The Shocking Truth About Equilibrium Stages—Are You Doing It Wrong? Part 1
DOFPro Team

The Shocking Truth About Equilibrium Stages
Are You Doing It Wrong?
Part 1 – Principles and Equations
Part 2 - Two VLE examples
Part 3 - A Liquid-Liquid Example
- Explain the mass-balance equations for a single stage in a separator
- Provide three examples
- Most separation equipment has multiple stages.
- The overall split fraction is calculated by chaining together the individual stages.
- The full calculations require both mass balances and energy balances.
- These videos only describe the mass balances.
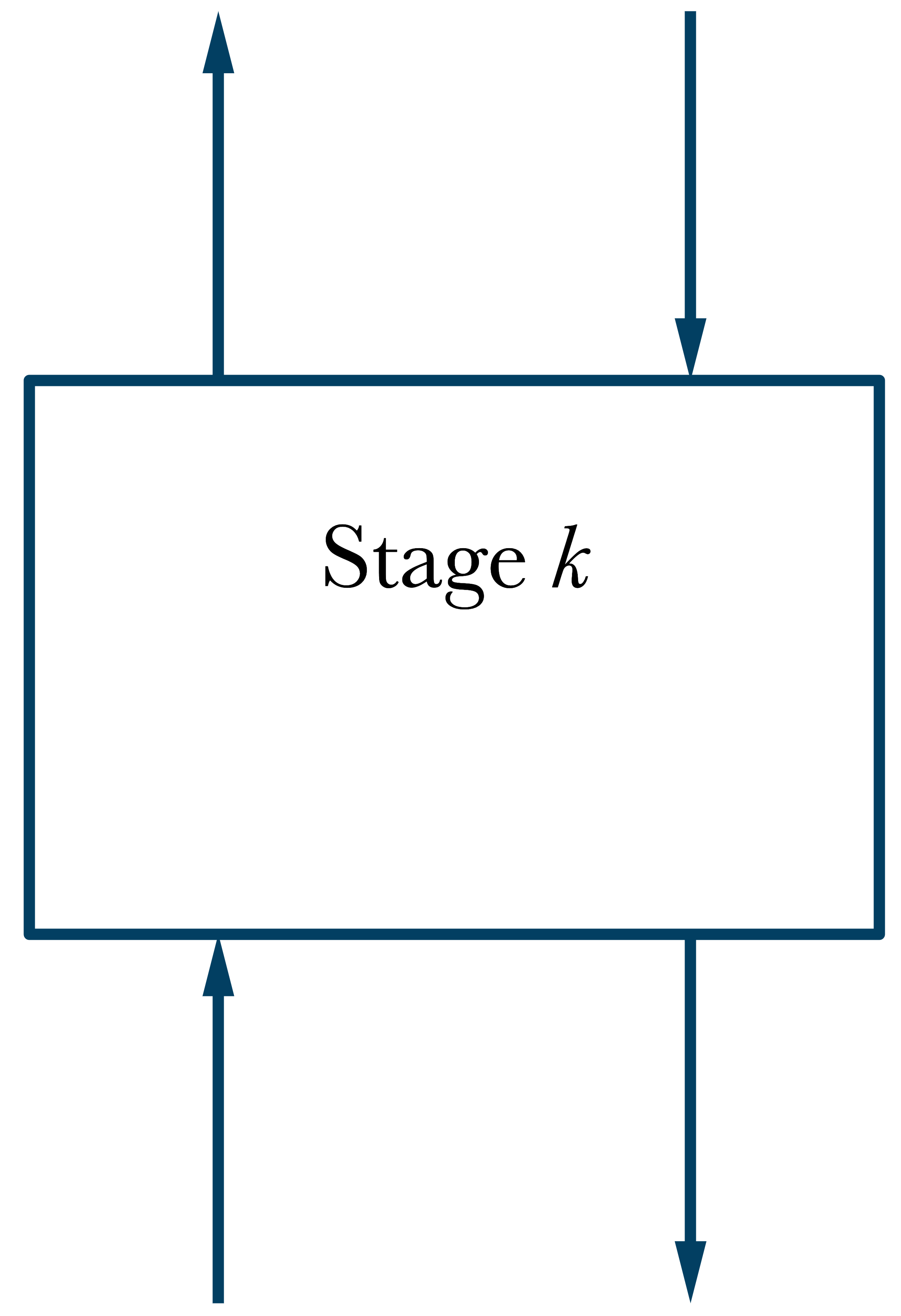
\(\dot{n}_{k-1}^2\)
\(\dot{n}_{k+1}^1\)
\(\dot{n}_{k}^2\)
\(\dot{n}_{k}^1\)
\(T_k,\ P_k\)
Mole Balance in General
\(\dot{n}_{k+1}^1 + \dot{n}_{k-1}^2 = \dot{n}_{k}^1 + \dot{n}_{k}^2 = \dot{n}_\mathrm{tot}\)
\(x_{i,k+1}\dot{n}_{k+1}^1 + y_{i,k-1}\dot{n}_{k-1}^2 = x_{i,k}\dot{n}_{k}^1 + y_{i,k}\dot{n}_{k}^2\)
\(z_{i,k} = \dfrac{{x_{i,k+1}}\dot{n}_{k+1}^1 + {y_{i,k-1}}\dot{n}_{k-1}^2}{(\dot{n}_{k+1}^1 + \dot{n}_{k-1}^2)\text{ or }\dot{n}_\mathrm{tot}} = \dfrac{{x_{i,k}}\dot{n}_{k}^1 + {y_{i,k}}\dot{n}_{k}^2}{(\dot{n}_{k}^1 + \dot{n}_{k}^2)\text{ or }\dot{n}_\mathrm{tot}}\)
\(\dfrac{\dot{n}_{k}^1}{{\dot{n}_\mathrm{tot}}} = \dfrac{\dot{n}_{k}^1}{\dot{n}_{k}^1 + \dot{n}_{k}^2}, \hspace{30mm}\dfrac{\dot{n}_{k}^2}{{\dot{n}_\mathrm{tot}}} = \dfrac{\dot{n}_{k}^2}{\dot{n}_{k}^1 + \dot{n}_{k}^2}\)
Phase Equilibria
Phase Diagrams \(\hspace{30mm}\) All at \(T_k\), \(P_k\)
Raoult’s law \(y_{i,k}P_k = x_{i,k}p_{i,k}^*\)

\(\dot{n}_{k-1}^v\)
\(\dot{n}_{k+1}^l\)
\(\dot{n}_{k}^v\)
\(\dot{n}_{k}^l\)
\(T_k,\ P_k\)
Mole Balance for Distillation
\(\dot{n}_{k+1}^l + \dot{n}_{k-1}^v = \dot{n}_{k}^l + \dot{n}_{k}^v = \dot{n}_\mathrm{tot}\)
\(x_{i,k+1}\dot{n}_{k+1}^l + y_{i,k-1}\dot{n}_{k-1}^v = x_{i,k}\dot{n}_{k}^l + y_{i,k}\dot{n}_{k}^v\)
\(z_{i,k} = \dfrac{{x_{i,k+1}}\dot{n}_{k+1}^l + {y_{i,k-1}}\dot{n}_{k-1}^v}{(\dot{n}_{k+1}^l + \dot{n}_{k-1}^v)\text{ or }\dot{n}_\mathrm{tot}} = \dfrac{{x_{i,k}}\dot{n}_{k}^l + {y_{i,k}}\dot{n}_{k}^v}{(\dot{n}_{k}^l + \dot{n}_{k}^v)\text{ or }\dot{n}_\mathrm{tot}}\)
\(\mathcal{L}_k = \dfrac{\dot{n}_{k}^l}{{\dot{n}_\mathrm{tot}}}, \hspace{30mm} \mathcal{V} = \dfrac{\dot{n}_{k}^v}{{\dot{n}_\mathrm{tot}}}\)
Phase Equilibria
Phase Diagrams \(\hspace{25mm}\) All at \(T_k\), \(P_k\)
Raoult’s law \(y_{i,k}P_k = x_{i,k}p_{i,k}^*\)

\(\dot{n}_{k-1}^v\)
\(\dot{n}_{k+1}^l\)
\(\dot{n}_{k}^v\)
\(\dot{n}_{k}^l\)
\(T_k,\ P_k\)
Types of Calculations
Given \(T_k\) and \(x_{i,k}\), find \(P_k\) and \(y_{i,k}\). BUBL P
Given \(P_k\) and \(x_{i,k}\), find \(T_k\) and \(y_{i.k}\). BUBL T
Given \(T_k\) and \(y_{i,k}\), find \(P_k\) and \(x_{i,k}\). DEW P
Given \(P_k\) and \(y_{i,k}\), find \(T_k\) and \(x_{i,k}\). DEW T
Given \(T_k\), \(P_k\), and \(z_{i,k}\), find \(\mathcal{L}_k\), \(\mathcal{V}_k\),
\(x_{i,k}\), and \(y_{i,k}\).
FLASH
Examples
- Distillation Column – Binary Phase Diagram
- Distillation Column – Raoult’s Law
- Liquid-Liquid Extraction – Ternary Phase Diagram
The Takeaways
- Equilibrium Separation Stages find common use in the chemical process industry.
- The mass balances are quite similar among the different types, and use the principles that have been explained in earlier videos.
- The principal differences are the means used to calculate the equilibrium compositions of the exiting streams.
Thanks for watching!
The previous video is in the link in the upper left. The next video in the series, is in the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.

