How to Have a Quickie with Your Data Part 1
DOFPro Team

Data Fitting Tools
- Statistics Videos much more complete
- Functions
average(values)andstdev.s(values)slope(y-values,x-values)andintercept(y-values,x-values)
- Solver
- Set up fitting function
- Set up array of fitting values. Keep magnitudes roughly equal.
- Calculate \(\Sigma{(y_i – y_{i\text{-fit}})^2}\). SSE
- Minimize with Solver
Demo for Average and Std. Dev.
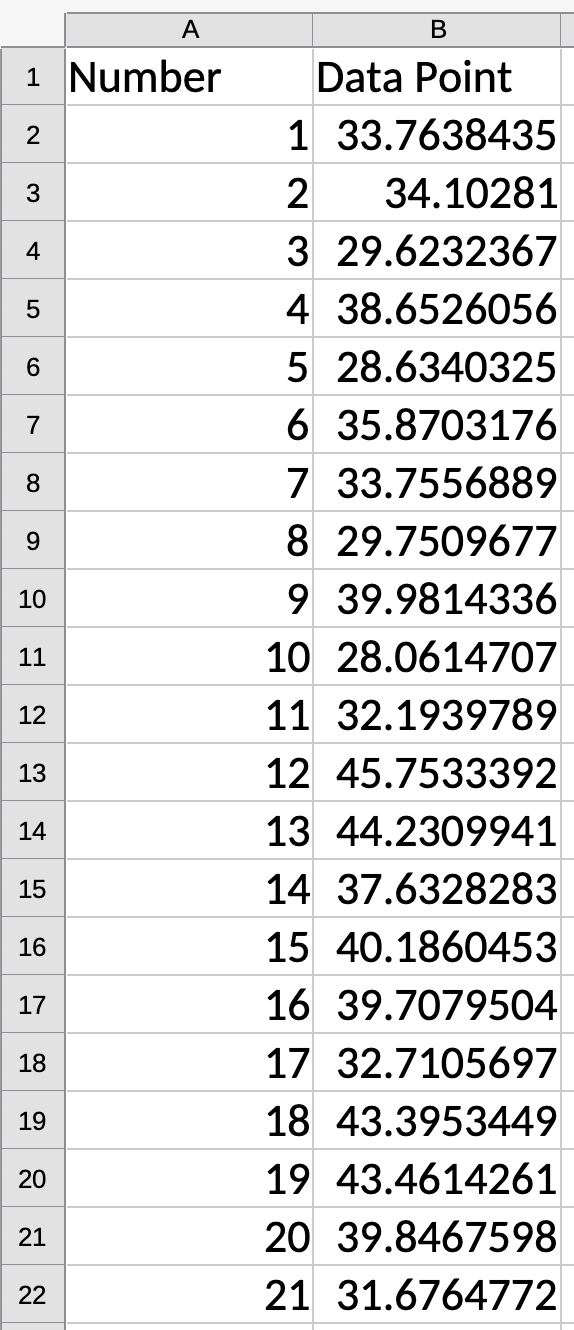
Demo for Slope and Intercept
Have data for \(2^\mathrm{nd}\)-order kinetics. Fit for \(C_{\mathrm{A}0}\), \(k\), \(k_0\), and \(E_A\).
\[C_\mathrm{A} = \dfrac{C_{\mathrm{A}0}}{1+C_{\mathrm{A}0} kt}\]
\[\dfrac{1}{\mathrm{C}_A} = \dfrac{1}{C_{\mathrm{A}0}} + kt\]
\[\mathrm{slope} = k\]
\[\mathrm{intercept} = \dfrac{1}{C_{\mathrm{A}0}}\]
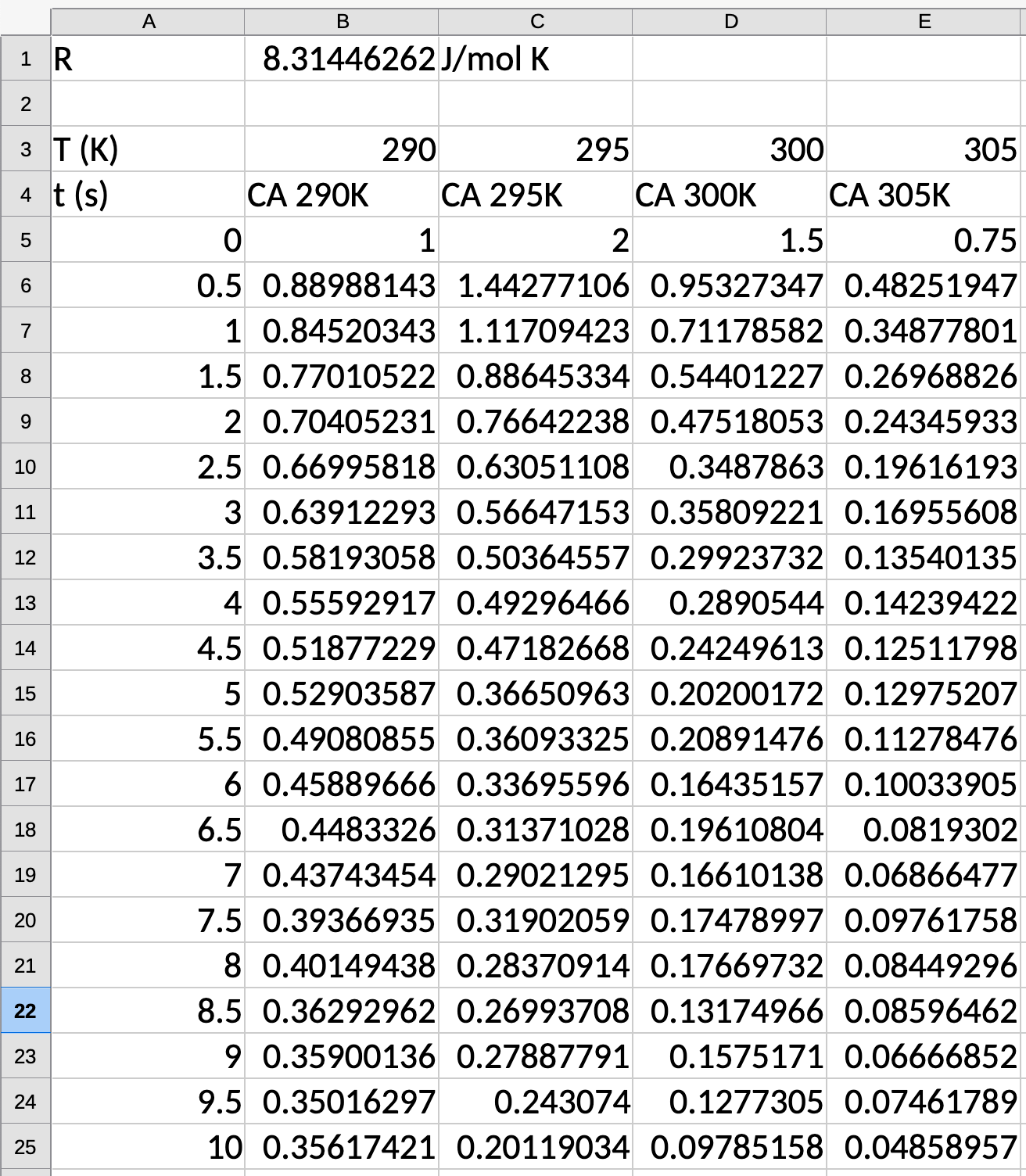
The Takeaways
- Spreadsheets have built-in functions to calculate the mean and the standard deviation of a set of data.
- Spreadsheets have built-in functions to calculate the slope and intercept of a set of paired data.
Thanks for watching!
The previous video in the series is the link in the upper left. The next video in the series is the link the upper right. To learn more about Chemical and Thermal Processes, visit the website linked in the description.

