The Shocking Truth About Equilibrium Stages—Are You Doing It Wrong? Part 3
DOFPro Team

The Shocking Truth About Equilibrium Stages
Are You Doing It Wrong?
Part 1 – Principles and Equations
Part 2 - Two VLE examples
Part 3 - A Liquid-Liquid Example
- Explain the mass-balance equations for a single stage in a separator
- Provide three examples
- Most separation equipment has multiple stages.
- The overall split fraction is calculated by chaining together the individual stages.
- The full calculations require both mass balances and energy balances.
- These videos only describe the mass balances.
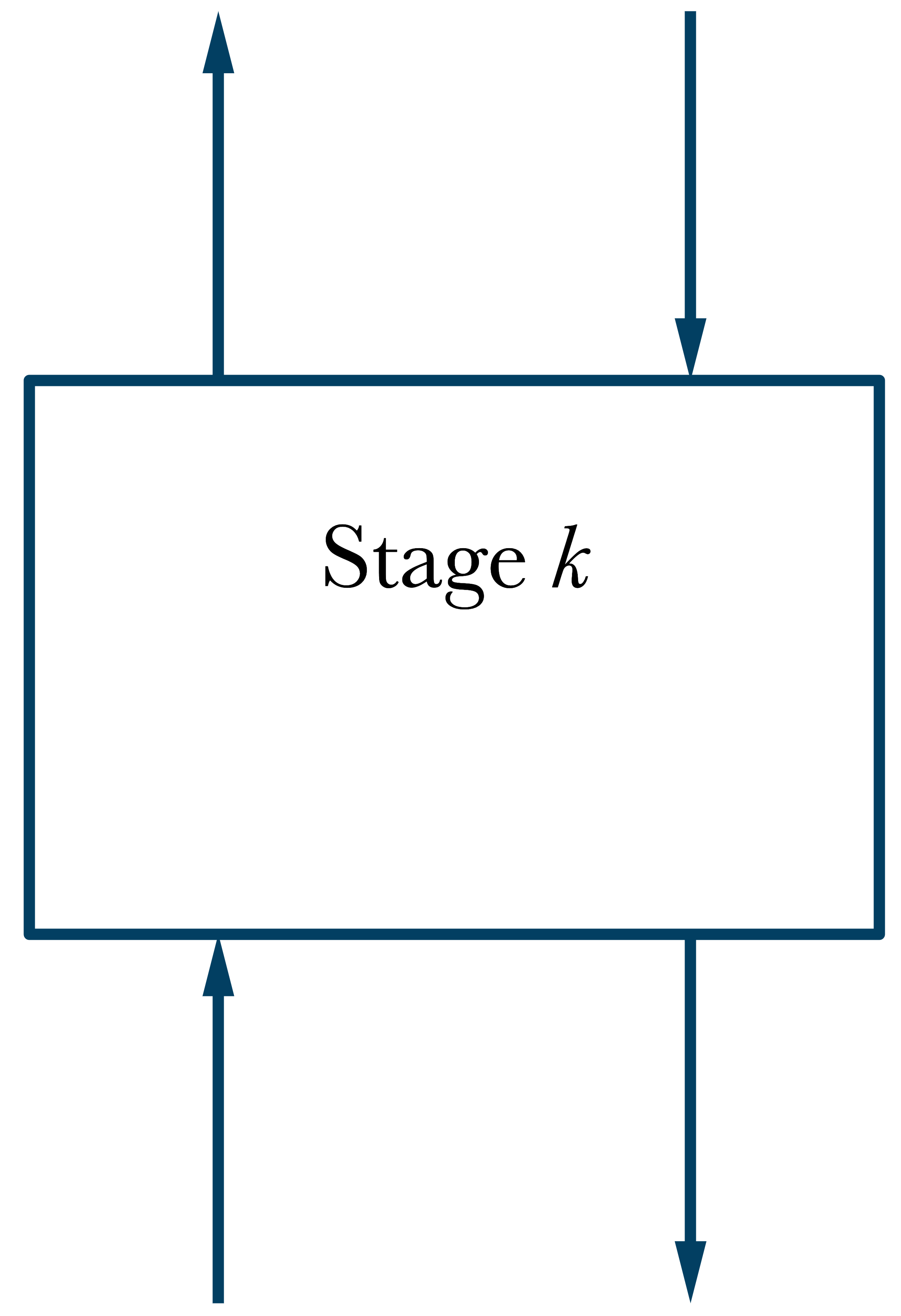
\(\dot{n}_{2}^{l_2}\)
\(\dot{n}_{1}^{l_1}\)
\(\dot{n}_{4}^{l_4}\)
\(\dot{n}_{3}^{l_3}\)
\(T_k,\ P_k\)
Example: Liquid-Liquid Extraction Phase Diagram
An equilibrium stage in a liquid-liquid extraction process has an equimolar feed of 50.0% water and 50.0% acetic acid and a flow rate of 180 mol/min. This feed needs to have most of the water removed. To this end, the other feed is 100% chloroform at 120 mol/min. The stage is operating at 298 K and 1 atm. Determine the flow rates and compositions of the product streams. How well does this first stage remove the water? The water-acetic acid-chloroform ternary phase diagram is given below.
\(\dot{n}_\mathrm{tot} = \dot{n}_1^{l_1} + \dot{n}_2^{l_2} = 180 + 120 = 300\ \mathrm{mol/min}\)
\(\dot{n}_{i,1}^{l_1} = x_i^{l_1} \dot{n}_1^{l_1} = 180 x_i^{l_1}\ \ \ \ (i=\mathrm{AA,\ CL,\ H_2O})\)
\(\dot{n}_{i,2}^{l_2} = x_i^{l_2} \dot{n}_2^{l_2} = 120 x_i^{l_2}\ \ \ \ (i=\mathrm{AA,\ CL,\ H_2O})\)
\(z_i = \dfrac{\dot{n}_{i,1}^{l_1} + \dot{n}_{i,2}^{l_2}}{\dot{n}_\mathrm{tot}}=\dfrac{180 x_i^{l_1} + 120 x_i^{l_2}}{300}\ \ \ \ (i=\mathrm{AA,\ CL,\ H_2O})\)
\(z_\mathrm{AA} = 0.300,\ \ \ \ \ \ z_\mathrm{CL} = 0.400,\ \ \ \ \ \ z_\mathrm{H2O} = 0.300\)
\(\mathcal{L}^3 = \dfrac{z_i - x_i^{l_4}}{x_i^{l_3} - x_i^{l_4}},\ \ \ \ \ \ \mathcal{L}^4 = 1 - \mathcal{L}^3\)
\(\text{Need }x_\mathrm{AA}^{l_3},\ x_\mathrm{CL}^{l_3},\ x_\mathrm{H_2O}^{l_3},\ x_\mathrm{AA}^{l_4},\ x_\mathrm{CL}^{l_4},\ x_\mathrm{H_2O}^{l_4}\)
Ternary Phase Diagram
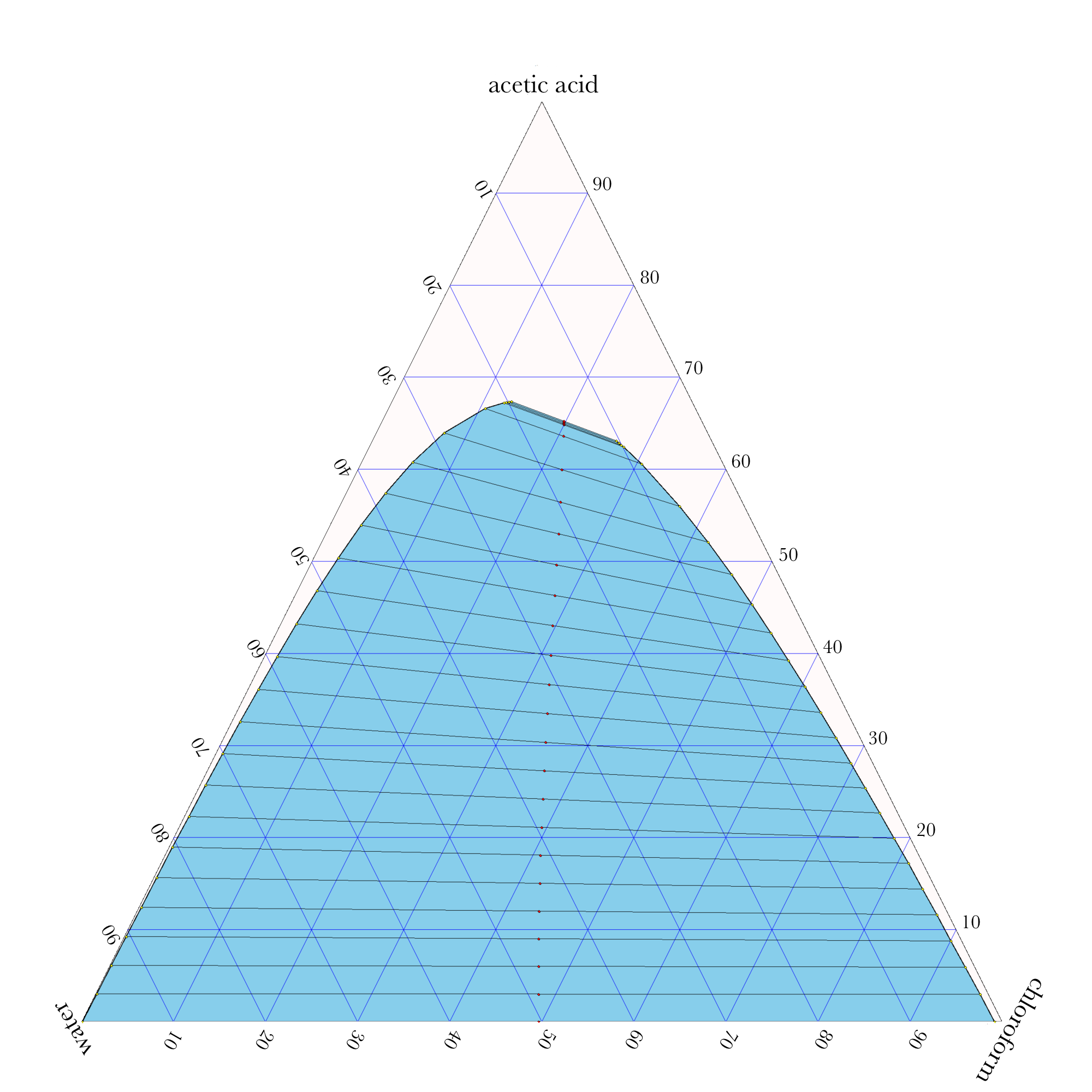
\((30.0\%,\ 40.0\%,\ 30.0\%)\)
•
Ternary Phase Diagram
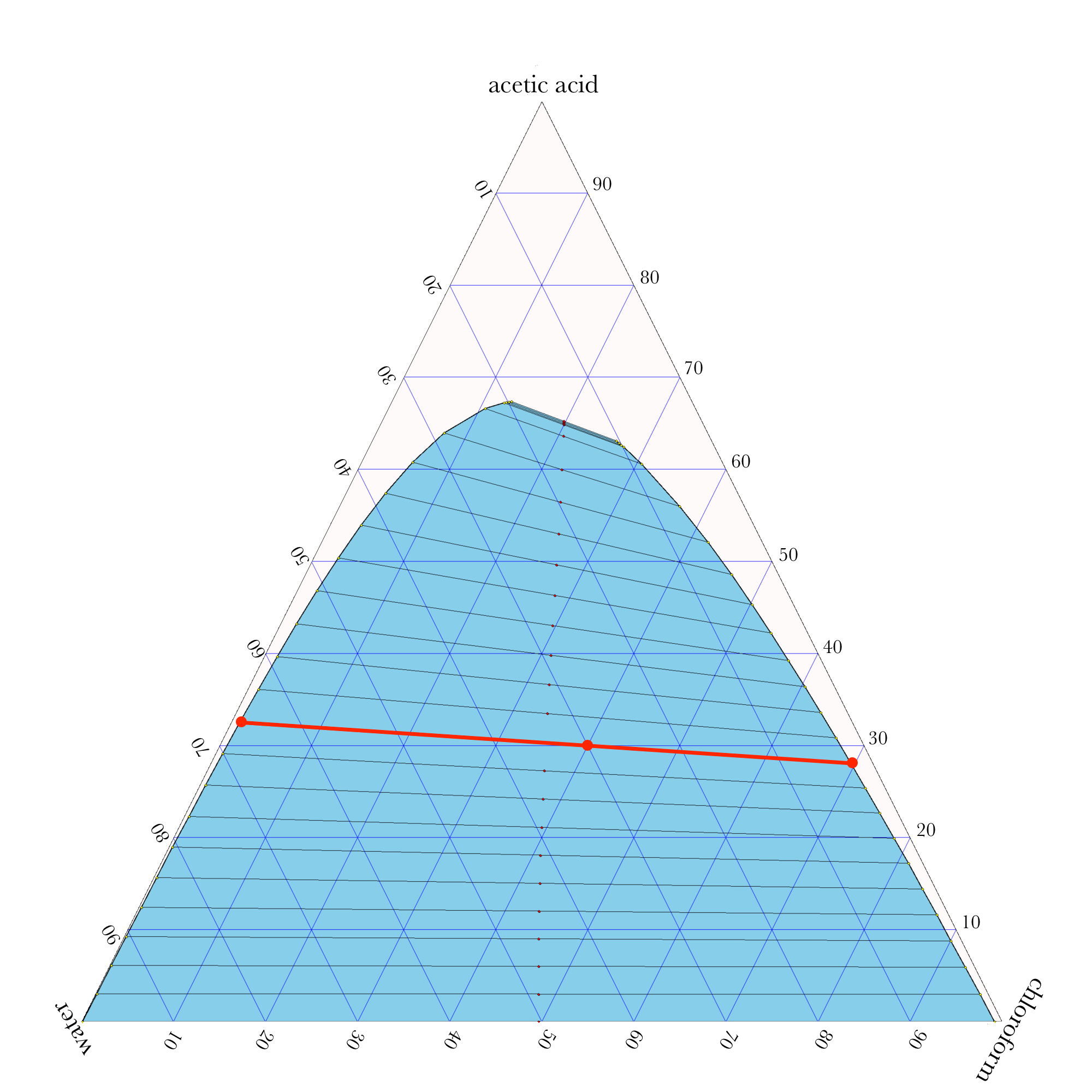
\((30.0\%,\ 40.0\%,\ 30.0\%)\)
\((32.6\%,\ 1.0\%,\ 66.4\%)\)
\((28.1\%,\ 69.6\%,\ 2.3\%)\)
\(z_\mathrm{AA} = 30.0\%,\ \ \ \ z_\mathrm{CL} = 40.0\%,\ \ \ \ z_\mathrm{H2O} = 30.0\%\)
\(x_\mathrm{AA}^{l_3} = 32.6\%,\ \ \ \ x_\mathrm{CL}^{l_3} = 1.0\%,\ \ \ \ x_\mathrm{H_2O}^{l_3} = 66.4\%\)
\(x_\mathrm{AA}^{l_4} = 28.1\%,\ \ \ \ x_\mathrm{CL}^{l_4} = 69.6\%,\ \ \ \ x_\mathrm{H_2O}^{l_4} = 2.3\%\)
The liquid-fraction values for each species were calculated with \(\mathcal{L}^3 = \dfrac{z_i - x_i^{l_4}}{x_i^{l_3} - x_i^{l_4}}\) and then averaged.
\(\mathcal{L}_\mathrm{mean}^3 = 43.1\%,\ \ \ \ \mathcal{L}_\mathrm{mean}^4 = 56.9\%\)
\(\dot{n}_3^{l_3} = \mathcal{L}_\mathrm{mean}^3 \dot{n}_\mathrm{tot} = 0.431 \mathbf{\cdot} 300 = 129\ \mathrm{mol/min}\)
\(\dot{n}_4^{l_4} = \mathcal{L}_\mathrm{mean}^4 \dot{n}_\mathrm{tot} = 0.569 \mathbf{\cdot} 300 = 171\ \mathrm{mol/min}\)
Solution Spreadsheet
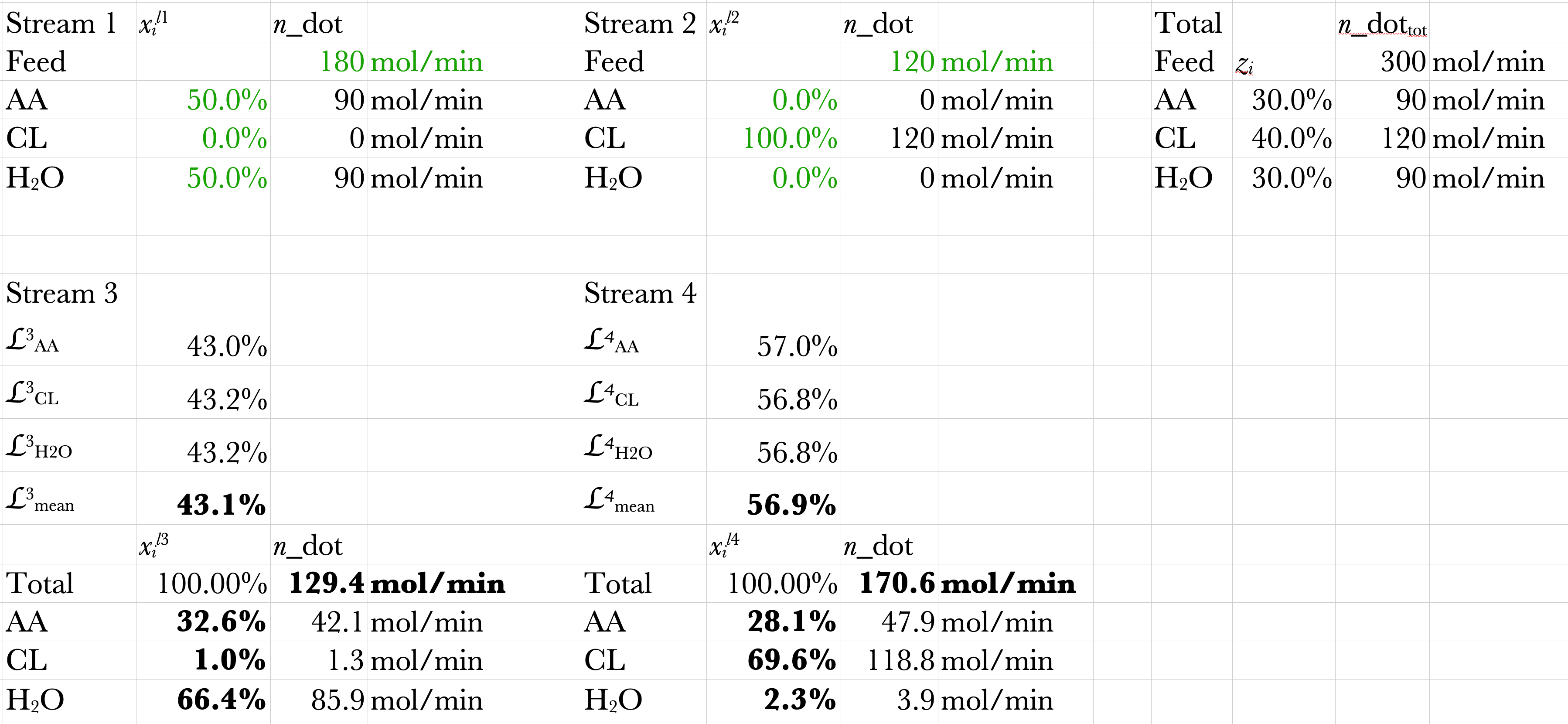
DWSIM Solutions
Here is the PFD and results of using DWSIM to solve this example problem.
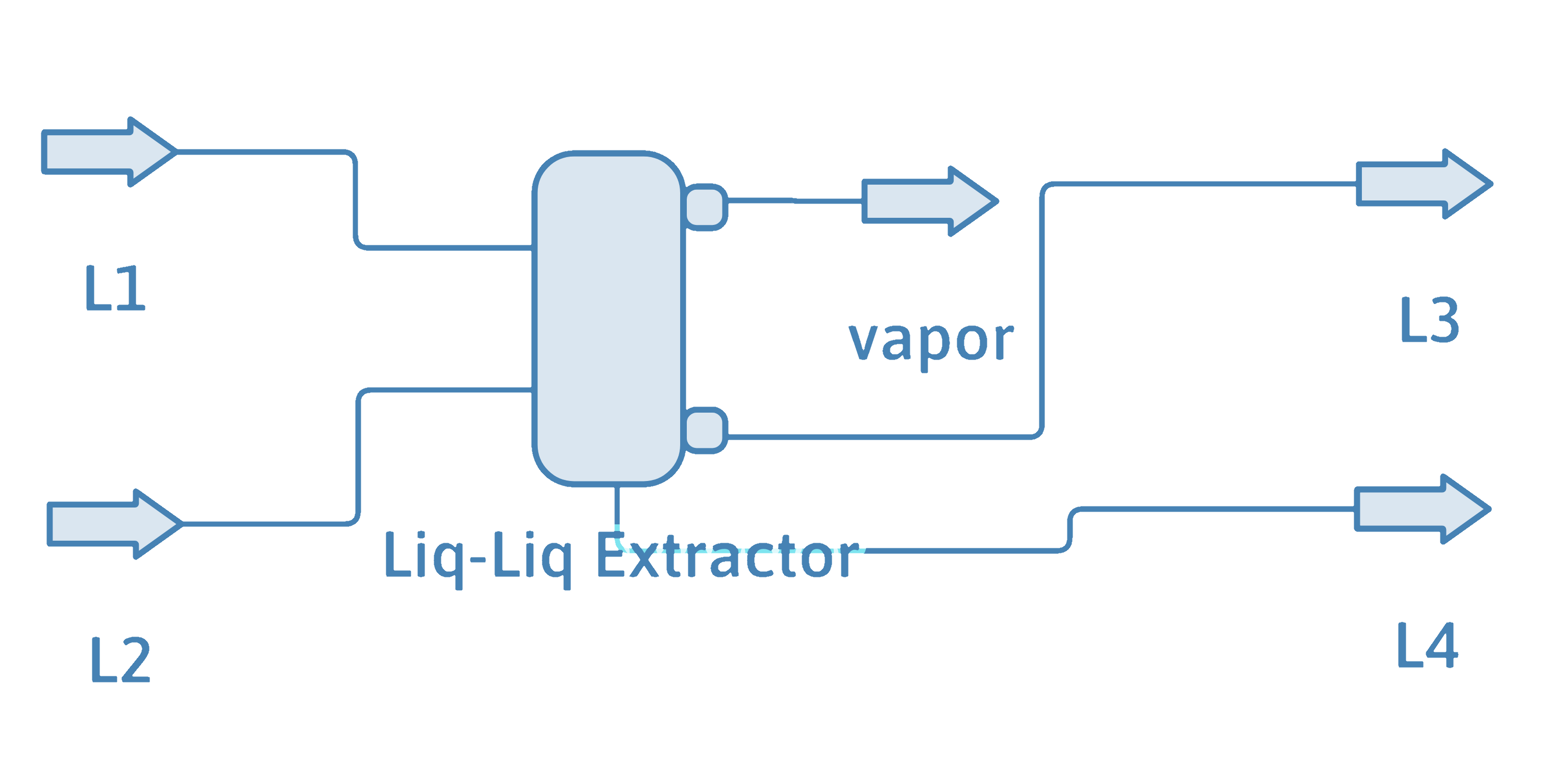
Stream 3
\(\dot{n} = 128.8\ \mathrm{mol/min}\)
\(x_\mathrm{AA} = 32.4\%\)
\(x_\mathrm{CL} = 0.92\%\)
\(x_\mathrm{H_2O} = 66.7\%\)
Stream 4
\(\dot{n} = 171.2\ \mathrm{mol/min}\)
\(x_\mathrm{AA} = 28.2\%\)
\(x_\mathrm{CL} = 69.4\%\)
\(x_\mathrm{H_2O} = 2.41\%\)
Calculation results for Material Stream L1
Compounds: { Acetic acid, Chloroform, Water, }
Specification: Temperature and Pressure
Temperature: 298.15 K
Pressure: 101325 Pa
Property Package: Modified UNIFAC (NIST)
Vapor Phase Molar Fraction 0
Liquid Phase 1 Molar Fraction 1
Liquid Phase 2 Molar Fraction 0
[Liquid Phase] Mass Flow 0.117101 kg/s
[Liquid Phase] Molar Flow 3 mol/s
[Liquid Phase] Volumetric Flow 0.00011491 m3/s
[Liquid Phase] Acetic acid Mole Frac 0.5
[Liquid Phase] Chloroform Mole Frac 0
[Liquid Phase] Water Mole Frac 0.5
[Liquid Phase] Acetic acid Mole Flow 1.5 mol/s [Liquid Phase] Chloroform Mole Flow 0 mol/s [Liquid Phase] Water Mole Flow 1.5 mol/s
Calculation results for Material Stream L2
Compounds: { Acetic acid, Chloroform, Water, }
Specification: Temperature and Pressure
Temperature: 298.15 K
Pressure: 101325 Pa
Property Package: Modified UNIFAC (NIST)
Vapor Phase Molar Fraction 0
Liquid Phase 1 Molar Fraction 1
Liquid Phase 2 Molar Fraction 0
[Liquid Phase] Mass Flow 0.238755 kg/s
[Liquid Phase] Molar Flow 2 mol/s
[Liquid Phase] Volumetric Flow 0.00016021 m3/s
[Liquid Phase] Acetic acid Mole Frac 0
[Liquid Phase] Chloroform Mole Frac 1
[Liquid Phase] Water Mole Frac 0
[Liquid Phase] Acetic acid Mole Flow 0 mol/s
[Liquid Phase] Chloroform Mole Flow 2 mol/s
[Liquid Phase] Water Mole Flow 0 mol/s
Calculation results for Material Stream L3
Compounds: { Acetic acid, Chloroform, Water, }
Specification: Pressure and Enthalpy
Pressure: 101325 Pa
Enthalpy: -1431.7079925153 kJ/kg
Property Package: Modified UNIFAC (NIST)
Stream Temperature 296.397 K
Stream Pressure 101325 Pa
Vapor Phase Molar Fraction 0
Liquid Phase 1 Molar Fraction 1
Liquid Phase 2 Molar Fraction 0
[Liquid Phase] Mass Flow 0.069894 kg/s
[Liquid Phase] Molar Flow 2.14627 mol/s
[Liquid Phase] Volumetric Flow 6.8864E-05 m3/s
[Liquid Phase] Acetic acid Mole Frac 0.323883
[Liquid Phase] Chloroform Mole Frac 0.00922527
[Liquid Phase] Water Mole Frac 0.666891
[Liq. Ph.] Acetic acid Mole Flow 0.695141 mol/s
[Liq. Ph.] Chloroform Mole Flow 0.01980 mol/s
[Liq. Ph.] Water Mole Flow 1.43133 mol/s
Calculation results for Material Stream L4
Compounds: { Acetic acid, Chloroform, Water, }
Specification: Pressure and Enthalpy
Pressure: 101325 Pa
Enthalpy: -361.464104380024 kJ/kg
Property Package: Modified UNIFAC (NIST)
Stream Temperature 296.397 K
Stream Pressure 101325 Pa
Vapor Phase Molar Fraction 0
Liquid Phase 1 Molar Fraction 1
Liquid Phase 2 Molar Fraction 0
[Liquid Phase] Mass Flow 0.285962 kg/s
[Liquid Phase] Molar Flow 2.85373 mol/s
[Liquid Phase] Volumetric Flow 0.00021699 m3/s
[Liquid Phase] Acetic acid Mole Frac 0.282037
[Liquid Phase] Chloroform Mole Frac 0.693899
[Liquid Phase] Water Mole Frac 0.0240638
[Liq. Ph.] Acetic acid Mole Flow 0.804859 mol/s
[Liq. Ph.] Chloroform Mole Flow 1.9802 mol/s
[Liq. Ph.] Water Mole Flow 0.0686717 mol/s
The Takeaways
- Liquid-Liquid extraction works very similarly to distillation stages and other equilibrium separation stages.
- The biggest challenge is getting and using the phase-equilibrium data.
- Using chemical-process-flowsheet software such as DWSIM removes most of the tedium from the process, if you know what you are doing.
Thanks for watching!
The previous video is in the link in the upper left. The next video in the series, is in the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.

