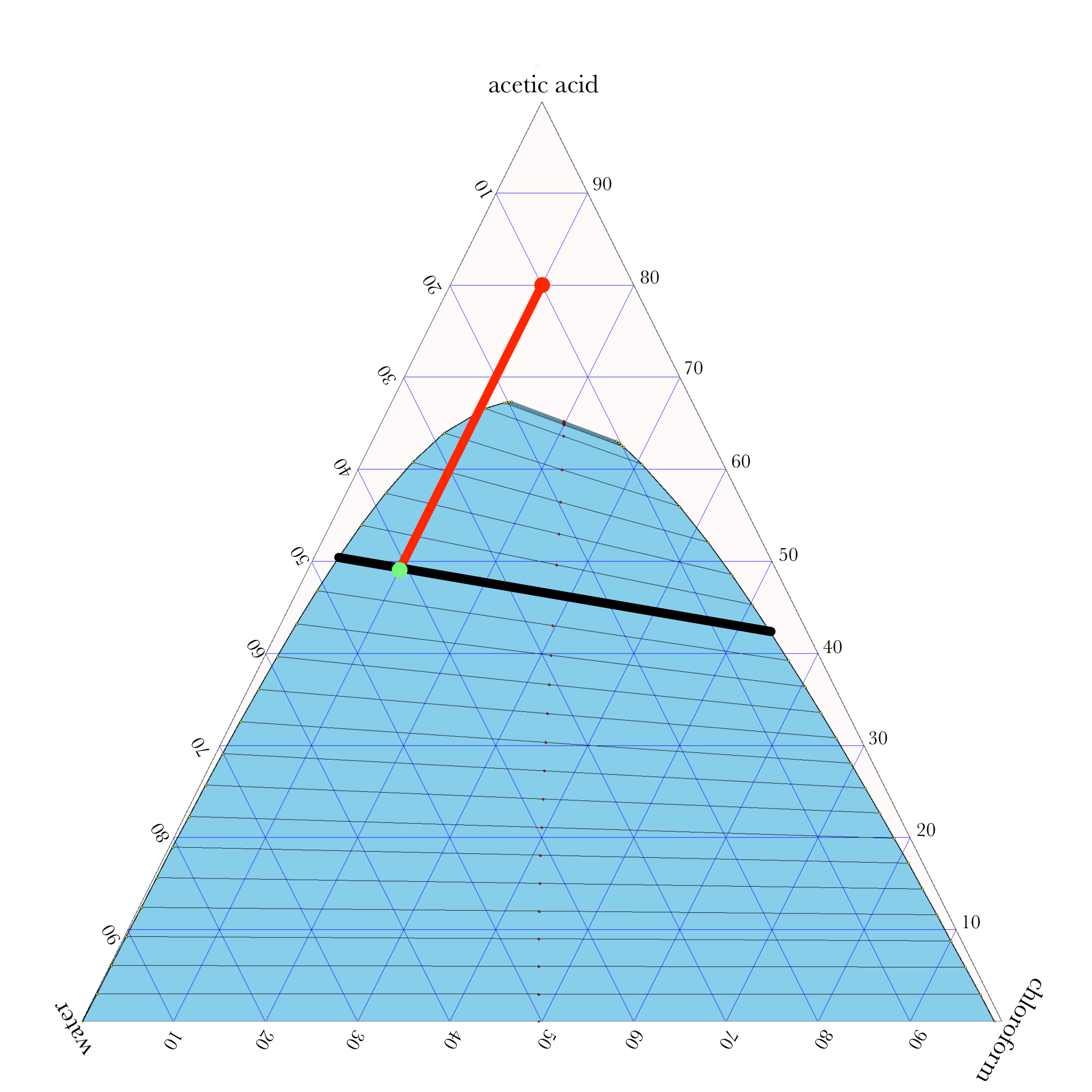
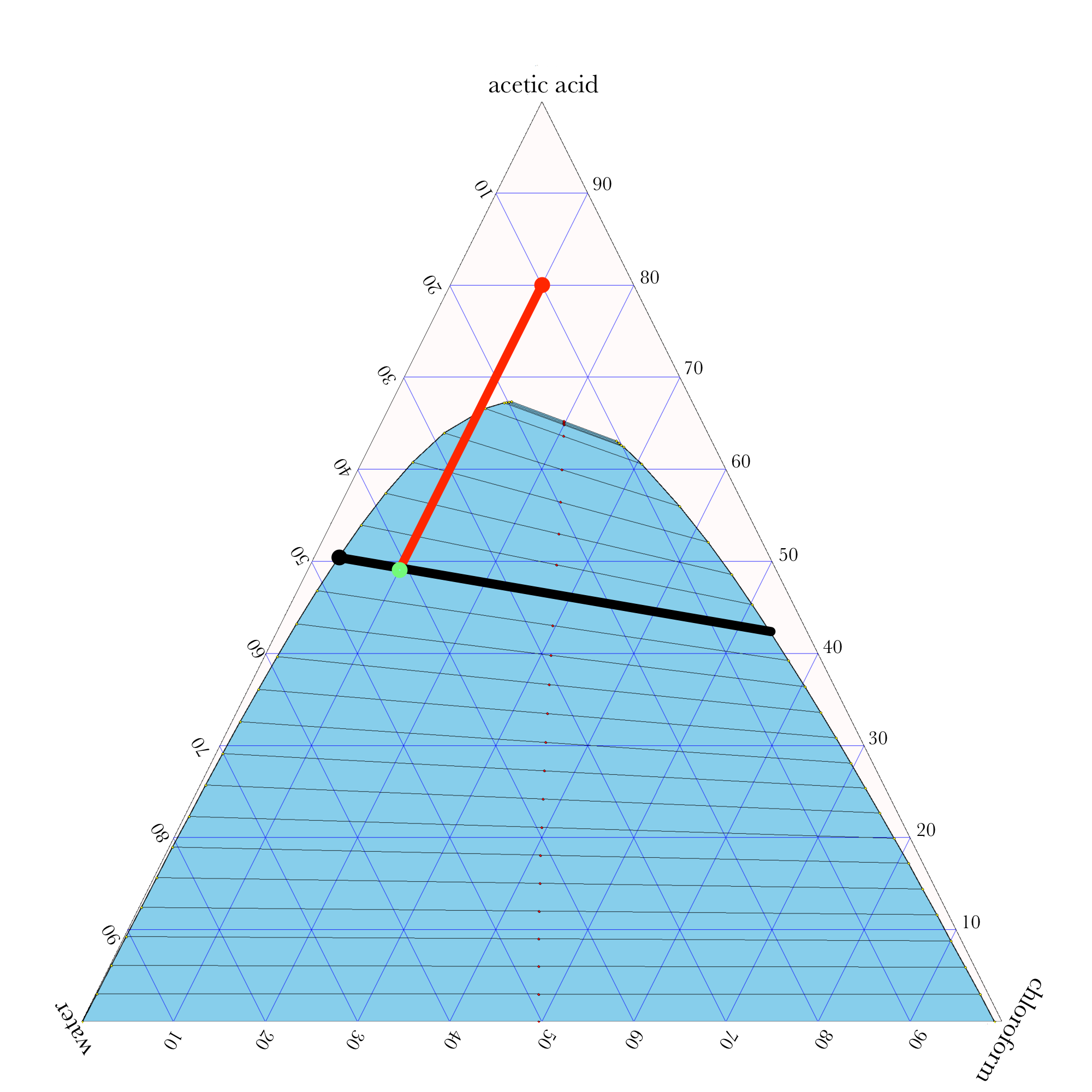
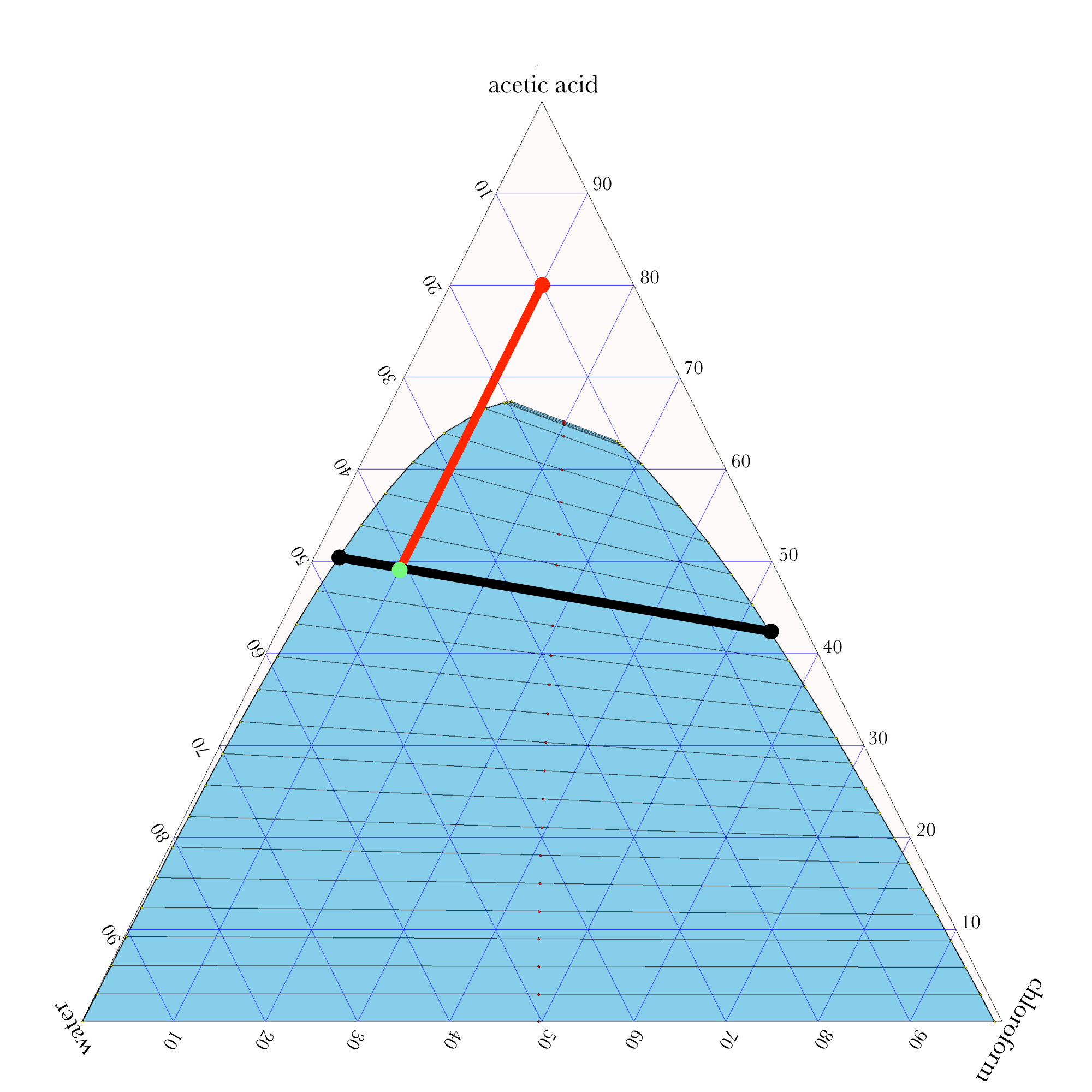
- In ternary phase diagrams, you read the composition by looking at composition lines that are normal to the species of interest. The mass or mole fractions have to add up to one or one hundred percent.
- The tie lines in a ternary diagram are not parallel like they are in a binary phase diagram.
- The lever rule can be used to calculate the relative amounts of the two phases.
Thanks for watching!
The previous video in the series is in the link in the upper left. The next video in the series is in the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.
The DOFPro Team

