Show Some Flash and Dew the Dew with a Little Bubbly!
DOFPro Team

Introduction
- The five most common vapor-liquid phase equilibrium calculations
- Performed on binary Txy and Pxy phase diagrams
- Bubble P (BUBL P)
- Dew P (DEW P)
- Bubble T (BUBL T)
- Dew T (DEW T)
- Flash (FLASH)
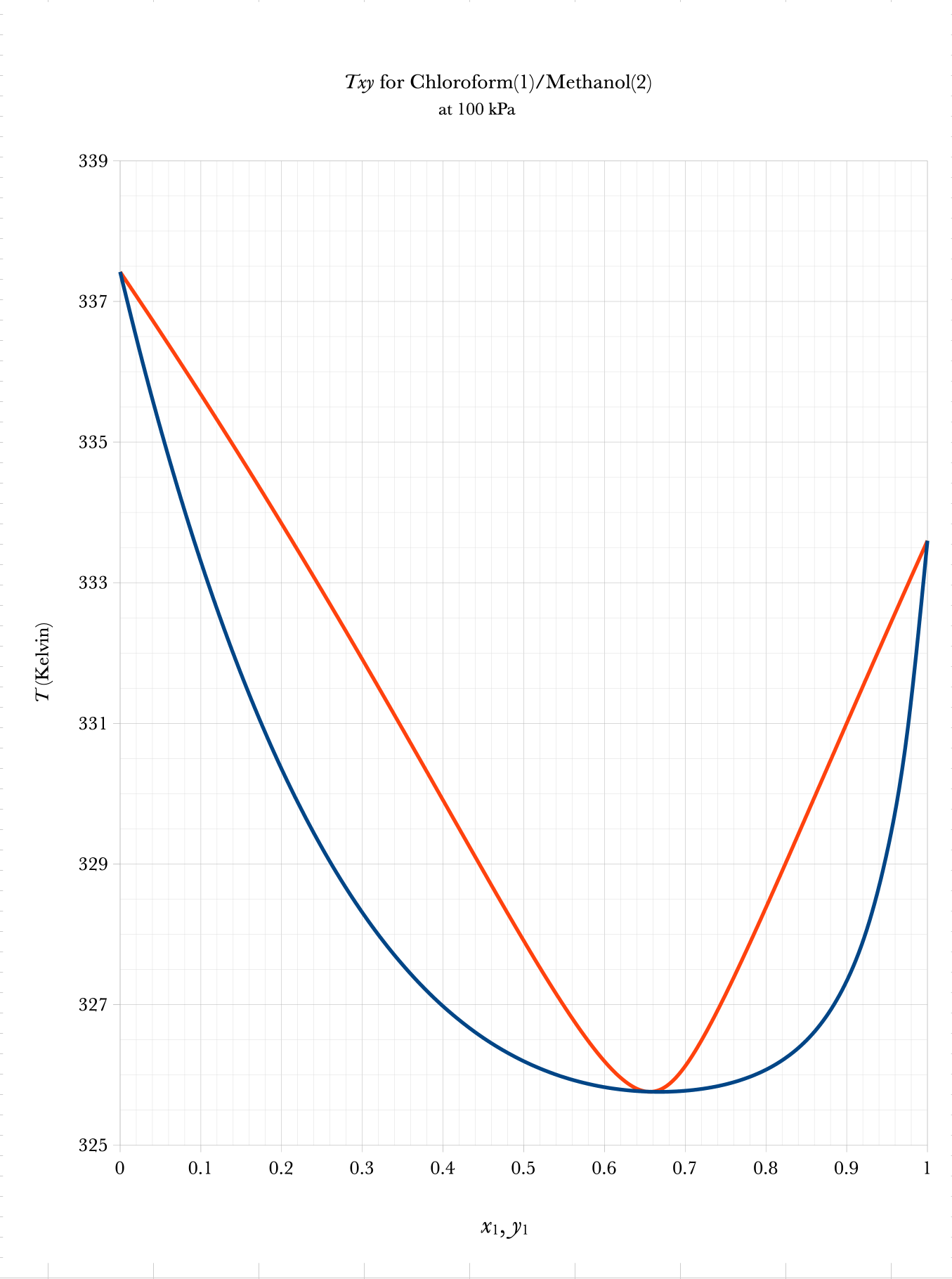
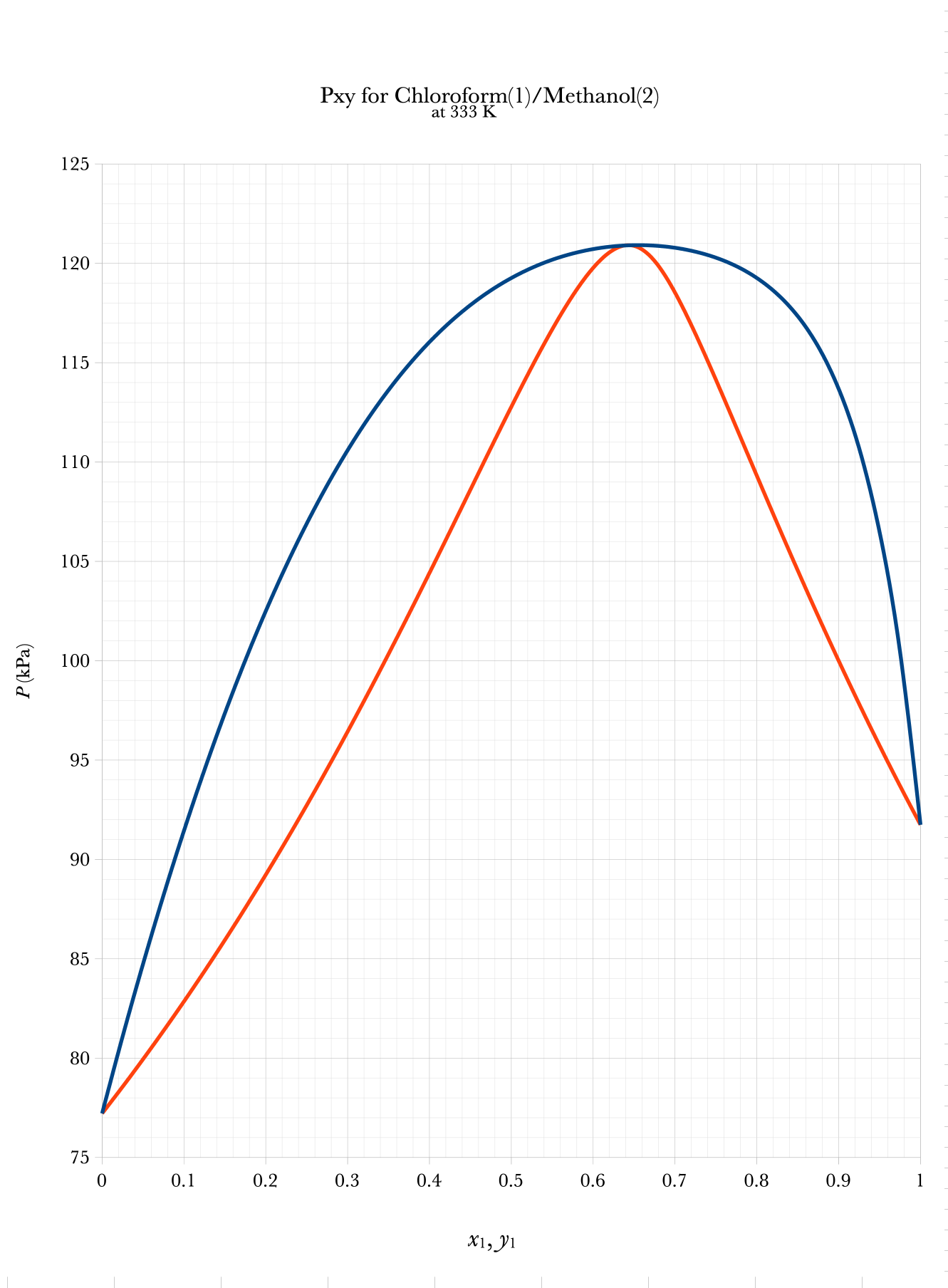
Txy and Pxy Binary Phase Diagrams
\(\mathrm{vapor}\)
\(2\text{–}\phi\)
\(\mathrm{liquid}\)
\(\mathrm{vapor}\)
\(2\text{–}\phi\)
\(\mathrm{liquid}\)
The Lever Rule for Phase Diagrams
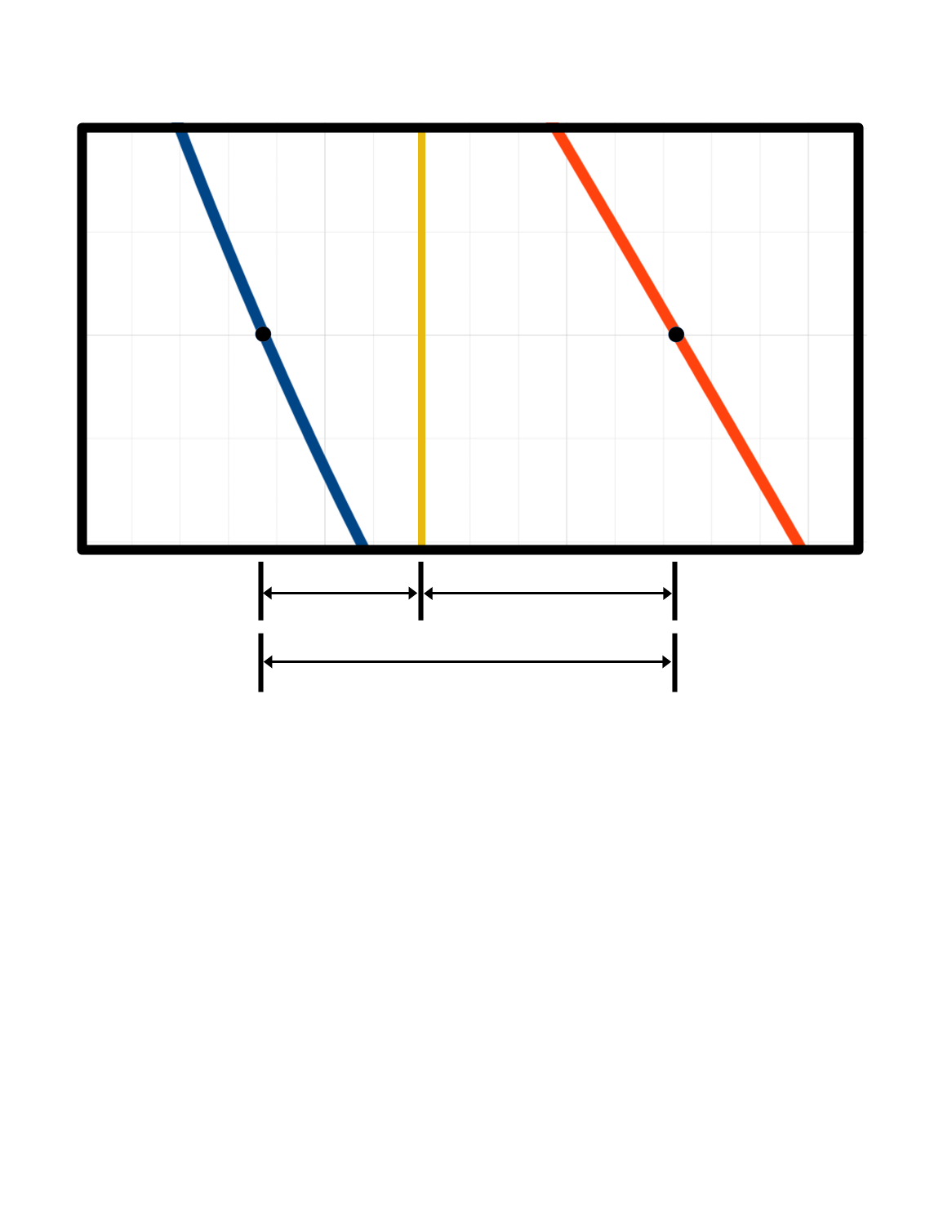
\(\text{Phase }\alpha\)
\(\text{Phase }\beta\)
\(2\text{–}\phi\)
\(x_\mathrm{O}\)
\(x_\alpha\)
\(x_\beta\)
\(l_1 = x_\mathrm{O} - x_\alpha\)
\(l_2 = x_\beta - x_\mathrm{O}\)
\(l_3 = x_\beta - x_\alpha\)
If
\(\ \ \ \ n_\mathrm{total} = n_\alpha + n_\beta\)
Then
\(\ \ \ \ n_\alpha l_1 = n_\beta l_2\)
\(\ \ \ \ \dfrac{n_\alpha}{n_\mathrm{total}} = \dfrac{l_2}{l_3}\)
\(\ \ \ \ \dfrac{n_\beta}{n_\mathrm{total}} = \dfrac{l_1}{l_3}\)
If \(\alpha\) is a liquid and \(\beta\) is a vapor, then
\(\mathcal{V} \equiv \dfrac{n_\beta}{n_\mathrm{total}} = \dfrac{l_1}{l_3}\) and \(\mathcal{L} \equiv \dfrac{n_\alpha}{n_\mathrm{total}} = \dfrac{l_2}{l_3}\)
BUBL P, BUBL T, DEW P, DEW T, and FLASH
For binary VLE mixtures, five types of calculations are usually performed:
- BUBL P – Given all \(x_i\)’s and \(T\), calculate \(P\) at which the first bubble forms, and \(y_i\)’s of the bubble.
- BUBL T – Given all \(x_i\)’s and \(P\), calculate \(T\) at which the first bubble forms, and \(y_i\)’s of the bubble.
- DEW P – Given all \(y_i\)’s and \(T\), calculate \(P\) at which the first droplet condenses, and \(x_i\)’s of the droplet.
- DEW T – Given all \(y_i\)’s and \(P\), calculate \(T\) at which the first droplet condenses, and \(x_i\)’s of the droplet.
- FLASH – Given overall composition (\(z_i\)’s) and \(T\) and \(P\), calculate \(x_i\)’s and \(y_i\)’s, and relative amounts of vapor, \(\mathcal{V}\), and liquid, \(\mathcal{L}\).
Graphical BUBL P, DEW P
Overall
Composition
Subcooled
Liquid
Superheated
Vapor
\(2\text{–}\phi\)
Bubble
Pressure
Bubble
Composition
Dew
Pressure
Dew
Composition
Low Boiling
Azeotrope
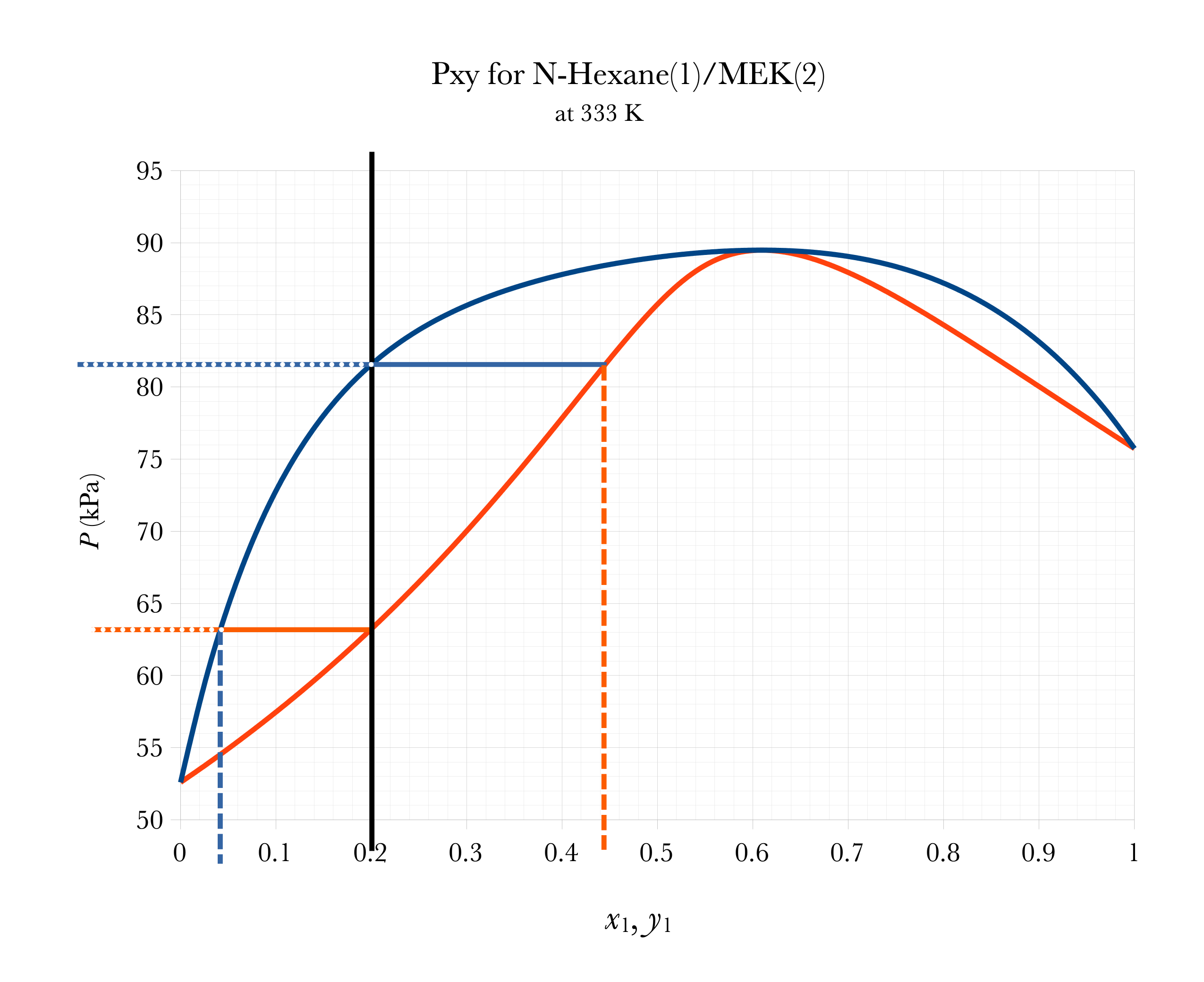
Graphical FLASH on Pxy
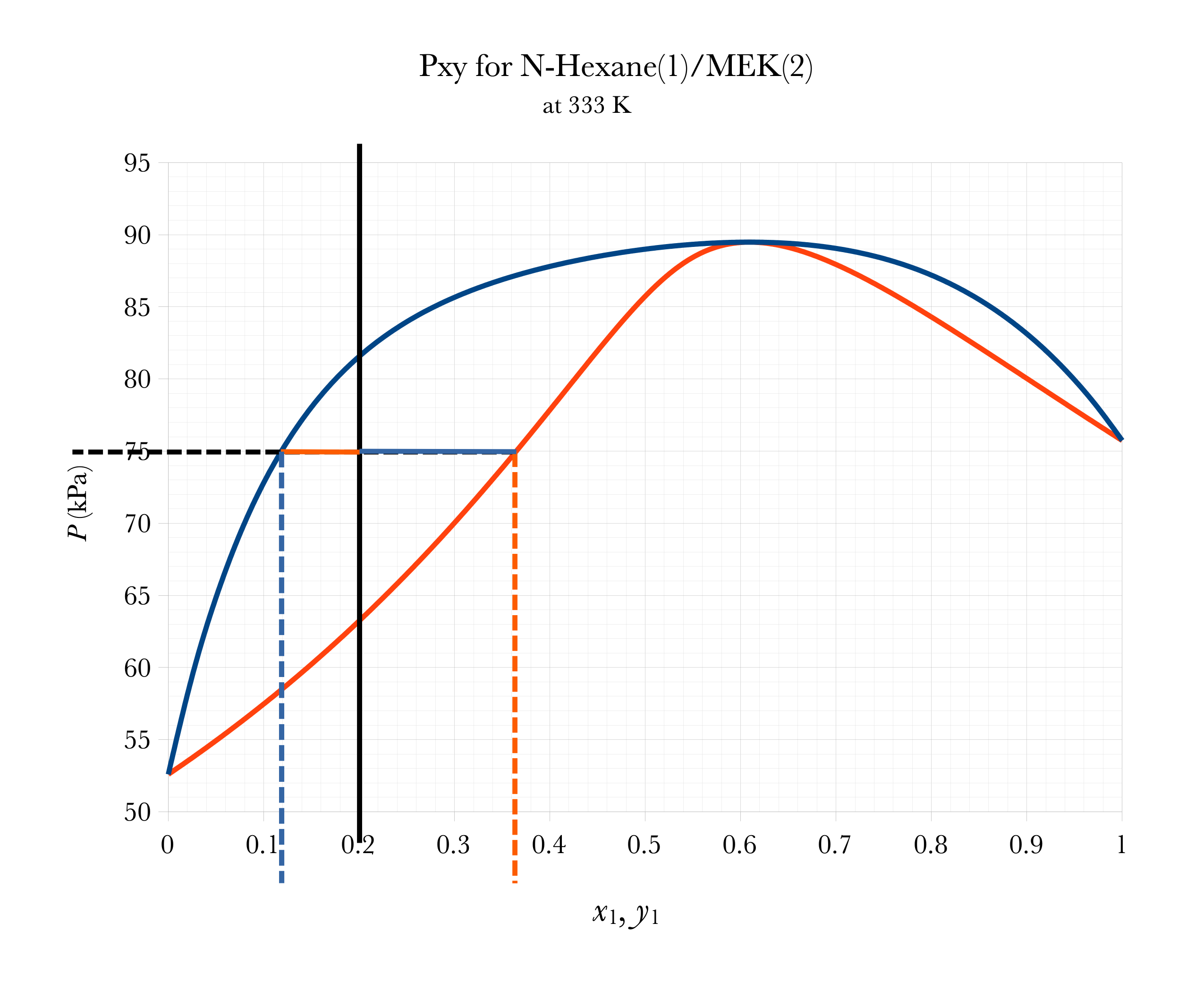
Overall
Composition
Subcooled
Liquid
Superheated
Vapor
\(2\text{–}\phi\)
Flash Temperature
Flash
Pressure
Liquid
Composition
Vapor
Composition
\(a\)
\(a \approx 0.20 - 0.12 = 0.08\)
\(b\)
\(b \approx 0.37-0.20 = 0.17\)
\(\mathcal{V} = \dfrac{a}{a+b} \approx 32\%\)
\(\mathcal{L} = 1-\mathcal{V} = \dfrac{b}{a+b} \approx 68\%\)
Graphical FLASH on Txy
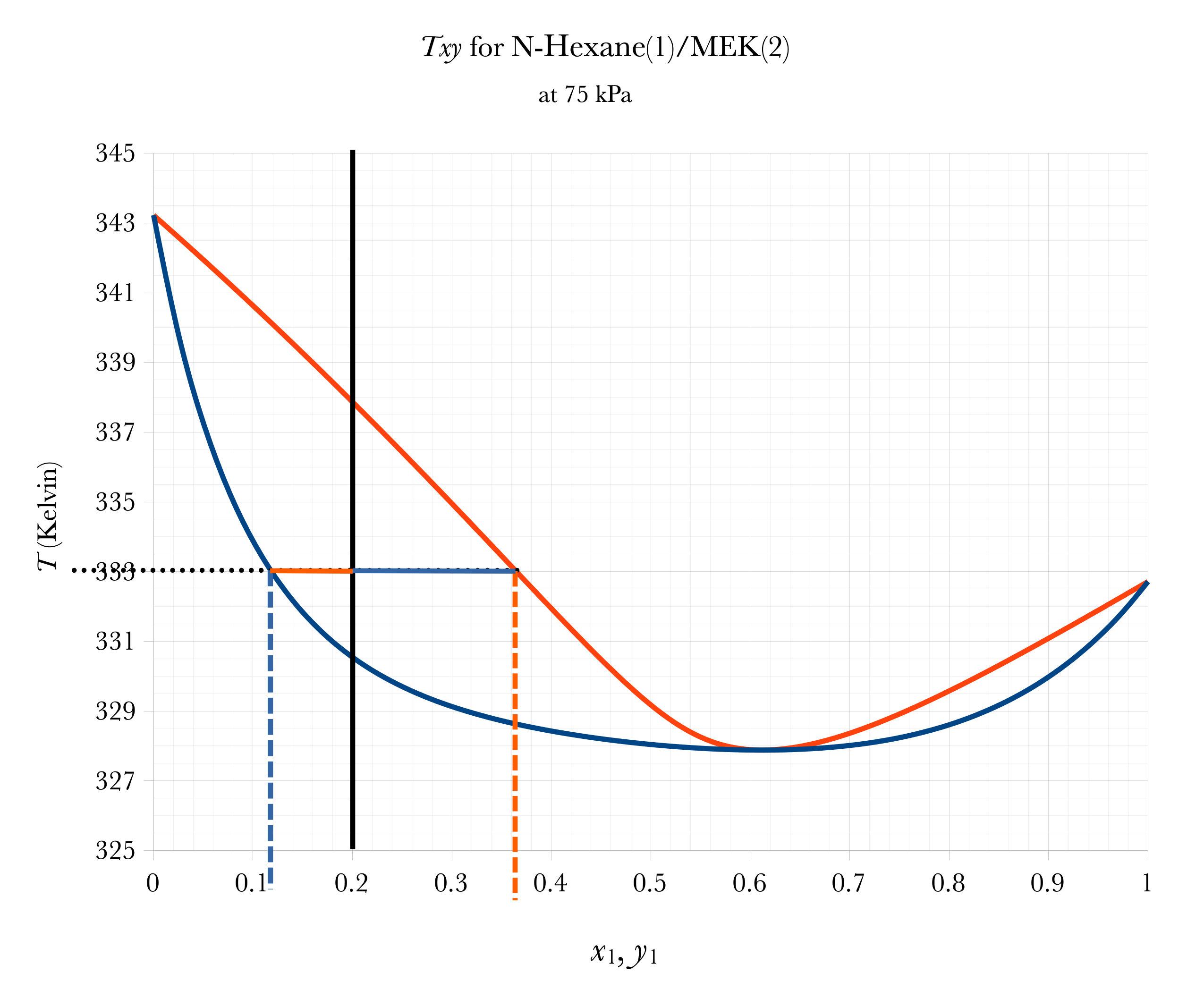
Overall
Composition
Subcooled
Liquid
Superheated
Vapor
\(2\text{–}\phi\)
Flash Pressure
Flash
Temperature
Liquid
Composition
Vapor
Composition
\(a\)
\(a \approx 0.20 - 0.12 = 0.08\)
\(b\)
\(b \approx 0.37-0.20 = 0.17\)
\(\mathcal{V} = \dfrac{a}{a+b} \approx 32\%\)
\(\mathcal{L} = 1-\mathcal{V} = \dfrac{b}{a+b} \approx 68\%\)
Graphical BUBL T, DEW T
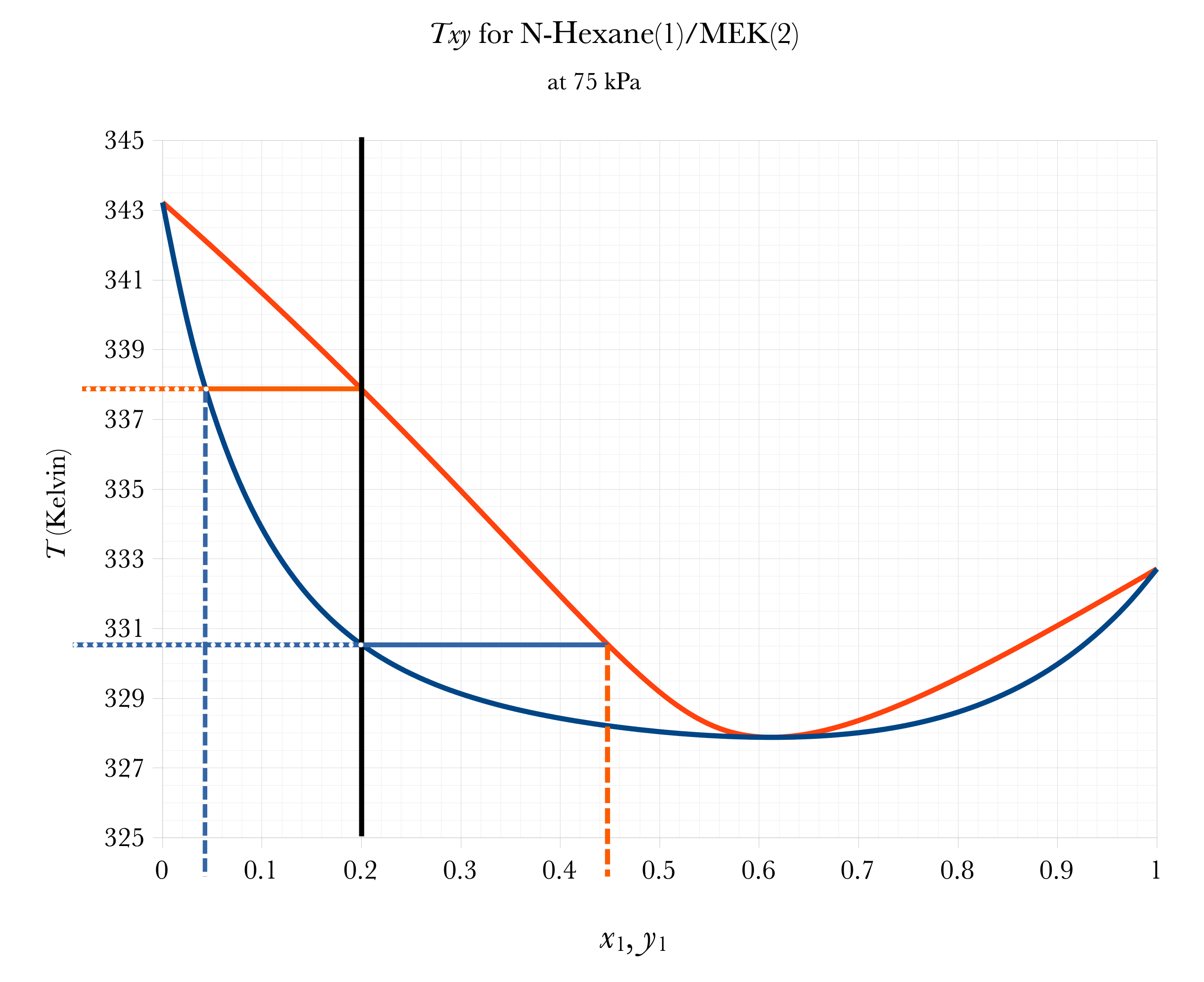
Overall
Composition
Subcooled
Liquid
Superheated
Vapor
\(2\text{–}\phi\)
Dew
Temperature
Dew
Composition
Bubble
Temperature
Bubble
Composition
Low Boiling
Azeotrope
The Takeaways
- A \(Pxy\) binary phase diagram can be used for Bubble P and Dew P graphical calculations.
- A \(Txy\) binary phase diagram can be used for Bubble T and Dew T graphical calculations.
- Either a \(Pxy\) or \(Txy\) binary phase diagram can be used for a Flash calculation, and when using the same \(T\) and \(P\) they should agree with each other.
- It is not easy to automate using graphical binary phase diagrams, and they don’t work for more than two species.
Thanks for watching!
The previous video is in the link in the upper left. The next video in the series, is in the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.

