Dew Your Bubbles Have Flash Part 3
DOFPro Team

Dew Your Bubbles Have Flash?
- Use Raoult’s law to perform vapor-liquid phase equilibria calculations.
- Not limited to binaries
- Multicomponent systems conforming to Raoult’s law
- Aliphatic hydrocarbon mixtures
- Aromatic hydrocarbon mixtures
- Part 1 – The basics and BUBL P and DEW P
- Part 2 – BUBL T and DEW T
- Part 3 – FLASH and Pxy and Txy diagrams
Graphical FLASH on Pxy
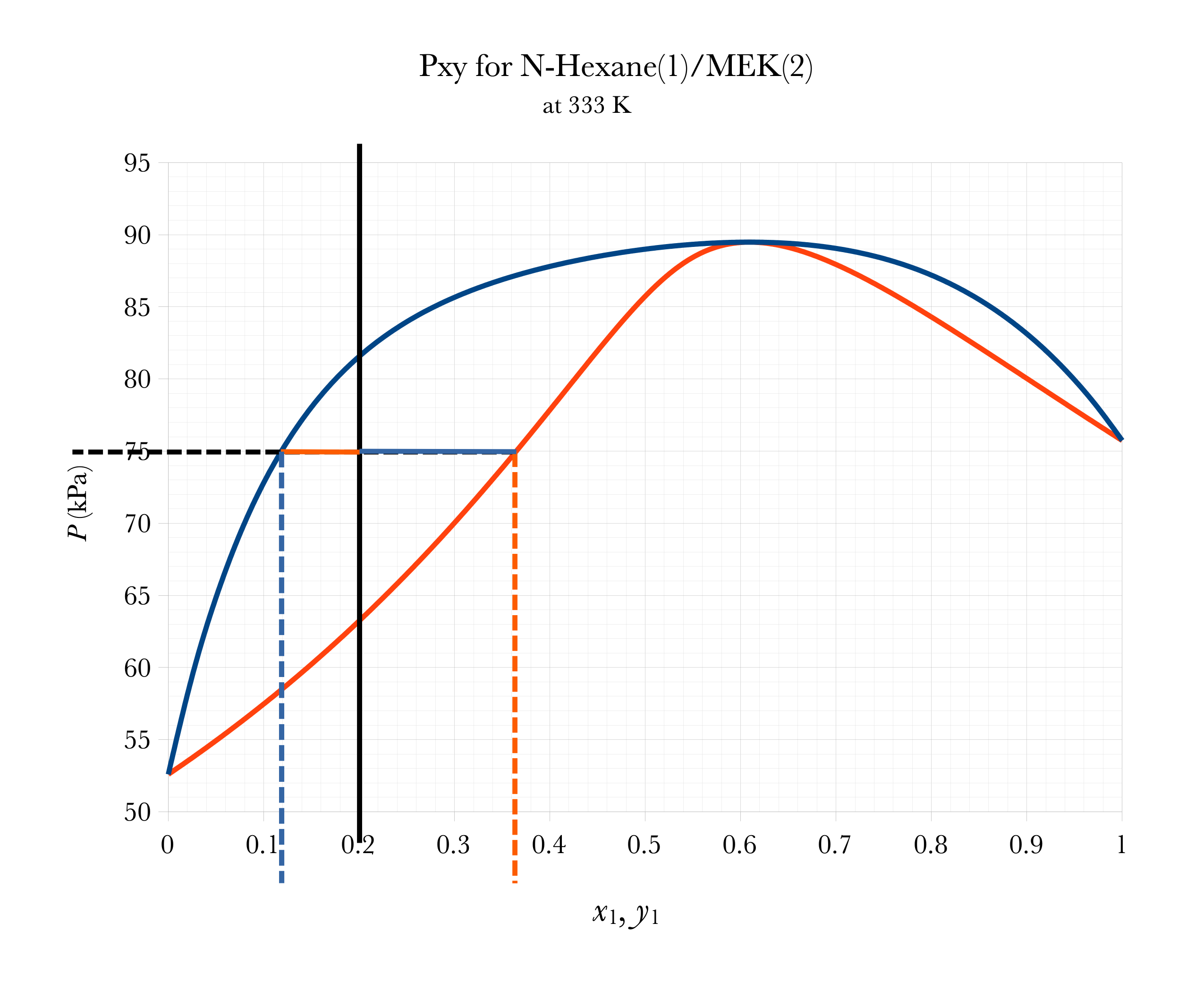
Overall
Composition
Subcooled
Liquid
Superheated
Vapor
\(2\text{–}\phi\)
Flash Temperature
Flash
Pressure
Liquid
Composition
Vapor
Composition
\(a\)
\(b\)
\(\mathcal{V} = \dfrac{a}{a+b}\)
\(\mathcal{L} = 1-\mathcal{V} = \dfrac{b}{a+b}\)
FLASH
Given \(z_i\)’s and \(T\) and \(P\), calculate \(x_i\)’s and \(y_{i}\)’s, and relative amounts of vapor, \(\mathcal{V}\), and liquid, \(\mathcal{L}\).
\[ \mathcal{V} \equiv \frac{n_\mathrm{{vapor}}}{n_\mathrm{{total}}} \tag{1}\]
and
\[ \mathcal{L} \equiv \frac{n_\mathrm{{liquid}}}{n_\mathrm{{total}}} \tag{2}\]
so
\[ \mathcal{L} + \mathcal{V} = 1 \tag{3}\]
and
\[ z_{i} = x_{i}\mathcal{L} + y_{i}\mathcal{V}\ \ \ (i=1, 2, 3, ..., N) \tag{4}\]
or
\[ z_{i} = x_{i}(1-\mathcal{V}) + y_{i}\mathcal{V}\ \ \ (i= 1, 2, 3, ..., N) \tag{5}\]
We will now use the K-value or distribution coefficient, defined as
\[ K_{i} \equiv \frac{p_{i}^{*}}{P} \tag{6}\]
which for Raoult’s law yields
\[ K_{i} = \frac{y_i}{x_i} \tag{7}\]
or
\[ x_{i} = \frac{y_i}{K_i} \tag{8}\]
Substitution into Equation 5 yields
\[ y_{i} = \frac{z_{i}K_{i}}{1+\mathcal{V} (K_{i} - 1) }\ \ \ (i = 1, 2, 3, ..., N) \tag{9}\]
or
\[ \sum_{i=1}^{N} \frac{z_{i}K_{i}}{1 + \mathcal{V} (K_{i} -1)} = 1 \tag{10}\]
which is solved for \(\mathcal{V}\) by numerical methods (such as Goal Seek). Then find the \(y_{i}\) from Equation 9 and the \(x_{i}\) from Equation 8.
For a binary, there is an analytical method for solving for \(\mathcal{L}\) and \(\mathcal{V}\). Discovering the method is left as an exercise to the viewer.
Example
The binary system benzene(1)/toluene(2) conforms closely to Raoult’s law. Vapor pressures for the pure species are given by the following Antoine equations:
\[ \log_{10} p_\mathrm{b}^{*} / \mathrm{bar} = 4.01814 - \frac{1203.835}{T /\mathrm{K} - 53.226} \]
\[ \log_{10}p_\mathrm{t}^{*} / \mathrm{bar} = 4.07827 - \frac{1343.943}{T / \mathrm{K} - 53.773} \]
Prepare a graph showing \(P\) vs. \(x_b\) and \(P\) vs. \(y_b\) for a temperature of 100°C.
Prepare a graph showing \(T\) vs. \(x_b\) and \(T\) vs. \(y_b\) for a pressure of 1 bar
For a \(Pxy\) diagram, we need \(P\) as a function of \(x_1\), and \(P\) as a function of \(y_1\), or \(x_1\) and \(y_1\) as a function of \(P\). From Equation 11
\[ P = \sum_{i=1}^{N} x_{i}p_{i}^{*} + (1-x_{i})p_{2}^{*} = (p_{1}^{*} - p_{2}^{*}) x_1 + p_{2}^{*} \]
which is a linear function of \(x_1\) that runs from \(p_{2}^{*}\) at \(x_1\) = 0 to \(p_{1}^{*}\) at \(x_1\) =1.
From Equation 12
\[ P = \frac{1}{\sum\limits_{i=1}^{N} \frac{y_i}{p_{1}^{*}}} = \frac{1}{\frac{y_1}{p_{1}^{*}} + \frac{1-y_1}{p_{2}^{*}}} = \frac{p_{1}^{*}p_{2}^{*}}{(p_{2}^{*} - p_{1}^{*}) y_1 + p_{1}^{}*} \]
\(p_{1}^{*}\) and \(p_{2}^{*}\) are determined from the Antoine equations.
\(P\) vs. \(x_1\) and \(P\) vs. \(y_1\) for a temperature of 100°C.
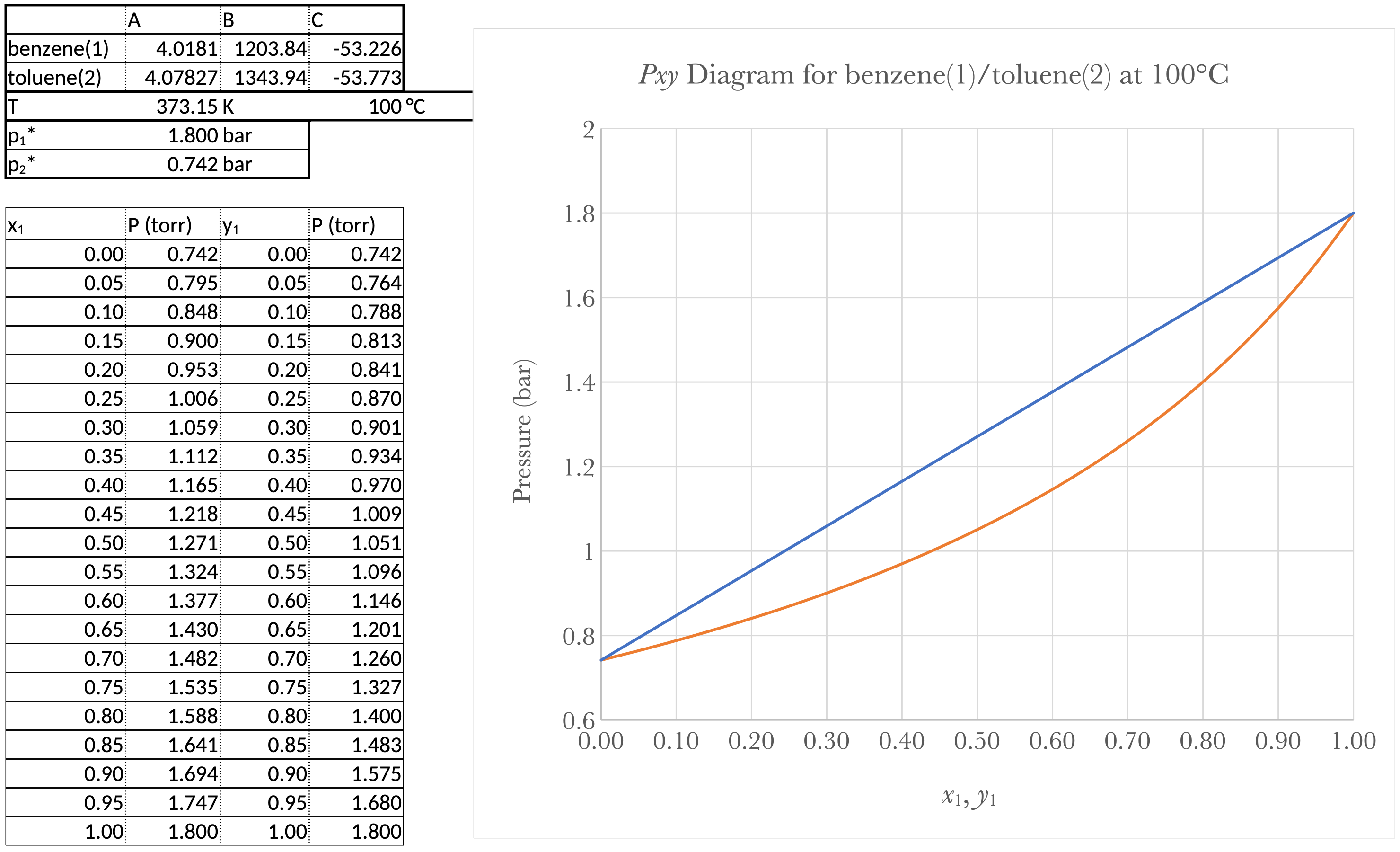
For a \(T_{xy}\) diagram, we either need to use Goal Seek or we need \(x_1\) and \(y_1\) as a function of \(T\). From From Equation 11
\[ P = (p_{1}^{*} - p_{2}^{*}) x_1 + p_{2}^{*} \]
\[ \Rightarrow x_1 = \frac{P-p_{2}^{*}}{p_{1}^{*} - p_{2}^{*}} \]
Pick a series of temperatures between \(T_\text{1-bp}\) and \(T_\text{2-bp}\) and calculate \(p_{1}^{*}\) and \(p_{2}^{*}\) and then \(x_1\).
From Equation 12
\[ P = \frac{p_{1}^{*}p_{2}^{*}}{(p_{2}^{*}-p_{1}^{*}) y_{1} + p_{1}^{*}} \]
\[ \Rightarrow y_1 = \frac{p_{1}^{*}}{P} \frac{P-p_{2}^{*}}{p_{1}^{*} - p_{2}^{*}} \]
Pick a series of temperatures between \(T_\text{1-bp}\) and \(T_\text{2-bp}\) and calculate \(p_{1}^{*}\) and \(p_{2}^{*}\) and then \(y_1\).
\(T\) vs. \(x_1\) and \(T\) vs. \(y_1\) for a pressure of 1 bar.
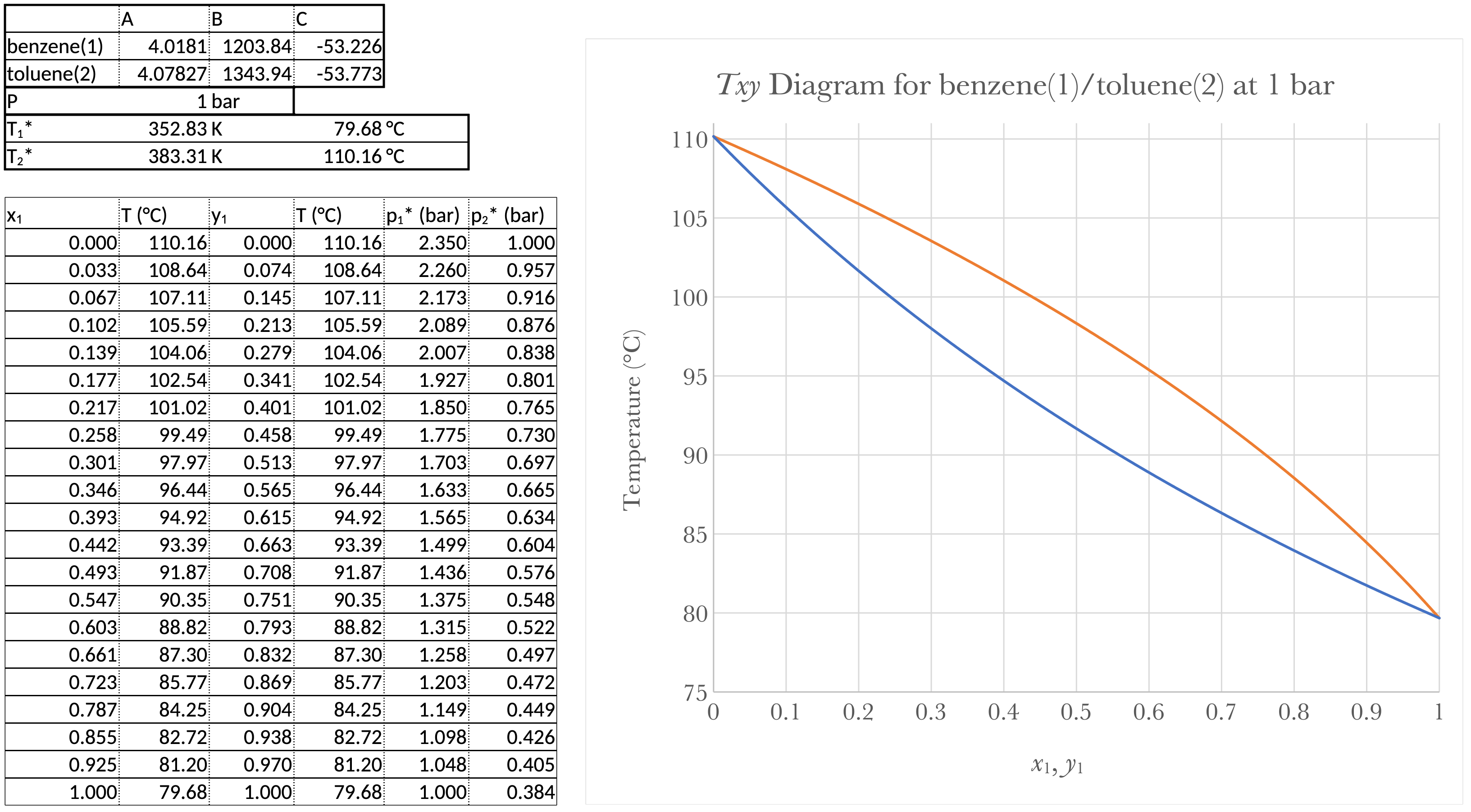
The Takeaways
- Vapor-Liquid Equilibria (VLE) calculations for binaries can be done with a Pxy or Txy phase diagram.
- For mixtures that are ideal solutions in the liquid phase and ideal gases in the vapor phase, VLE calculations can be done with Raoult’s law and the Antoine equation.
- The five classes of calculations from easiest to hardest are: Bubble P, Dew P, Bubble T, Dew T, and Flash.
- However, flash calculations for binary mixtures are relatively straightforward.
Thanks for watching!
The previous in the series video is the link in the upper left. The next video in the series is the link the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.

Equations for cross references
The originals are found in the Visuals for Part 1
\[ P = \sum_{n=1}^{N} x_{i}p_{i}^{*} \tag{11}\]
\[ P = \frac {1} {\sum\limits_{i=i}^{N}\frac{y_i} {p_{i}^{*}}} \tag{12}\]
